Differential Mechanisms of Antianalgesia Induced by Endomorphin-1 and Endomorphin-2 in the Ventral Periaqueductal Gray of the Rat
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

NINDS Custom Collection II
ACACETIN ACEBUTOLOL HYDROCHLORIDE ACECLIDINE HYDROCHLORIDE ACEMETACIN ACETAMINOPHEN ACETAMINOSALOL ACETANILIDE ACETARSOL ACETAZOLAMIDE ACETOHYDROXAMIC ACID ACETRIAZOIC ACID ACETYL TYROSINE ETHYL ESTER ACETYLCARNITINE ACETYLCHOLINE ACETYLCYSTEINE ACETYLGLUCOSAMINE ACETYLGLUTAMIC ACID ACETYL-L-LEUCINE ACETYLPHENYLALANINE ACETYLSEROTONIN ACETYLTRYPTOPHAN ACEXAMIC ACID ACIVICIN ACLACINOMYCIN A1 ACONITINE ACRIFLAVINIUM HYDROCHLORIDE ACRISORCIN ACTINONIN ACYCLOVIR ADENOSINE PHOSPHATE ADENOSINE ADRENALINE BITARTRATE AESCULIN AJMALINE AKLAVINE HYDROCHLORIDE ALANYL-dl-LEUCINE ALANYL-dl-PHENYLALANINE ALAPROCLATE ALBENDAZOLE ALBUTEROL ALEXIDINE HYDROCHLORIDE ALLANTOIN ALLOPURINOL ALMOTRIPTAN ALOIN ALPRENOLOL ALTRETAMINE ALVERINE CITRATE AMANTADINE HYDROCHLORIDE AMBROXOL HYDROCHLORIDE AMCINONIDE AMIKACIN SULFATE AMILORIDE HYDROCHLORIDE 3-AMINOBENZAMIDE gamma-AMINOBUTYRIC ACID AMINOCAPROIC ACID N- (2-AMINOETHYL)-4-CHLOROBENZAMIDE (RO-16-6491) AMINOGLUTETHIMIDE AMINOHIPPURIC ACID AMINOHYDROXYBUTYRIC ACID AMINOLEVULINIC ACID HYDROCHLORIDE AMINOPHENAZONE 3-AMINOPROPANESULPHONIC ACID AMINOPYRIDINE 9-AMINO-1,2,3,4-TETRAHYDROACRIDINE HYDROCHLORIDE AMINOTHIAZOLE AMIODARONE HYDROCHLORIDE AMIPRILOSE AMITRIPTYLINE HYDROCHLORIDE AMLODIPINE BESYLATE AMODIAQUINE DIHYDROCHLORIDE AMOXEPINE AMOXICILLIN AMPICILLIN SODIUM AMPROLIUM AMRINONE AMYGDALIN ANABASAMINE HYDROCHLORIDE ANABASINE HYDROCHLORIDE ANCITABINE HYDROCHLORIDE ANDROSTERONE SODIUM SULFATE ANIRACETAM ANISINDIONE ANISODAMINE ANISOMYCIN ANTAZOLINE PHOSPHATE ANTHRALIN ANTIMYCIN A (A1 shown) ANTIPYRINE APHYLLIC -

Cholecystokinin Octapeptide Antagonized Opioid Analgesia Mediated by /T- and R- but Not Cs-Receptors in the Spinal Cord of the Rat
Brain Research, 523 (1990) 5-10 5 Elsevier BRES 15696 Cholecystokinin octapeptide antagonized opioid analgesia mediated by /t- and r- but not cS-receptors in the spinal cord of the rat Xiao-Jing Wang, Xiao-Hong Wang and Ji-Sheng Han Department of Physiology, Beijing Medical University, Beijing (People's Republic of China) (Accepted 23 January 1990) Key words: Cholecystokinin octapeptide; (N-MePhea,o-Pro4) Morphiceptin; (N-MeTyrl,N-MeArg7,D-Leu 8) Dynorphin(1-8) ethylamide; (D-Pen 2'5) Enkephalin; Proglumide; Intrathecal injection; Opioid analgesia; Antiopioid effect Intrathecal (ith) injection of cholecystokinin octapeptide (CCK-8) to the rat with single dose of 4 or 40 ng, or successive doses from 0.1 to 1/ag at 10 rain intervals produced neither analgesia nor hyperalgesia. However, the analgesia produced by ith injection of PL017, a specific /~-reeeptor agonist or 66A-078, a specific r-receptor agonist could be markedly antagonized by CCK-8 at a dose as small as 4 ng. In contrast, analgesia produced by ith injection of 6-agonist DPDPE could not be blocked by CCK-8 even at a dose as high as 40 ng. Since the effect of CCK-8 could be totally reversed by the CCK receptor antagonist proglumide, this effect is most probably mediated by CCK receptors. INTRODUCTION MATERIALS AND METHODS Cholecystokinin octapeptide (CCK-8) has been known Surgical procedures and intrathecal injection of drugs Male Wistar rats weighing 200-250 g were anesthetized with as a neuropeptide of abundant and wide distribution in chlorohydrate (0.4 g/kg, i.p.). PE-10 polyethiene catheter of 7.5 cm CNS 1 with some important physiological functions in- long was implanted through the atlanto-occipital membrane down to cluding the anti-opioid effect4'9. -

Lack of Rapid Antidepressant Effects of Kir4.1 Channel Inhibitors in A
Pharmacology, Biochemistry and Behavior 176 (2019) 57–62 Contents lists available at ScienceDirect Pharmacology, Biochemistry and Behavior journal homepage: www.elsevier.com/locate/pharmbiochembeh Lack of rapid antidepressant effects of Kir4.1 channel inhibitors in a chronic social defeat stress model: Comparison with (R)-ketamine T Zhongwei Xionga,b, Kai Zhanga, Tamaki Ishimaa, Qian Rena, Min Maa, Yaoyu Pua, Lijia Changa, ⁎ Jincao Chenb, Kenji Hashimotoa, a Division of Clinical Neuroscience, Chiba University Center for Forensic Mental Health, Chiba 260-8670, Japan b Department of Neurosurgery, Zhongnan Hospital of Wuhan University, Wuhan University, Wuhan 430071, PR China ARTICLE INFO ABSTRACT Keywords: A recent study demonstrated a key role of astroglial potassium channel Kir4.1 in the lateral habenula in de- Antidepressant pression. We investigated whether Kir4.1 protein is altered in the brain regions from susceptible mice after a Kir4.1 channel chronic social defeat stress (CSDS). Furthermore, we compared the rapid and sustained antidepressant actions of (R)-Ketamine Kir4.1 inhibitors (quinacrine and sertraline) and (R)-ketamine, (R)-enantiomer of rapid-acting antidepressant Social defeat stress model (R,S)-ketamine, in a CSDS model. Western blot analysis of Kir4.1 protein in the brain regions (prefrontal cortex, nucleus accumbens, hippocampus) from CSDS susceptible mice and control mice (no CSDS) was performed. Quinacrine (15, or 30 mg/kg), sertraline (20 mg/kg), (R)-ketamine (10 mg/kg), or vehicle was administered intraperitoneally to CSDS susceptible mice. Subsequently, locomotion test, tail suspension test (TST), forced swimming test (FST) and 1% sucrose preference test (SPT) were performed. There were no changes of Kir4.1 protein in the all regions between two groups. -

Diamandis Thesis
!"!#$ CHEMICAL GENETIC INTERROGATION OF NEURAL STEM CELLS: PHENOTYPE AND FUNCTION OF NEUROTRANSMITTER PATHWAYS IN NORMAL AND BRAIN TUMOUR INITIATING NEURAL PRECUSOR CELLS by Phedias Diamandis A thesis submitted in conformity with the requirements for the degree of Doctor of Philosophy. Department of Molecular Genetics University of Toronto © Copyright by Phedias Diamandis 2010 Phenotype and Function of Neurotransmitter Pathways in Normal and Brain Tumor Initiating Neural Precursor Cells Phedias Diamandis Doctor of Philosophy Department of Molecular Genetics University of Toronto 2010 &'(!)&*!% The identification of self-renewing and multipotent neural stem cells (NSCs) in the mammalian brain brings promise for the treatment of neurological diseases and has yielded new insight into brain cancer. The complete repertoire of signaling pathways that governs these cells however remains largely uncharacterized. This thesis describes how chemical genetic approaches can be used to probe and better define the operational circuitry of the NSC. I describe the development of a small molecule chemical genetic screen of NSCs that uncovered an unappreciated precursor role of a number of neurotransmitter pathways commonly thought to operate primarily in the mature central nervous system (CNS). Given the similarities between stem cells and cancer, I then translated this knowledge to demonstrate that these neurotransmitter regulatory effects are also conserved within cultures of cancer stem cells. I then provide experimental and epidemiologically support for this hypothesis and suggest that neurotransmitter signals may also regulate the expansion of precursor cells that drive tumor growth in the brain. Specifically, I first evaluate the effects of neurochemicals in mouse models of brain tumors. I then outline a retrospective meta-analysis of brain tumor incidence rates in psychiatric patients presumed to be chronically taking neuromodulators similar to those identified in the initial screen. -

(12) Patent Application Publication (10) Pub. No.: US 2015/0202317 A1 Rau Et Al
US 20150202317A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2015/0202317 A1 Rau et al. (43) Pub. Date: Jul. 23, 2015 (54) DIPEPTDE-BASED PRODRUG LINKERS Publication Classification FOR ALPHATIC AMNE-CONTAINING DRUGS (51) Int. Cl. A647/48 (2006.01) (71) Applicant: Ascendis Pharma A/S, Hellerup (DK) A638/26 (2006.01) A6M5/9 (2006.01) (72) Inventors: Harald Rau, Heidelberg (DE); Torben A 6LX3/553 (2006.01) Le?mann, Neustadt an der Weinstrasse (52) U.S. Cl. (DE) CPC ......... A61K 47/48338 (2013.01); A61 K3I/553 (2013.01); A61 K38/26 (2013.01); A61 K (21) Appl. No.: 14/674,928 47/48215 (2013.01); A61M 5/19 (2013.01) (22) Filed: Mar. 31, 2015 (57) ABSTRACT The present invention relates to a prodrug or a pharmaceuti Related U.S. Application Data cally acceptable salt thereof, comprising a drug linker conju (63) Continuation of application No. 13/574,092, filed on gate D-L, wherein D being a biologically active moiety con Oct. 15, 2012, filed as application No. PCT/EP2011/ taining an aliphatic amine group is conjugated to one or more 050821 on Jan. 21, 2011. polymeric carriers via dipeptide-containing linkers L. Such carrier-linked prodrugs achieve drug releases with therapeu (30) Foreign Application Priority Data tically useful half-lives. The invention also relates to pharma ceutical compositions comprising said prodrugs and their use Jan. 22, 2010 (EP) ................................ 10 151564.1 as medicaments. US 2015/0202317 A1 Jul. 23, 2015 DIPEPTDE-BASED PRODRUG LINKERS 0007 Alternatively, the drugs may be conjugated to a car FOR ALPHATIC AMNE-CONTAINING rier through permanent covalent bonds. -

Five Decades of Research on Opioid Peptides: Current Knowledge and Unanswered Questions
Molecular Pharmacology Fast Forward. Published on June 2, 2020 as DOI: 10.1124/mol.120.119388 This article has not been copyedited and formatted. The final version may differ from this version. File name: Opioid peptides v45 Date: 5/28/20 Review for Mol Pharm Special Issue celebrating 50 years of INRC Five decades of research on opioid peptides: Current knowledge and unanswered questions Lloyd D. Fricker1, Elyssa B. Margolis2, Ivone Gomes3, Lakshmi A. Devi3 1Department of Molecular Pharmacology, Albert Einstein College of Medicine, Bronx, NY 10461, USA; E-mail: [email protected] 2Department of Neurology, UCSF Weill Institute for Neurosciences, 675 Nelson Rising Lane, San Francisco, CA 94143, USA; E-mail: [email protected] 3Department of Pharmacological Sciences, Icahn School of Medicine at Mount Sinai, Annenberg Downloaded from Building, One Gustave L. Levy Place, New York, NY 10029, USA; E-mail: [email protected] Running Title: Opioid peptides molpharm.aspetjournals.org Contact info for corresponding author(s): Lloyd Fricker, Ph.D. Department of Molecular Pharmacology Albert Einstein College of Medicine 1300 Morris Park Ave Bronx, NY 10461 Office: 718-430-4225 FAX: 718-430-8922 at ASPET Journals on October 1, 2021 Email: [email protected] Footnotes: The writing of the manuscript was funded in part by NIH grants DA008863 and NS026880 (to LAD) and AA026609 (to EBM). List of nonstandard abbreviations: ACTH Adrenocorticotrophic hormone AgRP Agouti-related peptide (AgRP) α-MSH Alpha-melanocyte stimulating hormone CART Cocaine- and amphetamine-regulated transcript CLIP Corticotropin-like intermediate lobe peptide DAMGO D-Ala2, N-MePhe4, Gly-ol]-enkephalin DOR Delta opioid receptor DPDPE [D-Pen2,D- Pen5]-enkephalin KOR Kappa opioid receptor MOR Mu opioid receptor PDYN Prodynorphin PENK Proenkephalin PET Positron-emission tomography PNOC Pronociceptin POMC Proopiomelanocortin 1 Molecular Pharmacology Fast Forward. -
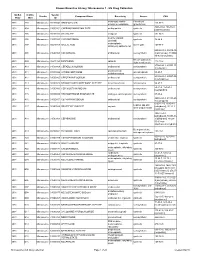
Known Bioactive Library: Microsource 1 - US Drug Collection
Known Bioactive Library: Microsource 1 - US Drug Collection ICCB-L ICCB-L Vendor Vendor Compound Name Bioactivity Source CAS Plate Well ID antifungal, inhibits Penicillium 2091 A03 Microsource 00200046 GRISEOFULVIN 126-07-8 mitosis in metaphase griseofulvum 3505-38-2, 486-16-8 2091 A04 Microsource 01500161 CARBINOXAMINE MALEATE antihistaminic synthetic [carbinoxamine] 2091 A05 Microsource 00200331 SALSALATE analgesic synthetic 552-94-3 muscle relaxant 2091 A06 Microsource 01500162 CARISOPRODOL synthetic 78-44-4 (skeletal) antineoplastic, 2091 A07 Microsource 00210369 GALLIC ACID insect galls 149-91-7 astringent, antibacterial 66592-87-8, 50370-12- 2091 A08 Microsource 01500163 CEFADROXIL antibacterial semisynthetic 2 [anhydrous], 119922- 89-9 [hemihydrate] Rheum palmatum, 2091 A09 Microsource 00211468 DANTHRON cathartic 117-10-2 Xyris semifuscata 27164-46-1, 25953-19- 2091 A10 Microsource 01500164 CEFAZOLIN SODIUM antibacterial semisynthetic 9 [cefazolin] glucocorticoid, 2091 A11 Microsource 00300024 HYDROCORTISONE adrenal glands 50-23-7 antiinflammatory 64485-93-4, 63527-52- 2091 A12 Microsource 01500165 CEFOTAXIME SODIUM antibacterial semisynthetic 6 [cefotaxime] 2091 A13 Microsource 00300029 DESOXYCORTICOSTERONE ACETATE mineralocorticoid adrenocortex 56-47-3 58-71-9, 153-61-7 2091 A14 Microsource 01500166 CEPHALOTHIN SODIUM antibacterial semisynthetic [cephalothin] 2091 A15 Microsource 00300034 TESTOSTERONE PROPIONATE androgen, antineoplastic semisynthetic 57-85-2 24356-60-3, 21593-23- 2091 A16 Microsource 01500167 CEPHAPIRIN SODIUM -

CHIRAL Handbook HPLC & SFC Separations
CHIRAL Handbook HPLC & SFC Separations Table of Contents Chapters Pages Introduction to Chiral Separations 2 Method Development 3 Guide for HPLC 3 Guide for SFC 12 Regis Chiral Columns 19 Pirkle-Type Chiral Stationary Phases 19 Polysaccharide Coated Chiral Stationary Phases 25 Crown-Ether Chiral Stationary Phases 26 Guide for ChiroSil Method Development 27 Frequently Asked Questions 30 HPLC & SFC Chiral Applications 33 References 304 Index 309 www.bgb-shop.com/regis 1 Introduction to Chiral Separations A resource built from nearly 50 years of experience Regis is pleased to present its inaugural edition of the Chiral Handbook. This definitive resource contains over 800 chiral applications using a variety of chiral column types as well as method development guidance and a Frequently Asked Question section. Don’t see the application you are looking for? Regis maintains a dedicated chiral separations laboratory that provides free chiral screening to identify the best column for your separation. For the latest applications and resources, please contact Regis or visit our website (www.chiral.com) for the most up-to-date information. Since 1970, Regis Technologies, Inc. has been a leader in HPLC and SFC chiral separations and purification services. We are proud to be a trusted supplier of high quality chromatography products and unique chiral stationary phases. Regis offers three different types of Chiral Stationary Phases (CSPs): • Covalently bonded Pirkle-Type • Coated Polysaccharide • Covalently bonded 18 Crown-ether All columns are available in analytical to preparative sizes as well as custom sizes. Chiral Separations One of the biggest challenges is finding the best column for separating compounds of various types, as there is no universal chiral stationary phase, and predicting which chiral stationary phase will provide the best separation is difficult. -

Appendix B – New Zealand and United Kingdom: Legislation
APPENDIX B – NEW ZEALAND AND UNITED KINGDOM: LEGISLATION AND POLICY (Appendices N.Z. - 35 to U.K. -41) SCOPE OF PRACTICE FOR REGISTERED NURSES IN THE EXTENDED CLASS (NURSE PRACTITIONERS): A JURISDICTIONAL REVIEW - NOVEMBER 2007 TABLE OF CONTENTS Page NZ – 35 Nursing Council of New Zealand: Scope of Practice………….. 3 NZ – 36 Nurse Council of New Zealand: Nurse Practitioner Endorsement – Guidelines for Applicants …………….………… 9 NZ – 37 Extract from New Zealand Gazette, 10/11/2005, No. 188 p. 4750: Medicines (Designated Prescriber: Nurse Preactitioners) Notice 2005 .…………………….………………………………….. 34 NZ – 38 Regulations of New Zealand: Medicines (Designated Prescriber: Nurse Practitioners) Regulation 2005 - Schedule Substances That are Nurse Practitioner Medicines if They are Prescription Medicines ………………………….……...…………. 35 UK – 39 Nursing and Midwifery Council: Advanced Nursing Practice Update 19, June 2007……………………………………..………. 71 UK – 40 Nursing and Midwifery Council: Nurse Prescribing and the Supply and Administration of Medication Position Statement..…………………………………………………….……. 73 UK – 41 Nursing and Midwifery Council: Guidelines for the Administraion of Medicine Nurse , January 2004…………………………………………………………..……... 82 2 ABC000002002 Scopes of Practice Page 1 of 6 Appendix N.Z.- 35 Nurs(n9 Counci,t of New ZeaLand Scopes of practice :_;, On 18th September 2004 the current registers were replaced by four scopes of practice under the Health Practitioners ._...... Competence Assurance Act 2003. There has been a smooth .... transition for nurses to the new scopes of practice. Every nurse who was on the register or roll was transferred onto the new _ register with the appropriate scope and conditions on 18 September. These are outlined in the table below. The four scopes of practice are" registered nurse, nurse ._... -

Blockade of Tolerance to Morphine but Not to Opioids by a Nitric Oxide Synthase Inhibitor (Nitric Oxide/Opiate/Dependence/Naloxone Benzoylhydrazone/U50,488H)
Proc. Natl. Acad. Sci. USA Vol. 90, pp. 5162-5166, June 1993 Pharmacology Blockade of tolerance to morphine but not to opioids by a nitric oxide synthase inhibitor (nitric oxide/opiate/dependence/naloxone benzoylhydrazone/U50,488H) YURI A. KOLESNIKOV, CHAIM G. PICK, GRAZYNA CISZEWSKA, AND GAVRIL W. PASTERNAK The Cotzias Laboratory of Neuro-Oncology, Memorial Sloan-Kettering Cancer Center, Departments of Neurology and Neuroscience and Pharmacology, Cornell University Medical College, New York, NY 10021 Communicated by Paul A. Marks, February 2, 1993 (received for review December 3, 1992) ABSTRACT The nitric oxide synthase inhibitor NG-nitro- the NOS inhibitor NO2Arg on the development of tolerance L-arginine (NO2Arg) blocks morphine tolerance in mice. After to morphine and extended these studies to K1 (trans-3,4- implantation of morphine pellets the analgesic response de- dichloro-N-methyl-N-[2-(1-pyrrolindinyl)cyclohexyl]-ben- creases from 100% on the first day to 0% on the third. zeneacetamide; U50,488H) and K3 [naloxone benzoylhydra- Coadministration of NO2Arg along with the pellets markedly zone (NalBzoH)] analgesics. retards the development of tolerance; 60% of mice are anal- gesic after 3 days, and 50% of mice are analgesic after 5 days. In a daily injection paradigm the analgesic response to mor- MATERIALS AND METHODS phine is reduced from 60% to 0% by 5 days. Concomitant Male CD-1 mice (25-35 g; Charles River Breeding Labora- administration of morphine along with NO2Arg at doses of 2 tories) were used in all studies. Morphine sulfate was ob- mg/kg per day prevents tolerance for 4 weeks. A single tained from Mallinckrodt, and U50,488H was from Upjohn. -

Stembook 2018.Pdf
The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances FORMER DOCUMENT NUMBER: WHO/PHARM S/NOM 15 WHO/EMP/RHT/TSN/2018.1 © World Health Organization 2018 Some rights reserved. This work is available under the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 IGO licence (CC BY-NC-SA 3.0 IGO; https://creativecommons.org/licenses/by-nc-sa/3.0/igo). Under the terms of this licence, you may copy, redistribute and adapt the work for non-commercial purposes, provided the work is appropriately cited, as indicated below. In any use of this work, there should be no suggestion that WHO endorses any specific organization, products or services. The use of the WHO logo is not permitted. If you adapt the work, then you must license your work under the same or equivalent Creative Commons licence. If you create a translation of this work, you should add the following disclaimer along with the suggested citation: “This translation was not created by the World Health Organization (WHO). WHO is not responsible for the content or accuracy of this translation. The original English edition shall be the binding and authentic edition”. Any mediation relating to disputes arising under the licence shall be conducted in accordance with the mediation rules of the World Intellectual Property Organization. Suggested citation. The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances. Geneva: World Health Organization; 2018 (WHO/EMP/RHT/TSN/2018.1). Licence: CC BY-NC-SA 3.0 IGO. Cataloguing-in-Publication (CIP) data. -

Supplement Ii to the Japanese Pharmacopoeia Seventeenth Edition
SUPPLEMENT II TO THE JAPANESE PHARMACOPOEIA SEVENTEENTH EDITION O‹cial from June 28, 2019 English Version THE MINISTRY OF HEALTH, LABOUR AND WELFARE Notice: This English Version of the Japanese Pharmacopoeia is published for the convenience of users unfamiliar with the Japanese language. When and if any discrepancy arises between the Japanese original and its English translation, the former is authentic. Printed in Japan The Ministry of Health, Labour and Welfare Ministerial Notification No. 49 Pursuant to Paragraph 1, Article 41 of Act on Securing Quality, Efficacy and Safety of Products Including Pharmaceuticals and Medical Devices (Act No. 145, 1960), this notification stated that a part of the Japanese Pharmacopoeia was revised as follows*. NEMOTO Takumi The Minister of Health, Labour and Welfare June 28, 2019 A part of the Japanese Pharmacopoeia (Ministerial Notification No. 64, 2016) was revised as follows*. (The text referred to by the term ``as follows'' are omitted here. All of the revised Japanese Pharmacopoeia in accordance with this notification (hereinafter referred to as ``new Pharmacopoeia'' in Supplement 2) are made available for public exhibition at the Pharmaceutical Evaluation Division, Pharmaceutical Safety and Environmen- tal Health Bureau, Ministry of Health, Labour and Welfare, at each Regional Bureau of Health and Welfare, and at each Prefectural Office in Japan). Supplementary Provisions (Effective Date) Article 1 This Notification is applied from June 28, 2019. (Transitional measures) Article 2 In the case of drugs which are listed in the Japanese Pharmacopoeia (hereinafter referred to as ``previous Pharmacopoeia'') [limited to those listed in new Pharmacopoeia] and drugs which have been approved as of June 28, 2019 as prescribed under Paragraph 1, Article 14 of Act on Securing Quality, Efficacy and Safety of Products Including Pharmaceuticals and Medical Devices [including drugs the Minister of Health, Labour and Welfare specifies (the Ministry of Health and Welfare Ministerial Notification No.