Immunosuppressant Ingredients, Immunostimulant Ingredients
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Newer Trends in the Management of Genital Herpes
Review NNewerewer trendstrends inin thethe mmanagementanagement ofof genitalgenital herpesherpes Article AAmiyamiya KKumarumar NNath,ath, DDevinderevinder MohanMohan ThappaThappa Department of Dermatology ABSTRACT and STD, Jawaharlal Institute of Postgraduate Medical Management of genital herpes is complex. Apart from using the standard antivirals, an Education and Research (JIPMER), Pondicherry - 605 ideal management protocol also needs to address various aspects of the disease, including 006, India the psychological morbidity. Oral acyclovir, valacyclovir or famciclovir are recommended for routine use. Long-term suppressive therapy is effective in reducing the number of AAddressddress forfor ccorrespondence:orrespondence: recurrences and the risk of transmission to others. Severe or disseminated disease may Dr. Devinder Mohan Thappa, require intravenous therapy. Resistant cases are managed with foscarnet or cidofovir. Genital Department of Dermatology herpes in human immunodeÞ ciency virus-infected individuals usually needs a longer duration and STD, JIPMER, of antiviral therapy along with continuation of highly active anti retroviral therapy (HAART). Pondicherry - 605 006, India. E-mail: [email protected] Genital herpes in late pregnancy increases the risk of neonatal herpes. Antiviral therapy and/or cesarean delivery are indicated depending on the clinical circumstance. Acyclovir appears to be safe in pregnancy. But, there is limited data regarding the use of valacyclovir and famciclovir in pregnancy. Neonatal herpes requires a higher dose of acyclovir given intravenously for a longer duration. Management of the sex partner, counseling and prevention advice are equally important in appropriate management of genital herpes. Vaccines till date have been marginally effective. Helicase–primase inhibitors, needle-free mucosal vaccine and a new microbicide product named VivaGel may become promising treatment options in the future. -

Metabolic Factors Affecting Tumor Immunogenicity: What Is Happening at the Cellular Level?
International Journal of Molecular Sciences Review Metabolic Factors Affecting Tumor Immunogenicity: What Is Happening at the Cellular Level? Rola El Sayed 1 , Yolla Haibe 2, Ghid Amhaz 2, Youssef Bouferraa 2 and Ali Shamseddine 2,* 1 Global Health Institute, American University of Beirut, Beirut 11-0236, Lebanon; [email protected] 2 Division of Hematology/Oncology, Department of Internal Medicine, American University of Beirut Medical Center, Beirut 11-0236, Lebanon; [email protected] (Y.H.); [email protected] (G.A.); [email protected] (Y.B.) * Correspondence: [email protected]; Tel.: +961-1-350-000 (ext. 5390) Abstract: Immunotherapy has changed the treatment paradigm in multiple solid and hematologic malignancies. However, response remains limited in a significant number of cases, with tumors de- veloping innate or acquired resistance to checkpoint inhibition. Certain “hot” or “immune-sensitive” tumors become “cold” or “immune-resistant”, with resultant tumor growth and disease progres- sion. Multiple factors are at play both at the cellular and host levels. The tumor microenvironment (TME) contributes the most to immune-resistance, with nutrient deficiency, hypoxia, acidity and different secreted inflammatory markers, all contributing to modulation of immune-metabolism and reprogramming of immune cells towards pro- or anti-inflammatory phenotypes. Both the tumor and surrounding immune cells require high amounts of glucose, amino acids and fatty acids to fulfill their energy demands. Thus, both compete over one pool of nutrients that falls short on needs, obliging cells to resort to alternative adaptive metabolic mechanisms that take part in shaping their inflammatory phenotypes. Aerobic or anaerobic glycolysis, oxidative phosphorylation, tryptophan catabolism, glutaminolysis, fatty acid synthesis or fatty acid oxidation, etc. -

Us 8530498 B1 3
USOO853 0498B1 (12) UnitedO States Patent (10) Patent No.: US 8,530,498 B1 Zeldis (45) Date of Patent: *Sep. 10, 2013 (54) METHODS FORTREATING MULTIPLE 5,639,476 A 6/1997 OShlack et al. MYELOMAWITH 5,674,533 A 10, 1997 Santus et al. 3-(4-AMINO-1-OXO-1,3-DIHYDROISOINDOL- 395 A 22 N. 2-YL)PIPERIDINE-2,6-DIONE 5,731,325 A 3/1998 Andrulis, Jr. et al. 5,733,566 A 3, 1998 Lewis (71) Applicant: Celgene Corporation, Summit, NJ (US) 5,798.368 A 8, 1998 Muller et al. 5,874.448 A 2f1999 Muller et al. (72) Inventor: Jerome B. Zeldis, Princeton, NJ (US) 5,877,200 A 3, 1999 Muller 5,929,117 A 7/1999 Muller et al. 5,955,476 A 9, 1999 Muller et al. (73) Assignee: Celgene Corporation, Summit, NJ (US) 6,020,358 A 2/2000 Muller et al. - 6,071,948 A 6/2000 D'Amato (*) Notice: Subject to any disclaimer, the term of this 6,114,355 A 9, 2000 D'Amato patent is extended or adjusted under 35 SS f 1939. All et al. U.S.C. 154(b) by 0 days. 6,235,756 B1 5/2001 D'Amatoreen et al. This patent is Subject to a terminal dis- 6,281.230 B1 8/2001 Muller et al. claimer 6,316,471 B1 1 1/2001 Muller et al. 6,326,388 B1 12/2001 Man et al. 6,335,349 B1 1/2002 Muller et al. (21) Appl. No.: 13/858,708 6,380.239 B1 4/2002 Muller et al. -

The Effects of Atopy and Asthma on in Vivo Human Nasal Responses to Toll
The effects of atopy and asthma on in vivo human nasal responses to Toll-like receptor agonists Dr Akhilesh Jha A thesis submitted for the degree of Doctor of Philosophy (PhD) Centre for Respiratory Infection National Heart and Lung Institute (NHLI), St Mary’s Campus Imperial College London, Norfolk Place, London W2 1PG July 2018 Abstract Acute respiratory viral infections cause significant morbidity and mortality, especially in vulnerable individuals, and it is important to study viral pathogenesis and the host immune response in humans. Toll-like receptors (TLRs) play a critical role in the detection of viral nucleic acids, and airway TLR receptors respond to nucleic acid patterns in the RNA viruses that cause respiratory infections. However, a reliable method of measuring mucosal innate immune responses to viral infections is lacking. TLR3 agonists (poly(I:C) and poly-ICLC) and the combined TLR7/8 agonist (resiquimod, R848) are synthetic analogues of double stranded RNA (dsRNA) and single stranded RNA (ssRNA) respectively. Nasal challenge with these TLR agonists was carried out, and serial sampling using nasosorption and nasal curettage was performed. Mucosal immune responses were measured and the effect of different host factors (e.g. asthma) on these responses was studied. Poly(I:C) and poly-ICLC were well tolerated but failed to induce significant and reliable nasal mucosal innate immune responses. R848 at a higher dose (10 µg/100 µL per nostril) induced significant mucosal interferon and cytokine responses but caused mild to moderate flu-like symptoms in three out of nine volunteers. A lower dose of R848 (0.02 µg/kg/100 µL, mean dose 1.5 µg/100 µL) was subsequently utilised in three groups of volunteers: healthy non-atopic (n=12), allergic rhinitis (n=12) and allergic asthma (n=11). -
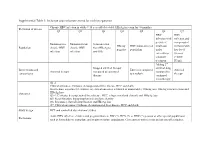
Inclusion and Exclusion Criteria for Each Key Question
Supplemental Table 1: Inclusion and exclusion criteria for each key question Chronic HBV infection in adults ≥ 18 year old (detectable HBsAg in serum for >6 months) Definition of disease Q1 Q2 Q3 Q4 Q5 Q6 Q7 HBV HBV infection with infection and persistent compensated Immunoactive Immunotolerant Seroconverted HBeAg HBV mono-infected viral load cirrhosis with Population chronic HBV chronic HBV from HBeAg to negative population under low level infection infection anti-HBe entecavir or viremia tenofovir (<2000 treatment IU/ml) Adding 2nd Stopped antiviral therapy antiviral drug Interventions and Entecavir compared Antiviral Antiviral therapy compared to continued compared to comparisons to tenofovir therapy therapy continued monotherapy Q1-2: Clinical outcomes: Cirrhosis, decompensated liver disease, HCC and death Intermediate outcomes (if evidence on clinical outcomes is limited or unavailable): HBsAg loss, HBeAg seroconversion and Outcomes HBeAg loss Q3-4: Cirrhosis, decompensated liver disease, HCC, relapse (viral and clinical) and HBsAg loss Q5: Renal function, hypophosphatemia and bone density Q6: Resistance, flare/decompensation and HBeAg loss Q7: Clinical outcomes: Cirrhosis, decompensated liver disease, HCC and death Study design RCT and controlled observational studies Acute HBV infection, children and pregnant women, HIV (+), HCV (+) or HDV (+) persons or other special populations Exclusions such as hemodialysis, transplant, and treatment failure populations. Co treatment with steroids and uncontrolled studies. Supplemental Table 2: Detailed Search Strategy: Ovid Database(s): Embase 1988 to 2014 Week 37, Ovid MEDLINE(R) In-Process & Other Non- Indexed Citations and Ovid MEDLINE(R) 1946 to Present, EBM Reviews - Cochrane Central Register of Controlled Trials August 2014, EBM Reviews - Cochrane Database of Systematic Reviews 2005 to July 2014 Search Strategy: # Searches Results 1 exp Hepatitis B/dt 26410 ("hepatitis B" or "serum hepatitis" or "hippie hepatitis" or "injection hepatitis" or 2 178548 "hepatitis type B").mp. -

Multinational Evaluation of Mycophenolic Acid, Tacrolimus
View metadata, citation and similar papers at core.ac.uk brought to you by CORE providedORIGINAL by University of QueenslandPAPER eSpace ISSN 1425-9524 © Ann Transplant, 2016; 21: 1-11 DOI: 10.12659/AOT.895664 Received: 2015.08.15 Accepted: 2015.09.01 Multinational Evaluation of Mycophenolic Published: 2016.01.05 Acid, Tacrolimus, Cyclosporin, Sirolimus, and Everolimus Utilization Authors’ Contribution: ABCDEF Kyle M. Gardiner School of Pharmacy, University of Queensland, Brisbane, QLD, Australia Study Design A ACDEF Susan E. Tett Data Collection B Statistical Analysis C ACDEF Christine E. Staatz Data Interpretation D Manuscript Preparation E Literature Search F Funds Collection G Corresponding Author: Christine E. Staatz, e-mail: [email protected] Source of support: Departmental funding only Background: Increasing immunosuppressant utilization and expenditure is a worldwide challenge as more people success- fully live with transplanted organs. Our aims were to characterize utilization of mycophenolate, tacrolimus, cy- closporin, sirolimus, and everolimus in Australian transplant recipients from 2007 to 2013; to identify specific patterns of usage; and to compare Australian utilization with Norwegian, Danish, Swedish, and the Netherlands use. Material/Methods: Australian utilization and expenditure data were captured through national Pharmaceutical Benefits Scheme and Highly Specialized Drug administrative databases. Norwegian, Danish, Swedish, and the Netherlands uti- lization were retrieved from their healthcare databases. Utilization was compared as defined daily dose per 1000 population per day (DDD/1000 population/day). Data on kidney transplant recipients, the predominant patient group prescribed these medicines, were obtained from international transplant registries. Results: From 2007–2013 Australian utilization of mycophenolic acid, tacrolimus and everolimus increased 2.7-fold, 2.2- fold, and 2.3-fold, respectively. -

WHO Drug Information Vol 22, No
WHO Drug Information Vol 22, No. 1, 2008 World Health Organization WHO Drug Information Contents Challenges in Biotherapeutics Miglustat: withdrawal by manufacturer 21 Regulatory pathways for biosimilar Voluntary withdrawal of clobutinol cough products 3 syrup 22 Pharmacovigilance Focus Current Topics WHO Programme for International Drug Proposed harmonized requirements: Monitoring: annual meeting 6 licensing vaccines in the Americas 23 Sixteen types of counterfeit artesunate Safety and Efficacy Issues circulating in South-east Asia 24 Eastern Mediterranean Ministers tackle Recall of heparin products extended 10 high medicines prices 24 Contaminated heparin products recalled 10 DacartTM development terminated and LapdapTM recalled 11 ATC/DDD Classification Varenicline and suicide attempts 11 ATC/DDD Classification (temporary) 26 Norelgestromin-ethynil estradiol: infarction ATC/DDD Classification (final) 28 and thromboembolism 12 Emerging cardiovascular concerns with Consultation Document rosiglitazone 12 Disclosure of transdermal patches 13 International Pharmacopoeia Statement on safety of HPV vaccine 13 Cycloserine 30 IVIG: myocardial infarction, stroke and Cycloserine capsules 33 thrombosis 14 Erythropoietins: lower haemoglobin levels 15 Recent Publications, Erythropoietin-stimulating agents 15 Pregabalin: hypersensitivity reactions 16 Information and Events Cefepime: increased mortality? 16 Assessing the quality of herbal medicines: Mycophenolic acid: pregnancy loss and contaminants and residues 36 congenital malformation 17 Launch -

Modulating the Immune System Through Nanotechnology
Modulating the immune system through nanotechnology Tamara G. Dacobaa,b*, Ana Oliveraa,b*, Dolores Torresb, José Crecente- Campoa,b#, María José Alonsoa,b## aCenter for Research in Molecular Medicine and Chronic Diseases (CIMUS), Campus Vida, Universidade de Santiago de Compostela, Santiago de Compostela 15782, Spain. bDepartment of Pharmacology, Pharmacy and Pharmaceutical Technology, School of Pharmacy, Campus Vida, Universidade de Santiago de Compostela, Santiago de Compostela 15782, Spain. *These authors contributed equally to this work. #Corresponding author e-mail address: [email protected] ##Corresponding author e-mail address: [email protected] 1 Abstract Nowadays, nanotechnology-based modulation of the immune system is presented as a cutting-edge strategy, which may lead to significant improvements in the treatment of severe diseases. In particular, efforts have been focused on the development of nanotechnology- based vaccines, which could be used for immunization or generation of tolerance. In this review, we highlight how different immune responses can be elicited by tuning nanosystems properties. In addition, we discuss specific formulation approaches designed for the development of anti-infectious and anti-autoimmune vaccines, as well as those intended to prevent the formation of antibodies against biologicals. Graphical abstract Keywords: nanotechnology; immune system; tolerance; stimulation; autoimmune disease; vaccine Highlights - Nanocarriers can be designed to target specific immune cells - Nanovaccines may help fighting diseases that are elusive to traditional vaccines - Nanocarriers can bias the immune response from humoral to cellular - Autoimmune disease treatments can be improved with nanotechnology-based approaches - The use of nanocarriers may help to avoid ADAs formation against biotherapeutics 2 1. Introduction The modulation of the immune system is the base of new and promising therapies for some of the most prevalent and/or severe diseases of our time, such as cancer, HIV, and diabetes. -

Study Protocol
PROTOCOL SYNOPSIS A Multicentre, Randomised, Double-blind, Placebo-controlled, Phase 3 Study Evaluating the Efficacy and Safety of Two Doses of Anifrolumab in Adult Subjects with Active Systemic Lupus Erythematosus International Coordinating Investigator Study site(s) and number of subjects planned Approximately 450 subjects are planned at approximately 173 sites. Study period Phase of development Estimated date of first subject enrolled Q2 2015 3 Estimated date of last subject completed Q2 2018 Study design This is a Phase 3, multicentre, multinational, randomised, double-blind, placebo-controlled study to evaluate the efficacy and safety of an intravenous treatment regimen of anifrolumab (150 mg or 300 mg) versus placebo in subjects with moderately to severely active, autoantibody-positive systemic lupus erythematosus (SLE) while receiving standard of care (SOC) treatment. The study will be performed in adult subjects aged 18 to 70 years of age. Approximately 450 subjects receiving SOC treatment will be randomised in a 1:2:2 ratio to receive a fixed intravenous dose of 150 mg anifrolumab, 300 mg anifrolumab, or placebo every 4 weeks (Q4W) for a total of 13 doses (Week 0 to Week 48), with the primary endpoint evaluated at the Week 52 visit. Investigational product will be administered as an intravenous (IV) infusion via an infusion pump over a minimum of 30 minutes, Q4W. Subjects must be taking either 1 or any combination of the following: oral corticosteroids (OCS), antimalarial, and/or immunosuppressants. Randomisation will be stratified using the following factors: SLE Disease Activity Index 2000 (SLEDAI-2K) score at screening (<10 points versus ≥10 points); Week 0 (Day 1) OCS dose 2(125) Revised Clinical Study Protocol Drug Substance Anifrolumab (MEDI-546) Study Code D3461C00005 Edition Number 5 Date 18 May 2016 (<10 mg/day versus ≥10 mg/day prednisone or equivalent); and results of a type 1 interferon (IFN) test (high versus low). -

Bioinformatics Review of the Role of HSV-1 in Alzheimer's Disease
Advances in Alzheimer’s Disease, 2020, 9, 57-75 https://www.scirp.org/journal/aad ISSN Online: 2169-2467 ISSN Print: 2169-2459 Bioinformatics Review of the Role of HSV-1 in Alzheimer’s Disease Brian T. Reiss1, Meade C. Eggleston1, Arah C. Godbole1, Julia A. Marut1, Cecelia M. McCann1, Jessica A. Cottrell1, Tinchun Chu1, Sulie L. Chang1,2* 1Department of Biological Sciences, Seton Hall University, South Orange, NJ, USA 2Institute of Neuroimmune Pharmacology, Seton Hall University, South Orange, NJ, USA How to cite this paper: Reiss, B.T., Eg- Abstract gleston, M.C., Godbole, A.C., Marut, J.A., McCann, C.M., Cottrell, J.A., Chu, T.C. and Alzheimer’s disease (AD) is a neurodegenerative disease characterized by the Chang, S.L. (2020) Bioinformatics Review progressive loss of cognitive functions in affected individuals. Brain tissue of the Role of HSV-1 in Alzheimer’s Dis- pathology is associated with the formation of senile plaques which result from ease. Advances in Alzheimer’s Disease, 9, 57-75. the over-production of amyloid β (Aβ), due to the cleavage of a membrane https://doi.org/10.4236/aad.2020.93005 bound glycoprotein. It is unclear what causes AD and its associated patholo- gies, but age and genetic predisposition play an import role in the likelihood Received: June 11, 2020 Accepted: September 6, 2020 of disease development. Studies have shown that the reactivation of latent Published: September 9, 2020 herpes simplex virus 1 (HSV-1) infection can lead to the neuropathy of acute herpes simplex encephalitis (HSE), which causes similar symptoms to AD. -

Polymorphisms of the Multidrug Pump ABCG2: a Systematic Review of Their Effect on Protein Expression, Function, and Drug Pharmacokinetics
1521-009X/46/12/1886–1899$35.00 https://doi.org/10.1124/dmd.118.083030 DRUG METABOLISM AND DISPOSITION Drug Metab Dispos 46:1886–1899, December 2018 Copyright ª 2018 by The American Society for Pharmacology and Experimental Therapeutics Minireview Polymorphisms of the Multidrug Pump ABCG2: A Systematic Review of Their Effect on Protein Expression, Function, and Drug Pharmacokinetics Niall Heyes, Parth Kapoor, and Ian D. Kerr School of Life Sciences, Queen’s Medical Centre, University of Nottingham, Nottingham, United Kingdom Received June 12, 2018; accepted September 20, 2018 Downloaded from ABSTRACT The widespread expression and polyspecificity of the multidrug to improve outcomes in cancer patients treated with tyrosine ABCG2 efflux transporter make it an important determinant of the kinase inhibitors, but the reasons for this are yet to be estab- pharmacokinetics of a variety of substrate drugs. Null ABCG2 lished, and this residue’s role in the mechanism of the protein is expression has been linked to the Junior blood group. Polymor- unexplored by current biochemical and structural approaches. phisms affecting the expression or function of ABCG2 may have Research into the less-common polymorphisms is confined to dmd.aspetjournals.org clinically important roles in drug disposition and efficacy. The in vitro studies, with several polymorphisms shown to decrease most well-studied single nucleotide polymorphism (SNP), Q141K resistance to anticancer agents such as SN-38 and mitoxantrone. (421C>A), is shown to decrease ABCG2 expression and activity, In this review, we present a systematic analysis of the effects of resulting in increased total drug exposure and decreased re- ABCG2 polymorphisms on ABCG2 function and drug pharmaco- sistance to various substrates. -

IMPDH Inhibitors for Anti-Tumor Therapy in Tuberous Sclerosis Complex
bioRxiv preprint doi: https://doi.org/10.1101/835199; this version posted November 11, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. IMPDH inhibitors for anti-tumor therapy in tuberous sclerosis complex Alexander J. Valvezan1, *, Spencer K. Miller1, Molly C. McNamara1, Margaret E. Torrence1, John M. Asara2, Elizabeth P. Henske3, Brendan D. Manning1,* 1Department of Genetics and Complex Diseases, Harvard T.H. Chan School of Public Health, Boston, MA, USA 2Division of Signal Transduction, Beth Israel Deaconess Medical Center, and Department of Medicine, Harvard Medical School, Boston, MA, USA 3Department of Medicine, Brigham and Women's Hospital and Harvard Medical School, Boston, MA, USA *Correspondence to: [email protected] or [email protected] 665 Huntington Ave, SPH2-117 Boston, MA 02115 617 432-5614 Running title: Repurposing mizoribine for tuberous sclerosis complex tumors Keywords: mTORC1, IMPDH, mizoribine, mycophenolate, tuberous sclerosis complex Financial support: This research was supported by grants from the National Institutes of Health (P01CA120964 to J.M.A. E.P.H. and B.D.M., P30CA006516 to J.M.A., and R35CA197459 to B.D.M.) and a Rothberg Courage Award from the TS Alliance to B.D.M. Conflict of interest statement: B.D.M. is a shareholder and scientific advisory board member for Navitor Pharmaceuticals and LAM Therapeutics. Word count: 5,171 Number of figures: 6 main figures, 4 supplemental figures 1 bioRxiv preprint doi: https://doi.org/10.1101/835199; this version posted November 11, 2019.