Journal of Vaccines & Vaccination
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

0 January to July 2021
0 www.journalsofindia.com January to July 2021 SCIENCE & TECH ............................................................................................................................................................... 6 1. REUSABLE LAUNCH VEHICLE TECHNOLOGY DEMONSTRATION PROGRAMME(RLV-TD) ................................................. 6 2. GAGANYAAN MISSION ..................................................................................................................................................... 6 3. MARS ORBITER MISSION (MOM) ..................................................................................................................................... 6 4. CHANDRAYAAN MISSION................................................................................................................................................. 7 5. SOLAR MISSION ............................................................................................................................................................... 8 6. ARTEMIS ACCORD ............................................................................................................................................................ 9 7. NATIONAL MISSION ON INTERDISCIPLINARY CYBER-PHYSICAL SYSTEM (NMICPS) ....................................................... 10 8. SMART ANTI-AIRFIELD WEAPON (SAAW) ...................................................................................................................... 10 9. AQUAPONICS ................................................................................................................................................................ -

Vaccination and Anaphylaxis
14 FORENSIC SCIENCE Croat Med J. 2017;58:14-25 https://doi.org/10.3325/cmj.2017.58.14 Vaccination and anaphylaxis: a Cristian Palmiere1, Camilla Tettamanti2, Maria Pia forensic perspective Scarpelli1 1CURML, University Center of Legal Medicine, Lausanne 25, Switzerland 2Department of Legal Medicine, Aim To review the available literature pertaining to fatali- University of Genova, Genova, Italy ties following vaccine administration and, in particular, cas- es of vaccine-related fatal anaphylaxis. Method The MEDLINE database was systematically searched up to March 2016 to identify all relevant articles pertaining to fatal cases of anaphylaxis following vaccine administration. Results Six papers pertaining to fatal anaphylaxis following vaccination were found relevant. Mast cell tryptase and to- tal IgE concentration was assessed exclusively in one case. Laryngeal edema was not detected in any of these cases, whereas eosinophil or mast cell infiltration was observed in lymphoid organs. In one case, immunohistochemical in- vestigations using anti-tryptase antibodies allowed pulmo- nary mast cells and degranulating mast cells with tryptase- positive material outside to be identified. Conclusion In any suspected IgE-mediated fatal anaphy- lactic cases, biochemical investigations should be system- atically performed for forensic purposes. Splenic tissue should be routinely sampled for immunohistochemical investigations in all suspected anaphylaxis-related deaths and mast cell/eosinophil infiltrations should be systemati- cally sought out in -

India: the World's Pharmacy Expands Its Reach in Global Health
New York | New Delhi | Rio de Janeiro Nairobi | Johannesburg | London India: The World’s Pharmacy Expands Its Reach in Global Health March 2021 This white paper was last updated on 1 March 2021 Global Health Strategies 18/1, 2nd Floor Shaheed Bhawan, Aruna Asaf Ali Marg, New Delhi, 110 067 www.globalhealthstrategies.com Twitter: @GHS Contents Executive Summary 02 India’s response to COVID-19 03 ∙ Pharmaceuticals and Biosimilars 04 ∙ Vaccines 05 ∙ Diagnostics 06 Evolution of India’s pharmaceutical industry 07 ∙ Milestones 08 Case Studies ∙ Hepatitis B Vaccine 10 ∙ Anti-retroviral Drugs 11 ∙ MenAfriVac 12 ∙ Complex Generics 13 ∙ Insulin 14 ∙ Monoclonal Antibodies 15 ∙ Vaccine for Rotavirus 16 ∙ Typhoid Conjugate Vaccine 17 Conclusion: Looking Forward 18 Executive Summary India’s pharmaceutical industry is already playing a pivotal role in the scale-up of pharmaceuticals and diagnostics to combat the global COVID-19 pandemic. It is poised to play an even more dominant role as biological products – preventive vaccines and cutting-edge biotechnology such as monoclonal antibodies– come to the fore. Even before the pandemic, Indian manufacturers produced vast quantities of generic antiviral drugs that turned HIV from a death sentence to a chronic manageable condition in developing countries. India’s global dominance in generic drugs and vaccine manufacturing has earned it the label “Pharmacy of the World”. COVID-19 only strengthens the case for this moniker. So far, India has supplied medicines to 133 countries to fight the pandemic. Six Indian manufacturers have been granted royalty-free licenses by Gilead to manufacture the first antiviral drug approved by the U.S. -

Production Flow Sheet
(Refer SOP No. QA-INS-010) Central Drugs Standard Control Organization Directorate General of Health Services, Ministry of Health and Family Welfare, Government of India FDA Bhavan, ITO, Kotla Road, New Delhi -110002 Central Inspection Plan Using Risk Based Approach Of Vaccine Manufacturing Units for 2019 Sl. Name and Vaccines manufactured (category Dt. of last Purpose of last Major Compliance AEFI reported/ Proposed time of No. address of the wise list) inspection inspection (grant deficiencies met till date, changes/ Inspection manufacturing with no of /renewal /post-approval detected if any Product site days & team changes, AEFI, follow- complaints, up, routine) failure if any A NORTH ZONE 1 M/s Biomed Pvt. 1.Oral Polio Vaccine IP, 03.10.2018 to To find out the root As per report Observations NSQ Reported 1st Quarter Ltd, 2. Vi olysaccharide Typhoid Vaccine 06.10.2018 & cause for the NSQ communicate Annual Inspection C-96, Site –I, (Bio TyphTM) IP, 3. Haemophilus Type- 08.10.2018 Reported d to the firm Bulandshahar B Conjugate Vaccine (PedaHib) for 2nd Quarter 4. Meningococal Polysaccharide Vaccine ADC(I) and compliance. Follow-up Road, Ind. Area, (Group A,C,Y,W 135) QuadriMeningo Ghaziabad. 5. Meningococal Polysaccharide Vaccine DIs of Inspection (Group A&C) Bivalent CDSCO, , and (if any) 6. Vi Conjugate Typhoid Vaccine (Peda DI, Uttar Typh), Pradesh 3rd Quarter 7. Vi Conjugate Typhoid Vaccine (Bio Annual Inspection TyphTM), 8. Haemophilus Type-B Conjugate 4th Quarter Vaccine (PedaHib) IP Follow-up 9. Meningococal Inspection Polysaccharide Vaccine (Group A,C,Y,W 135) IP QuadriMeningo (if any) 10. -

Requirements for Diphtheria. Tetanus. Pertussis and Combined Vaccines
0World Health Organization World Health Or_panizatior?. Technics; Rsporr Series. Xo . S0.l . 1990 Annex 2 REQUIREMENTS FOR DIPHTHERIA. TETANUS. PERTUSSIS AND COMBINED VACCINES (Requirements for Biological Substances Nos. 8 and 10) (Revised 1989) Page Introduction ....................................................................................................... 88 Requirements for diphtheria vaccine (adsorbed) ............................................ 91 General considerations ........................................................................... 91 Part A . Manufacturing requirements .......................................................... A .1 Definit~ons............................................................................. A.2 General manufacturing requirements .................................... A.3 Production control ................................................................ A.4 Filling and containers ........................................................... A.5 Control of final product ........................................................ A.6 Records ................................................................................. A.7 Samples ................................................................................. A.8 Labelling ................................................................................ A.9 Distribution and transport .................................................... A.10 Stability . storage and expir>-date ..................................... Part B . Kational control -

WHO Expert Committee on Biological Standardization of Publications
WHOWHOWHO Technical Technical Technical Report Report Report Series Series Series 941941941 BIOLOGICAL BIOLOGICAL BIOLOGICAL ThisThisThis report report report presents presents presents the the recommendationsthe recommendations recommendations of ofa ofWHOa WHOa WHO Expert Expert Expert WHOWHOWHO EXPERT EXPERT EXPERT COMMITTEE COMMITTEE COMMITTEE CommitteeCommitteeCommittee commissioned commissioned commissioned to tocoordinate tocoordinate coordinate activities activities activities leading leading leading to tothe tothe adoptionthe adoption adoption of ofinternational ofinternational international recommendations recommendations recommendations for for the for the the ONONON BIOLOGICAL BIOLOGICAL BIOLOGICAL productionproductionproduction and and andcontrol control control of ofvaccines ofvaccines vaccines and and andother other other biologicals biologicals biologicals STANDARDIZATION STANDARDIZATION STANDARDIZATION andand andthe the establishmentthe establishment establishment of ofinternational ofinternational international biological biological biological reference reference reference STANDARDIZATIONSTANDARDIZATIONSTANDARDIZATION materials.materials.materials. TheThe Thereport report report starts starts starts with with with a discussiona discussiona discussion of ofgeneral ofgeneral general issues issues issues brought brought brought to tothe tothe attentionthe attention attention of ofthe ofthe Committeethe Committee Committee and and andprovides provides provides information information information onon theon the statusthe -

The Historical Impact of Epidemic Typhus
THE HISTORICAL IMPACT OF EPIDEMIC TYPHUS by JOSEPH M. CONLON LCDR, MSC, USN [email protected] INTRODUCTION Louse-borne Typhus Fever is undoubtedly one of the oldest pestilential diseases of mankind. Called by many names and confused with other fevers, it is not until the late fifteenth century that it can be recognized with certainty as causing devastating epidemics. With Plague, Typhoid, and Dysentery, it was the scourge of armies and civilian populations throughout the Middle Ages and frequently played a decisive role in wars conducted in Europe from the 15th through the 20th centuries. The manner in which the course of European history has been affected by Typhus epidemics has been graphically portrayed by a number of authors. This paper will attempt a further analysis of the historical impact of Louse-borne Typhus and how its epidemic propagation has led many to regard Pediculus humanus corporis as having a more profound effect on human history than any other animal. EPIDEMIC TYPHUS FEVER (TABARILLO, CLASSIC OR EUROPEAN TYPHUS, JAIL FEVER, WAR FEVER) Causative Agent. Typhus fever is an acute specific infection caused by Rickettsia prowazeki as isolated and identified by DaRocha-Lima in 1916. Named in honor of H. T. Ricketts and L. von Prowazek, both of whom contracted typhus in the course of their investigations and died, R. prowazeki was originally believed to be a virus because of its minute size and difficulty of cultivation. R. prowazeki is now recognized as being morphologically and biochemically a type of bacterium. A rod-shaped microorganism, R. prowazeki is an obligate intracellular parasite whose cell wall contains muramic acid, diaminopimelic acid, and other components similar to those of the gram-negative bacteria. -
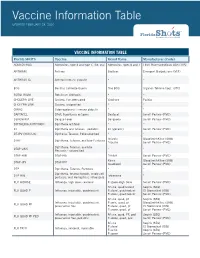
Vaccine Information Table UPDATED FEBRUARY 28, 2020
Vaccine Information Table UPDATED FEBRUARY 28, 2020 VACCINE INFORMATION TABLE Florida SHOTS Vaccine Brand Name Manufacturer (Code) ADENOVIRUS Adenovirus, type 4 and type 7, live, oral Adenovirus Types 4 and 7 TEVA Pharmaceuticals USA (TVA) ANTHRAX Anthrax Biothrax Emergent BioSolutions (MIP) ANTHRAX IG Anthrax immune globulin * * BCG Bacillus Calmette-Guerin Tice BCG Organon Teknika Corp. (OTC) BOTULINUM Botulinum Antitoxin * * CHOLERA LIVE Cholera, live attenuated Vaxchora PaxVax CHOLERA UNK Cholera, unspecified * * CMVIG Cytomegalovirus immune globulin * * DAPTACEL DTaP, 5 pertussis antigens Daptacel Sanofi Pasteur (PMC) DENGVAXIA Dengue Fever Dengvaxia Sanofi Pasteur (PMC) DIPTHERIA ANTITOXIN Diphtheria antitoxin * * DT Diphtheria and Tetanus - pediatric DT (generic) Sanofi Pasteur (PMC) DT-IPV (NON-US) Diptheria, Tetanus, Polio adsorbed * * Infanrix GlaxoSmithKline (SKB) DTAP Diphtheria, Tetanus, acellular Pertussis Tripedia Sanofi Pasteur (PMC) Diphtheria, Tetanus, acellular DTAP UNK * * Pertussis - unspecified DTAP-HIB DTaP-Hib Trihibit Sanofi Pasteur (PMC) Kinrix GlaxoSmithKline (SKB) DTAP-IPV DTaP-IPV Quadracel Sanofi Pasteur (PMC) DTP Diphtheria, Tetanus, Pertussis * * Diphtheria, tetanux toxoids, whole cell DTP-HIB Tetramune * pertussis, and Hemophilus influenza B FLU HIDOSE Influenza, high dose, seasonal Fluzone-High Dose Sanofi Pasteur (PMC) Afluria, quadrivalent Seqiris (SEQ) FLU QUAD P Influenza, injectable, quadrivalent Flulaval, quadrivalent ID Biomedical (IDB) Fluzone, quadrivalent Sanofi Pasteur (PMC) Afluria, quad, pf -

WHO Expert Committee on Biological Standardization Sixtieth Report
977 WHO Technical Report Series 977 WHO Expert on Biological Standardization Committee This report presents the recommendations of a WHO expert committee commissioned to coordinate activities leading to the adoption of international recommendations for the production and control of vaccines and other biologicals and the establishment of international biological reference materials. Thereport starts with a discussion of general issues brought to the attention of the Committee and provides information on WHO Expert Committee the status and development of reference materials for various antibodies, antigens, blood products and related substances, on Biological cytokines, growth factors, endocrinological substances and in vitro diagnostic devices. The second part of the report, of particular relevance to manufacturers and national regulatory Standardization authorities, contains revised WHO Recommendations for production and control of live attenuated influenza vaccines and for production and control of pneumococcal conjugate vaccines. New WHO Guidelines on the regulatory evaluation of similar biotherapeutic medicines are also provided. Sixtieth report Also included are a list of Recommendations, Guidelines and other documents for biological substances used in medicine, WHO and of International Standards and Reference Reagent for biological substances. Report Series Technical The World Health Organization was established in 1948 as a specialized agency of the SELECTED WHO PUBLICATIONS OF RELATED INTEREST United Nations serving as the directing and coordinating authority for international health matters and public health. One of WHO’s constitutional functions is to provide objective and reliable information and advice in the field of human health, a responsibility that it fulfils in part through its extensive programme of publications. WHO Expert Committee on Biological Standardization The Organization seeks through its publications to support national health strategies Fifty-ninth report. -

Coronavirus Vaccine Tracker
https://nyti.ms/2MHNdRL U.S.A. World Health Coronavirus Vaccine Tracker By Carl Zimmer, Jonathan Corum and Sui-Lee Wee Updated April 5, 2021 PHASE 1 PHASE 2 PHASE 3 AUTHORIZED APPROVED ABANDONED 50 35 23 5 8 4 Vaccines Vaccines Vaccines Vaccines Vaccines Vaccines testing safety in expanded in large-scale in early or approved abandoned and dosage safety trials efficacy tests limited use for full use after trials Vaccines typically require years of research and testing before reaching the clinic, but in 2020, scientists embarked on a race to produce safe and effective coronavirus vaccines in record time. Researchers are currently testing 86 vaccines in clinical trials on humans, and 23 have reached the final stages of testing. At least 77 preclinical vaccines are under active investigation in animals. New additions and recent updates April 5 The Walter Reed Army Institute of Research enters Phase 1. April 5 Japanʼs KM Biologics begins Phase 1/2. April 2 Franceʼs OSE Immunotherapeutics enters Phase 1. March 31 The Pfizer-BioNTech vaccine is highly effective in adolescents. March 31 Some Johnson & Johnson doses are delayed by a U.S. factory mix-up. March 30 Chinaʼs Jiangsu Rec-Biotechnology enters Phase 1. March 27 Turkeyʼs Middle East Technical and Bilkent University begin Phase 1. March 25 A vaccine from Chinaʼs Zhongyianke Biotech enters Phase 2. March 23 U.S. officials question the completeness of AstraZenecaʼs trial results. March 23 Daiichi Sankyo and the University of Tokyo enter Phase 1/2. March 22 AstraZenecaʼs vaccine is found to be 79% effective in a large U.S. -

Vaccines Innovative
2013 MeDiciNes iN DeVelopmeNT REPORT Vaccines A Report on the Prevention and Treatment of Disease Through Vaccines PRESENTED BY AMERICA’s biopharmACEUTICAL RESEARCH COMPANIES Nearly 300 Vaccines Are in Development; Research Focuses on Prevention and Treatment For many years, vaccines have been used • A monoclonal antibody vaccine that to successfully prevent devastating infec- targets both pandemic and seasonal tious diseases such as smallpox, measles influenza. and polio. According to data from the • A genetically-modified vaccine designed Vaccines in Development* U.S. Centers for Disease Control and for the treatment of pancreatic cancer. Prevention (CDC), 10 infectious diseases Application Submitted have been at least 90 percent eradicated • An irradiated vaccine for protection in the United States thanks to vaccines. Phase III against malaria. This has protected millions of children Phase II and families from needless illness. The development and regulatory path that these vaccine candidates face is Phase I These public health triumphs illustrate the complex. As with the development of all major contributions that vaccines have drugs, the majority of vaccines must pre- 137 made in saving countless lives around vail through years of clinical testing before the world. In the past several years, they can be approved for use by the gen- through our growing understanding of eral public. However, advances in other the molecular underpinnings of disease scientific fields, such as genomics and and technological advances, many new manufacturing technologies, are becom- vaccines have been developed, including ing increasingly useful in the development one against human papillomavirus (HPV) of new vaccines. The continued efforts 99 infections that can lead to cervical cancer, of researchers within biopharmaceutical a vaccine to guard against the anthrax companies and across the ecosystem, virus before exposure, and a vaccine to who are pursuing new techniques and prevent pneumococcal infections in high- strategies in vaccine development, cre- risk populations. -

Vaccine Beliefs of Complementary and Alternative Medical (CAM) Providers in Oregon
AN ABSTRACT OF THE DISSERTATION OF Sandra J. Bean for the degree of Doctor of Philosophy in Public Health presented on May 21, 2014. Title: Vaccine Beliefs of Complementary and Alternative Medical (CAM) Providers in Oregon Abstract approved: ______________________________________________________ Joseph A. Catania Health care providers, including complementary and alternative medical (CAM) practitioners, exert a significant influence on parental pediatric vaccination decisions. Use of CAM therapies is increasing in Oregon. Concomitantly, there has been a decade-long increase in parental vaccine refusal in Oregon, rising from 1 to 5 percent from 2000–2009. For example, in some Oregon schools, 70% of children are unvaccinated. I conducted hour-long interviews with 36 practitioners from five CAM modalities (i.e., acupuncture, chiropractic, homeopathy, midwifery, and naturopathy) to explore a range of associations between vaccination perceptions and vaccine recommendations to others. Data collected from the semi-structured interviews were sorted and analyzed by themes. Perceived susceptibility to and severity of either an illness or a vaccine as well as perceptions of vaccine efficacy, benefits, and risks influenced CAM provider vaccine recommendations to parents about vaccinating their children. The contextual factors of immunology beliefs, personal experience, reference group norms, and beliefs about industry and government were found to a lesser extent to influence whether a CAM provider promoted or opposed pediatric vaccination. The results