Epipen Administration Tip Sheet Part 2
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
An Intramuscular Injection Is an Injection Given Directly Into The
Depo Lupron and Testosterone are both given by intramuscular injection. The following is a guideline on their administration. Eileen Durham, RN, NP Version 12 Jan 2010 Description Intramuscular (IM) injections are given directly into the central area of selected muscles. There are a number of sites that are suitable for IM injections; there are three sites that are most commonly used in this procedure described below. The volume of viscosity of the medication to be injected determines the site that should be used. IM injections cause stretching of the muscle fiber so the larger the muscle used the less discomfort. Intramuscular Injection Sites Depo Lupron Depo Lupron 3 month preparation should only be injected into the Gluteus medius due to the viscosity and volume of the medication approx. 1.5 – 2 cc. Depo Lupron 1 month preparation can be injection into the vastus lateralis or the gluteus medius. Testosterone Testosterone administered to adolescents and adults can be injected into any of the sites listed below, as long as the volume is 1 cc or less. For a volume of 1.5 use the vastus lateralis or Gluteus medius, if the volume is 2 cc you must you the largest muscle the Gluteus medius. If the volume is greater then 2 cc you must divide the dose and give 2 injections as the maximum volume in the Gluteal muscle is 2 cc. Testosterone administered to infants and toddlers use only the anteriolateral aspect of the thigh. Deltoid muscle The deltoid muscle located laterally on the upper arm can be used for intramuscular injections. -

Emerging Evidence for a Central Epinephrine-Innervated A1- Adrenergic System That Regulates Behavioral Activation and Is Impaired in Depression
Neuropsychopharmacology (2003) 28, 1387–1399 & 2003 Nature Publishing Group All rights reserved 0893-133X/03 $25.00 www.neuropsychopharmacology.org Perspective Emerging Evidence for a Central Epinephrine-Innervated a1- Adrenergic System that Regulates Behavioral Activation and is Impaired in Depression ,1 1 1 1 1 Eric A Stone* , Yan Lin , Helen Rosengarten , H Kenneth Kramer and David Quartermain 1Departments of Psychiatry and Neurology, New York University School of Medicine, New York, NY, USA Currently, most basic and clinical research on depression is focused on either central serotonergic, noradrenergic, or dopaminergic neurotransmission as affected by various etiological and predisposing factors. Recent evidence suggests that there is another system that consists of a subset of brain a1B-adrenoceptors innervated primarily by brain epinephrine (EPI) that potentially modulates the above three monoamine systems in parallel and plays a critical role in depression. The present review covers the evidence for this system and includes findings that brain a -adrenoceptors are instrumental in behavioral activation, are located near the major monoamine cell groups 1 or target areas, receive EPI as their neurotransmitter, are impaired or inhibited in depressed patients or after stress in animal models, and a are restored by a number of antidepressants. This ‘EPI- 1 system’ may therefore represent a new target system for this disorder. Neuropsychopharmacology (2003) 28, 1387–1399, advance online publication, 18 June 2003; doi:10.1038/sj.npp.1300222 Keywords: a1-adrenoceptors; epinephrine; motor activity; depression; inactivity INTRODUCTION monoaminergic systems. This new system appears to be impaired during stress and depression and thus may Depressive illness is currently believed to result from represent a new target for this disorder. -

Injection Technique 1: Administering Drugs Via the Intramuscular Route
Copyright EMAP Publishing 2018 This article is not for distribution except for journal club use Clinical Practice Keywords Intramuscular injection/ Medicine administration/Absorption Practical procedures This article has been Injection technique double-blind peer reviewed Injection technique 1: administering drugs via the intramuscular route rugs administered by the intra- concerns that nurses are still performing Author Eileen Shepherd is clinical editor muscular (IM) route are depos- outdated and ritualistic practice relating to at Nursing Times. ited into vascular muscle site selection, aspirating back on the syringe Dtissue, which allows for rapid (Greenway, 2014) and skin cleansing. Abstract The intramuscular route allows absorption into the circulation (Dough- for rapid absorption of drugs into the erty and Lister, 2015; Ogston-Tuck, 2014). Site selection circulation. Using the correct injection Complications of poorly performed IM Four muscle sites are recommended for IM technique and selecting the correct site injection include: administration: will minimise the risk of complications. l Pain – strategies to reduce this are l Vastus lateris; outlined in Box 1; l Rectus femoris Citation Shepherd E (2018) Injection l Bleeding; l Deltoid; technique 1: administering drugs via l Abscess formation; l Ventrogluteal (Fig 1, Table 1). the intramuscular route. Nursing Times l Cellulitis; Traditionally the dorsogluteal (DG) [online]; 114: 8, 23-25. l Muscle fibrosis; muscle was used for IM injections but this l Injuries to nerves and blood vessels muscle is in close proximity to a major (Small, 2004); blood vessel and nerves, with sciatic nerve l Inadvertent intravenous (IV) access. injury a recognised complication (Small, These complications can be avoided if 2004). -

Self-Administering a Vitamin B12 Injection
Self-administering a Vitamin B12 injection This is usually given as an intramuscular injection, every 2-3 months. Alternatives to an intramuscular injection are: Oral Vitamin B12 at a dose of at least 1000mcg per day. • Available as tablets or a spray. They can be bought over the counter and available at most health food stores and pharmacies. • Oral Vitamin B12 is not recommended if: - If you have a bowel condition such as inflammatory bowel disease, Coeliac disease, small bowel overgrowth, bile acid malabsorption and short bowel (you will require injections) - You require an initial loading of B12 (soon after diagnosis) It is important to monitor your symptoms if you change to oral B12. If symptoms return, then the oral/sublingual dose can be increased to 2000mcg or you may need to consider starting back on injections. https://www.hollandandbarrett.com/shop/product/betteryou-pure-energy-b12-boost-oral-spray-60099160?skuid=099160 https://www.hollandandbarrett.com/search?query=%20Vitamin%20B12%20Tablets&utm_medium=cpc&utm_source=google&isSearch=true# gclid=EAIaIQobChMIh5nLgOH26AIVxLTtCh0JIA_GEAAYASAAEgJL-fD_BwE&gclsrc=aw.ds Subcutaneous (SC) injection; this is off-licence but still effective. It is considered an easier method of administration. It is how insulin and blood thinning medication are usually self-administered. Equipment needed to self-inject - Prescribed medicine - 1 syringe (2 ml) - 2 needles (1 for drawing up the drug and 1 for administration – you can use the same size needle for both). o For an IM injection; the needle gauge should be 19-25. The needle length is 1- 1 ½ inches (up to 3 inches for larger adults) o For a SC injection; the needle gauge should be 25-27. -

Cocaine Intoxication and Hypertension
THE EMCREG-INTERNATIONAL CONSENSUS PANEL RECOMMENDATIONS Cocaine Intoxication and Hypertension Judd E. Hollander, MD From the Department of Emergency Medicine, University of Pennsylvania, Philadelphia, PA. 0196-0644/$-see front matter Copyright © 2008 by the American College of Emergency Physicians. doi:10.1016/j.annemergmed.2007.11.008 [Ann Emerg Med. 2008;51:S18-S20.] with cocaine intoxication is analogous to that of the patient with hypertension: the treatment should be geared toward the Cocaine toxicity has been reported in virtually all organ patient’s presenting complaint. systems. Many of the adverse effects of cocaine are similar to When the medical history is clear and symptoms are mild, adverse events that can result from either acute hypertensive laboratory evaluation is usually unnecessary. In contrast, if the crisis or chronic effects of hypertension. Recognizing when the patient has severe toxicity, evaluation should be geared toward specific disease requires treatment separate from cocaine toxicity the presenting complaint. Laboratory evaluation may include a is paramount to the treatment of patients with cocaine CBC count; determination of electrolyte, glucose, blood urea intoxication. nitrogen, creatine kinase, and creatinine levels; arterial blood The initial physiologic effect of cocaine on the cardiovascular gas analysis; urinalysis; and cardiac marker determinations. system is a transient bradycardia as a result of stimulation of the Increased creatine kinase level occurs with rhabdomyolysis. vagal nuclei. Tachycardia typically ensues, predominantly from Cardiac markers are increased in myocardial infarction. Cardiac increased central sympathetic stimulation. Cocaine has a troponin I is preferred to identify acute myocardial13 infarction. cardiostimulatory effect through sensitization to epinephrine A chest radiograph should be obtained in patients with and norepinephrine. -

Atopic Children and Use of Prescribed Medication: a Comprehensive Study in General Practice
RESEARCH ARTICLE Atopic children and use of prescribed medication: A comprehensive study in general practice David H. J. Pols1*, Mark M. J. Nielen2, Arthur M. Bohnen1, Joke C. Korevaar2, Patrick J. E. Bindels1 1 Department of General Practice, Erasmus MC, University Medical Center Rotterdam, Rotterdam, The Netherlands, 2 NIVEL, Netherlands Institute for Health Services Research, Utrecht, The Netherlands a1111111111 * [email protected] a1111111111 a1111111111 a1111111111 Abstract a1111111111 Purpose A comprehensive and representative nationwide general practice database was explored to study associations between atopic disorders and prescribed medication in children. OPEN ACCESS Citation: Pols DHJ, Nielen MMJ, Bohnen AM, Korevaar JC, Bindels PJE (2017) Atopic children Method and use of prescribed medication: A All children aged 0±18 years listed in the NIVEL Primary Care Database in 2014 were comprehensive study in general practice. PLoS ONE 12(8): e0182664. https://doi.org/10.1371/ selected. Atopic children with atopic eczema, asthma and allergic rhinitis (AR) were journal.pone.0182664 matched with controls (not diagnosed with any of these disorders) within the same general Editor: Anthony Peter Sampson, University of practice on age and gender. Logistic regression analyses were performed to study the differ- Southampton School of Medicine, UNITED ences in prescribed medication between both groups by calculating odds ratios (OR); 93 dif- KINGDOM ferent medication groups were studied. Received: March 24, 2017 Accepted: July 13, 2017 Results Published: August 24, 2017 A total of 45,964 children with at least one atopic disorder were identified and matched with Copyright: © 2017 Pols et al. This is an open controls. Disorder-specific prescriptions seem to reflect evidence-based medicine guide- access article distributed under the terms of the lines for atopic eczema, asthma and AR. -

Efficacy/Risk Profile of Triamcinolone Acetonide in Severe Asthma
ARTICLE IN PRESS Respiratory Medicine CME (2008) 1, 111–115 respiratory MEDICINE CME CASE REPORT Efficacy/risk profile of triamcinolone acetonide in severe asthma: Lessons from one case study Cesar Picadoà Servei de Pneumologia, Hospital Clinic, Universitat de Barcelona, Villarroel 170, 08036 Barcelona, Spain Received 26 November 2007; accepted 22 January 2008 KEYWORDS Summary Insulin; Some prednisone-resistant asthma patients respond to intramuscular triamcinolone Refractory asthma; acetonide (TA). The use of TA has been questioned on the basis of its potential toxicity. Severe asthma; TA’s ratio of clinical efficacy to systemic effects compared with oral prednisone has not Steroid-dependent been clearly defined. asthma; We report the case of a prednisone-insensitive severe asthma patient with insulin- Triamcinolone dependent diabetes, hypertension and diabetic retinopathy treated with repeated sessions acetonide of laser therapy. By daily monitoring the needs of insulin doses we compared the systemic effects of prednisone and TA. The analysis of the balance between systemic effects (insulin doses) and beneficial effects (PEF values) show that TA has a better benefit/risk profile than prednisone. The patient has been followed up for 3 years and has received injections of TA repeated at intervals ranging from 21 to 60 days according to patient response and the evolution of PEF and clinical symptoms. During this period the patient lost 3 kg of weight, while presenting increased bone mineral density, normalized arterial tension, and improved proliferative diabetic retinopathy. The present case report shows that TA has a better benefit/risk profile than prednisone. TA can be a valuable alternative in the control of some patients with severe prednisone- resistant asthma. -

How to Administer Intramuscular, Intradermal, and Intranasal Influenza Vaccines
How to Administer Intramuscular, Intradermal, and Intranasal Influenza Vaccines Intramuscular injection (IM) Intradermal administration (ID) Intranasal administration (NAS) Inactivated Influenza Vaccines (IIV), including Inactivated Influenza Vaccine (IIV) Live Attenuated Influenza Vaccine (LAIV) recombinant hemagglutinin influenza vaccine (RIV3) 1 Gently shake the microinjection system before 1 FluMist (LAIV) is for intranasal administration 1 Use a needle long enough to reach deep into administering the vaccine. only. Do not inject FluMist. the muscle. Infants age 6 through 11 mos: 1"; 1 through 2 yrs: 1–1¼"; children and adults 2 Hold the system by placing the 2 Remove rubber tip protector. Do not remove 3 yrs and older: 1–1½". thumb and middle finger on dose-divider clip at the other end of the sprayer. the finger pads; the index finger 2 With your left hand*, bunch up the muscle. should remain free. 3 With the patient in an upright position, place the tip just inside the nostril 3 With your right hand*, insert the needle at a 3 Insert the needle perpendicular to the skin, to ensure LAIV is delivered into 90° angle to the skin with a quick thrust. in the region of the deltoid, in a short, quick the nose. The patient should movement. breathe normally. 4 Push down on the plunger and inject the entire contents of the syringe. There is no need to 4 Once the needle has been 4 With a single motion, depress plunger as aspirate. inserted, maintain light pressure rapidly as possible until the dose-divider clip on the surface of the skin and prevents you from going further. -

Epipen Injection Label
EPIPEN®- epinephrine injection EPIPEN Jr®- epinephrine injection Mylan Specialty L.P. ---------- EpiPen® (epinephrine) Auto-Injector 0.3 mg EpiPen® = one dose of 0.30 mg epinephrine (USP, 1:1000, 0.3 mL) EpiPen Jr® (epinephrine) Auto-Injector 0.15 mg EpiPenJr® = one dose of 0.15 mg epinephrine (USP, 1:2000, 0.3 mL) DESCRIPTION Each EpiPen® Auto-Injector delivers a single dose of 0.3 mg epinephrine injection, USP, 1:1000 (0.3 mL) in a sterile solution. Each EpiPen Jr® Auto-Injector delivers a single dose of 0.15 mg epinephrine injection, USP, 1:2000 (0.3 mL) in a sterile solution. The EpiPen and EpiPen Jr Auto-Injectors each contain 2 mL epinephrine solution. Approximately 1.7 mL remains in the auto- injector after activation and cannot be used. Each 0.3 mL in the EpiPen Auto-Injector contains 0.3 mg epinephrine, 1.8 mg sodium chloride, 0.5 mg sodium metabisulfite, hydrochloric acid to adjust pH, and Water for Injection. The pH range is 2.2-5.0. Each 0.3 mL in the EpiPen Jr Auto-Injector contains 0.15 mg epinephrine, 1.8 mg sodium chloride, 0.5 mg sodium metabisulfite, hydrochloric acid to adjust pH, and Water for Injection. The pH range is 2.2-5.0. Epinephrine is a sympathomimetic catecholamine. Chemically, epinephrine is B-(3, 4-dihydroxyphenyl)-a-methyl-aminoethanol, with the following structure: Epinephrine solution deteriorates rapidly on exposure to air or light, turning pink from oxidation to adrenochrome and brown from the Reference ID: 3176782 formation of melanin. -

Intramuscular Injections: GUIDELINES for BEST PRACTICE B
2.1 ANCC Contact Hours Abstract The administration of injec- tions is a fundamental nursing skill; however, it is not without risk. Children receive numer- ous vaccines, and pediatric nurses administer the major- ity of these vaccines via the intramuscular route, and thus must be knowledgeable about safe and evidence-based immunization programs. Nurses may not be aware of the potential consequences associated with poor injection practices, and historically have relied on their basic nursing training or the advice of colleagues as a substitute for newer evidence about how to administer injections today. Evidence-based nursing practice requires pediatric nurses to review current literature to establish best practices and thus improved patient outcomes. Key words: Child; Evidence-based nursing; Injection intramuscu- lar; Vaccination. AbbyPediatric Rishovd, DNP, PNP Intramuscular Injections: GUIDELINES FOR BEST PRACTICE B. Boissonnet / BSIP SA / Alamy Alamy / B. Boissonnet / BSIP SA March/April 2014 MCN 107 Copyright © 2014 Lippincott Williams & Wilkins. Unauthorized reproduction of this article is prohibited. ver the past decade, the childhood vaccine (2011), and the World Health Organization (2004) have schedule has increased in complexity. De- now all strongly discouraged the practice of aspirating for pending upon the combinations adminis- blood when administering an IM injection. A randomized tered, children now may receive as many control trial found that the standard injection technique as 24 immunizations via intramuscular consisting of slow aspiration and injection followed by O(IM) injection by 2 years of age (Centers withdrawal of the needle was more painful and took lon- for Disease Control and Prevention [CDC], 2011). Pedi- ger than rapid injection without aspiration (Ipp, Taddio, atric nurses administer the majority of injections and thus Sam, Gladbach, & Parkin, 2007). -
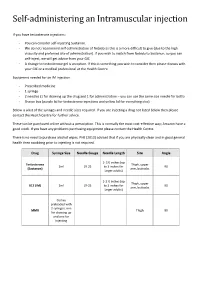
Self-Administering an Intramuscular Injection
Self-administering an Intramuscular injection If you have testosterone injections: - You can consider self-injecting Sustanon. - We do not recommend self-administration of Nebido as this is a more difficult to give (due to the high viscosity and preferred site of administration). If you wish to switch from Nebido to Sustanon, so you can self-inject, we will get advice from your GIC - A change to testosterone gel is an option. If this is something you wish to consider then please discuss with your GIC or a medical professional at the Health Centre. Equipment needed for an IM injection - Prescribed medicine - 1 syringe - 2 needles (1 for drawing up the drug and 1 for administration – you can use the same size needle for both) - Sharps box (purple lid for testosterone injections and yellow lid for everything else) Below is a list of the syringes and needle sizes required. If you are injecting a drug not listed below then please contact the Health Centre for further advice. These can be purchased online without a prescription. This is normally the most cost-effective way; Amazon have a good stock. If you have any problems purchasing equipment please contact the Health Centre. There is no need to purchase alcohol wipes. PHE (2013) advised that if you are physically clean and in good general health then swabbing prior to injecting is not required. Drug Syringe Size Needle Gauge Needle Length Site Angle 1- 1½ inches (up Testosterone Thigh, upper 2ml 19-25 to 3 inches for 90 (Sustanon) arm, buttocks larger adults) 1-1 ½ inches (up Thigh, upper B12 (IM) 2ml 19-25 to 3 inches for 90 arm, buttocks larger adults) Comes preloaded with 2 syringes, one MMR Thigh 90 for drawing up and one for injecting Where to inject The easiest site when self-administering an IM injection is the middle third of the vastus lateralis muscle of the thigh. -

Patients on Beta-Blockers
Systemic Allergic & Immunoglobulin Disorders Bryan L. Martin, DO, MMAS, FACAAI, FAAAAI, FACP, FACOI Emeritus Professor of Medicine and Pediatrics President, American College of Allergy, Asthma & Immunology Disclosures . None 2 Objectives . Pass the boards! . Recognize anaphylaxis and understand the new guidelines for the treatment of anaphylaxis . Compare and contrast anaphylactic and anaphylactoid reactions 3 The Patient . 45 year old woman goes to lunch with her friends . While eating she develops GI upset, dizziness, shortness of breath and hives . Her friends drive her to your nearby clinic Initial Evaluation . Your nurse gets the history of sudden onset of GI upset, dizziness, shortness of breath and hives while eating a shrimp salad. The patient now has audible wheezing, tachycardia, and a blood pressure of 110/60 . Your initial response . Take a detailed history of everything the patient had to eat for lunch . Ask if she has a history of any medical issues such as asthma, diabetes or heart disease . Give nebulized beta agonist . Give 50 mg of oral diphenhydramine . Give 0.3 ml of IM epinephrine Anaphylaxis presentation . Your nurse gets the history of sudden onset of GI upset, dizziness, shortness of breath and hives while eating lunch. The patient now has audible wheezing, tachycardia, and a blood pressure of 110/60 . Your initial response . Take a detailed history of everything the patient had to eat for lunch . Ask if she has a history of any medical issues such as asthma, diabetes or heart disease . Give nebulized beta agonist . Give 50 mg of oral diphenhydramine . Give 0.3 ml of IM epinephrine Initial Response to anaphylaxis .