Bose–Einstein Condensation and Superfluidity (Reference: Robert B
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Lecture 3: Fermi-Liquid Theory 1 General Considerations Concerning Condensed Matter
Phys 769 Selected Topics in Condensed Matter Physics Summer 2010 Lecture 3: Fermi-liquid theory Lecturer: Anthony J. Leggett TA: Bill Coish 1 General considerations concerning condensed matter (NB: Ultracold atomic gasses need separate discussion) Assume for simplicity a single atomic species. Then we have a collection of N (typically 1023) nuclei (denoted α,β,...) and (usually) ZN electrons (denoted i,j,...) interacting ∼ via a Hamiltonian Hˆ . To a first approximation, Hˆ is the nonrelativistic limit of the full Dirac Hamiltonian, namely1 ~2 ~2 1 e2 1 Hˆ = 2 2 + NR −2m ∇i − 2M ∇α 2 4πǫ r r α 0 i j Xi X Xij | − | 1 (Ze)2 1 1 Ze2 1 + . (1) 2 4πǫ0 Rα Rβ − 2 4πǫ0 ri Rα Xαβ | − | Xiα | − | For an isolated atom, the relevant energy scale is the Rydberg (R) – Z2R. In addition, there are some relativistic effects which may need to be considered. Most important is the spin-orbit interaction: µ Hˆ = B σ (v V (r )) (2) SO − c2 i · i × ∇ i Xi (µB is the Bohr magneton, vi is the velocity, and V (ri) is the electrostatic potential at 2 3 2 ri as obtained from HˆNR). In an isolated atom this term is o(α R) for H and o(Z α R) for a heavy atom (inner-shell electrons) (produces fine structure). The (electron-electron) magnetic dipole interaction is of the same order as HˆSO. The (electron-nucleus) hyperfine interaction is down relative to Hˆ by a factor µ /µ 10−3, and the nuclear dipole-dipole SO n B ∼ interaction by a factor (µ /µ )2 10−6. -

The Development of the Science of Superconductivity and Superfluidity
Universal Journal of Physics and Application 1(4): 392-407, 2013 DOI: 10.13189/ujpa.2013.010405 http://www.hrpub.org Superconductivity and Superfluidity-Part I: The development of the science of superconductivity and superfluidity in the 20th century Boris V.Vasiliev ∗Corresponding Author: [email protected] Copyright ⃝c 2013 Horizon Research Publishing All rights reserved. Abstract Currently there is a common belief that the explanation of superconductivity phenomenon lies in understanding the mechanism of the formation of electron pairs. Paired electrons, however, cannot form a super- conducting condensate spontaneously. These paired electrons perform disorderly zero-point oscillations and there are no force of attraction in their ensemble. In order to create a unified ensemble of particles, the pairs must order their zero-point fluctuations so that an attraction between the particles appears. As a result of this ordering of zero-point oscillations in the electron gas, superconductivity arises. This model of condensation of zero-point oscillations creates the possibility of being able to obtain estimates for the critical parameters of elementary super- conductors, which are in satisfactory agreement with the measured data. On the another hand, the phenomenon of superfluidity in He-4 and He-3 can be similarly explained, due to the ordering of zero-point fluctuations. It is therefore established that both related phenomena are based on the same physical mechanism. Keywords superconductivity superfluidity zero-point oscillations 1 Introduction 1.1 Superconductivity and public Superconductivity is a beautiful and unique natural phenomenon that was discovered in the early 20th century. Its unique nature comes from the fact that superconductivity is the result of quantum laws that act on a macroscopic ensemble of particles as a whole. -

Guide to Understanding Condensation
Guide to Understanding Condensation The complete Andersen® Owner-To-Owner™ limited warranty is available at: www.andersenwindows.com. “Andersen” is a registered trademark of Andersen Corporation. All other marks where denoted are marks of Andersen Corporation. © 2007 Andersen Corporation. All rights reserved. 7/07 INTRODUCTION 2 The moisture that suddenly appears in cold weather on the interior We have created this brochure to answer questions you may have or exterior of window and patio door glass can block the view, drip about condensation, indoor humidity and exterior condensation. on the floor or freeze on the glass. It can be an annoying problem. We’ll start with the basics and offer solutions and alternatives While it may seem natural to blame the windows or doors, interior along the way. condensation is really an indication of excess humidity in the home. Exterior condensation, on the other hand, is a form of dew — the Should you run into problems or situations not covered in the glass simply provides a surface on which the moisture can condense. following pages, please contact your Andersen retailer. The important thing to realize is that if excessive humidity is Visit the Andersen website: www.andersenwindows.com causing window condensation, it may also be causing problems elsewhere in your home. Here are some other signs of excess The Andersen customer service toll-free number: 1-888-888-7020. humidity: • A “damp feeling” in the home. • Staining or discoloration of interior surfaces. • Mold or mildew on surfaces or a “musty smell.” • Warped wooden surfaces. • Cracking, peeling or blistering interior or exterior paint. -

Phys 446: Solid State Physics / Optical Properties Lattice Vibrations
Solid State Physics Lecture 5 Last week: Phys 446: (Ch. 3) • Phonons Solid State Physics / Optical Properties • Today: Einstein and Debye models for thermal capacity Lattice vibrations: Thermal conductivity Thermal, acoustic, and optical properties HW2 discussion Fall 2007 Lecture 5 Andrei Sirenko, NJIT 1 2 Material to be included in the test •Factors affecting the diffraction amplitude: Oct. 12th 2007 Atomic scattering factor (form factor): f = n(r)ei∆k⋅rl d 3r reflects distribution of electronic cloud. a ∫ r • Crystalline structures. 0 sin()∆k ⋅r In case of spherical distribution f = 4πr 2n(r) dr 7 crystal systems and 14 Bravais lattices a ∫ n 0 ∆k ⋅r • Crystallographic directions dhkl = 2 2 2 1 2 ⎛ h k l ⎞ 2πi(hu j +kv j +lw j ) and Miller indices ⎜ + + ⎟ •Structure factor F = f e ⎜ a2 b2 c2 ⎟ ∑ aj ⎝ ⎠ j • Definition of reciprocal lattice vectors: •Elastic stiffness and compliance. Strain and stress: definitions and relation between them in a linear regime (Hooke's law): σ ij = ∑Cijklε kl ε ij = ∑ Sijklσ kl • What is Brillouin zone kl kl 2 2 C •Elastic wave equation: ∂ u C ∂ u eff • Bragg formula: 2d·sinθ = mλ ; ∆k = G = eff x sound velocity v = ∂t 2 ρ ∂x2 ρ 3 4 • Lattice vibrations: acoustic and optical branches Summary of the Last Lecture In three-dimensional lattice with s atoms per unit cell there are Elastic properties – crystal is considered as continuous anisotropic 3s phonon branches: 3 acoustic, 3s - 3 optical medium • Phonon - the quantum of lattice vibration. Elastic stiffness and compliance tensors relate the strain and the Energy ħω; momentum ħq stress in a linear region (small displacements, harmonic potential) • Concept of the phonon density of states Hooke's law: σ ij = ∑Cijklε kl ε ij = ∑ Sijklσ kl • Einstein and Debye models for lattice heat capacity. -

Chapter 3 Bose-Einstein Condensation of an Ideal
Chapter 3 Bose-Einstein Condensation of An Ideal Gas An ideal gas consisting of non-interacting Bose particles is a ¯ctitious system since every realistic Bose gas shows some level of particle-particle interaction. Nevertheless, such a mathematical model provides the simplest example for the realization of Bose-Einstein condensation. This simple model, ¯rst studied by A. Einstein [1], correctly describes important basic properties of actual non-ideal (interacting) Bose gas. In particular, such basic concepts as BEC critical temperature Tc (or critical particle density nc), condensate fraction N0=N and the dimensionality issue will be obtained. 3.1 The ideal Bose gas in the canonical and grand canonical ensemble Suppose an ideal gas of non-interacting particles with ¯xed particle number N is trapped in a box with a volume V and at equilibrium temperature T . We assume a particle system somehow establishes an equilibrium temperature in spite of the absence of interaction. Such a system can be characterized by the thermodynamic partition function of canonical ensemble X Z = e¡¯ER ; (3.1) R where R stands for a macroscopic state of the gas and is uniquely speci¯ed by the occupa- tion number ni of each single particle state i: fn0; n1; ¢ ¢ ¢ ¢ ¢ ¢g. ¯ = 1=kBT is a temperature parameter. Then, the total energy of a macroscopic state R is given by only the kinetic energy: X ER = "ini; (3.2) i where "i is the eigen-energy of the single particle state i and the occupation number ni satis¯es the normalization condition X N = ni: (3.3) i 1 The probability -

Sounds of a Supersolid A
NEWS & VIEWS RESEARCH hypothesis came from extensive population humans, implying possible mosquito exposure long-distance spread of insecticide-resistant time-series analysis from that earlier study5, to malaria parasites and the potential to spread mosquitoes, worsening an already dire situ- which showed beyond reasonable doubt that infection over great distances. ation, given the current spread of insecticide a mosquito vector species called Anopheles However, the authors failed to detect resistance in mosquito populations. This would coluzzii persists locally in the dry season in parasite infections in their aerially sampled be a matter of great concern because insecticides as-yet-undiscovered places. However, the malaria vectors, a result that they assert is to be are the best means of malaria control currently data were not consistent with this outcome for expected given the small sample size and the low available8. However, long-distance migration other malaria vectors in the study area — the parasite-infection rates typical of populations of could facilitate the desirable spread of mosqui- species Anopheles gambiae and Anopheles ara- malaria vectors. A problem with this argument toes for gene-based methods of malaria-vector biensis — leaving wind-powered long-distance is that the typical infection rates they mention control. One thing is certain, Huestis and col- migration as the only remaining possibility to are based on one specific mosquito body part leagues have permanently transformed our explain the data5. (salivary glands), rather than the unknown but understanding of African malaria vectors and Both modelling6 and genetic studies7 undoubtedly much higher infection rates that what it will take to conquer malaria. -

It's Just a Phase!
BASIS Lesson Plan Lesson Name: It’s Just a Phase! Grade Level Connection(s) NGSS Standards: Grade 2, Physical Science FOSS CA Edition: Grade 3 Physical Science: Matter and Energy Module *Note to teachers: Detailed standards connections can be found at the end of this lesson plan. Teaser/Overview Properties of matter are illustrated through a series of demonstrations and hands-on explorations. Students will learn to identify solids, liquids, and gases. Water will be used to demonstrate the three phases. Students will learn about sublimation through a fun experiment with dry ice (solid CO2). Next, they will compare the propensity of several liquids to evaporate. Finally, they will learn about freezing and melting while making ice cream. Lesson Objectives ● Students will be able to identify the three states of matter (solid, liquid, gas) based on the relative properties of those states. ● Students will understand how to describe the transition from one phase to another (melting, freezing, evaporation, condensation, sublimation) ● Students will learn that matter can change phase when heat/energy is added or removed Vocabulary Words ● Solid: A phase of matter that is characterized by a resistance to change in shape and volume. ● Liquid: A phase of matter that is characterized by a resistance to change in volume; a liquid takes the shape of its container ● Gas: A phase of matter that can change shape and volume ● Phase change: Transformation from one phase of matter to another ● Melting: Transformation from a solid to a liquid ● Freezing: Transformation -

High Pressure Band Structure, Density of States, Structural Phase Transition and Metallization in Cds
Chemical and Materials Engineering 5(1): 8-13, 2017 http://www.hrpub.org DOI: 10.13189/cme.2017.050102 High Pressure Band Structure, Density of States, Structural Phase Transition and Metallization in CdS J. Jesse Pius1, A. Lekshmi2, C. Nirmala Louis2,* 1Rohini College of Engineering, Nagercoil, Kanyakumari District, India 2Research Center in Physics, Holy Cross College, India Copyright©2017 by authors, all rights reserved. Authors agree that this article remains permanently open access under the terms of the Creative Commons Attribution License 4.0 International License Abstract The electronic band structure, density of high pressure studies due to the development of different states, metallization and structural phase transition of cubic designs of diamond anvil cell (DAC). With a modern DAC, zinc blende type cadmium sulphide (CdS) is investigated it is possible to reach pressures of 2 Mbar (200 GPa) using the full potential linear muffin-tin orbital (FP-LMTO) routinely and pressures of 5 Mbar (500 GPa) or higher is method. The ground state properties and band gap values achievable [3]. At such pressures, materials are reduced to are compared with the experimental results. The fractions of their original volumes. With this reduction in equilibrium lattice constant, bulk modulus and its pressure inter atomic distances; significant changes in bonding and derivative and the phase transition pressure at which the structure as well as other properties take place. The increase compounds undergo structural phase transition from ZnS to of pressure means the significant decrease in volume, which NaCl are predicted from the total energy calculations. The results in the change of electronic states and crystal structure. -
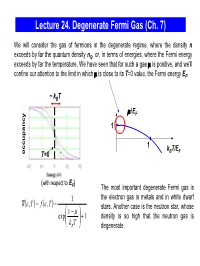
Lecture 24. Degenerate Fermi Gas (Ch
Lecture 24. Degenerate Fermi Gas (Ch. 7) We will consider the gas of fermions in the degenerate regime, where the density n exceeds by far the quantum density nQ, or, in terms of energies, where the Fermi energy exceeds by far the temperature. We have seen that for such a gas μ is positive, and we’ll confine our attention to the limit in which μ is close to its T=0 value, the Fermi energy EF. ~ kBT μ/EF 1 1 kBT/EF occupancy T=0 (with respect to E ) F The most important degenerate Fermi gas is 1 the electron gas in metals and in white dwarf nε()(),, T= f ε T = stars. Another case is the neutron star, whose ε⎛ − μ⎞ exp⎜ ⎟ +1 density is so high that the neutron gas is ⎝kB T⎠ degenerate. Degenerate Fermi Gas in Metals empty states ε We consider the mobile electrons in the conduction EF conduction band which can participate in the charge transport. The band energy is measured from the bottom of the conduction 0 band. When the metal atoms are brought together, valence their outer electrons break away and can move freely band through the solid. In good metals with the concentration ~ 1 electron/ion, the density of electrons in the electron states electron states conduction band n ~ 1 electron per (0.2 nm)3 ~ 1029 in an isolated in metal electrons/m3 . atom The electrons are prevented from escaping from the metal by the net Coulomb attraction to the positive ions; the energy required for an electron to escape (the work function) is typically a few eV. -

Lecture Notes for Quantum Matter
Lecture Notes for Quantum Matter MMathPhys c Professor Steven H. Simon Oxford University July 24, 2019 Contents 1 What we will study 1 1.1 Bose Superfluids (BECs, Superfluid He, Superconductors) . .1 1.2 Theory of Fermi Liquids . .2 1.3 BCS theory of superconductivity . .2 1.4 Special topics . .2 2 Introduction to Superfluids 3 2.1 Some History and Basics of Superfluid Phenomena . .3 2.2 Landau and the Two Fluid Model . .6 2.2.1 More History and a bit of Physics . .6 2.2.2 Landau's Two Fluid Model . .7 2.2.3 More Physical Effects and Their Two Fluid Pictures . .9 2.2.4 Second Sound . 12 2.2.5 Big Questions Remaining . 13 2.3 Curl Free Constraint: Introducing the Superfluid Order Parameter . 14 2.3.1 Vorticity Quantization . 15 2.4 Landau Criterion for Superflow . 17 2.5 Superfluid Density . 20 2.5.1 The Andronikoshvili Experiment . 20 2.5.2 Landau's Calculation of Superfluid Density . 22 3 Charged Superfluid ≈ Superconductor 25 3.1 London Theory . 25 3.1.1 Meissner-Ochsenfeld Effect . 27 3 3.1.2 Quantum Input and Superfluid Order Parameter . 29 3.1.3 Superconducting Vortices . 30 3.1.4 Type I and Type II superconductors . 32 3.1.5 How big is Hc ............................... 33 4 Microscopic Theory of Bosons 37 4.1 Mathematical Preliminaries . 37 4.1.1 Second quantization . 37 4.1.2 Coherent States . 38 4.1.3 Multiple orbitals . 40 4.2 BECs and the Gross-Pitaevskii Equation . 41 4.2.1 Noninteracting BECs as Coherent States . -

Chapter 13 Ideal Fermi
Chapter 13 Ideal Fermi gas The properties of an ideal Fermi gas are strongly determined by the Pauli principle. We shall consider the limit: k T µ,βµ 1, B � � which defines the degenerate Fermi gas. In this limit, the quantum mechanical nature of the system becomes especially important, and the system has little to do with the classical ideal gas. Since this chapter is devoted to fermions, we shall omit in the following the subscript ( ) that we used for the fermionic statistical quantities in the previous chapter. − 13.1 Equation of state Consider a gas ofN non-interacting fermions, e.g., electrons, whose one-particle wave- functionsϕ r(�r) are plane-waves. In this case, a complete set of quantum numbersr is given, for instance, by the three cartesian components of the wave vector �k and thez spin projectionm s of an electron: r (k , k , k , m ). ≡ x y z s Spin-independent Hamiltonians. We will consider only spin independent Hamiltonian operator of the type ˆ 3 H= �k ck† ck + d r V(r)c r†cr , �k � where thefirst and the second terms are respectively the kinetic and th potential energy. The summation over the statesr (whenever it has to be performed) can then be reduced to the summation over states with different wavevectork(p=¯hk): ... (2s + 1) ..., ⇒ r � �k where the summation over the spin quantum numberm s = s, s+1, . , s has been taken into account by the prefactor (2s + 1). − − 159 160 CHAPTER 13. IDEAL FERMI GAS Wavefunctions in a box. We as- sume that the electrons are in a vol- ume defined by a cube with sidesL x, Ly,L z and volumeV=L xLyLz. -

Capillary Condensation of Colloid–Polymer Mixtures Confined
INSTITUTE OF PHYSICS PUBLISHING JOURNAL OF PHYSICS: CONDENSED MATTER J. Phys.: Condens. Matter 15 (2003) S3411–S3420 PII: S0953-8984(03)66420-9 Capillary condensation of colloid–polymer mixtures confined between parallel plates Matthias Schmidt1,Andrea Fortini and Marjolein Dijkstra Soft Condensed Matter, Debye Institute, Utrecht University, Princetonplein 5, 3584 CC Utrecht, The Netherlands Received 21 July 2003 Published 20 November 2003 Onlineatstacks.iop.org/JPhysCM/15/S3411 Abstract We investigate the fluid–fluid demixing phase transition of the Asakura–Oosawa model colloid–polymer mixture confined between two smooth parallel hard walls using density functional theory and computer simulations. Comparing fluid density profiles for statepoints away from colloidal gas–liquid coexistence yields good agreement of the theoretical results with simulation data. Theoretical and simulation results predict consistently a shift of the demixing binodal and the critical point towards higher polymer reservoir packing fraction and towards higher colloid fugacities upon decreasing the plate separation distance. This implies capillary condensation of the colloid liquid phase, which should be experimentally observable inside slitlike micropores in contact with abulkcolloidal gas. 1. Introduction Capillary condensation denotes the phenomenon that spatial confinement can stabilize a liquid phase coexisting with its vapour in bulk [1, 2]. In order for this to happen the attractive interaction between the confining walls and the fluid particles needs to be sufficiently strong. Although the main body of work on this subject has been done in the context of confined simple liquids, one might expect that capillary condensation is particularly well suited to be studied with colloidal dispersions. In these complex fluids length scales are on the micron rather than on the ångstrom¨ scale which is typical for atomicsubstances.