Organic Reactions Winter 2013
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Chemistry 2000 Slide Set 1: Introduction to the Molecular Orbital Theory
Chemistry 2000 Slide Set 1: Introduction to the molecular orbital theory Marc R. Roussel January 2, 2020 Marc R. Roussel Introduction to molecular orbitals January 2, 2020 1 / 24 Review: quantum mechanics of atoms Review: quantum mechanics of atoms Hydrogenic atoms The hydrogenic atom (one nucleus, one electron) is exactly solvable. The solutions of this problem are called atomic orbitals. The square of the orbital wavefunction gives a probability density for the electron, i.e. the probability per unit volume of finding the electron near a particular point in space. Marc R. Roussel Introduction to molecular orbitals January 2, 2020 2 / 24 Review: quantum mechanics of atoms Review: quantum mechanics of atoms Hydrogenic atoms (continued) Orbital shapes: 1s 2p 3dx2−y 2 3dz2 Marc R. Roussel Introduction to molecular orbitals January 2, 2020 3 / 24 Review: quantum mechanics of atoms Review: quantum mechanics of atoms Multielectron atoms Consider He, the simplest multielectron atom: Electron-electron repulsion makes it impossible to solve for the electronic wavefunctions exactly. A fourth quantum number, ms , which is associated with a new type of angular momentum called spin, enters into the theory. 1 1 For electrons, ms = 2 or − 2 . Pauli exclusion principle: No two electrons can have identical sets of quantum numbers. Consequence: Only two electrons can occupy an orbital. Marc R. Roussel Introduction to molecular orbitals January 2, 2020 4 / 24 The hydrogen molecular ion The quantum mechanics of molecules + H2 is the simplest possible molecule: two nuclei one electron Three-body problem: no exact solutions However, the nuclei are more than 1800 time heavier than the electron, so the electron moves much faster than the nuclei. -

Theoretical Methods That Help Understanding the Structure and Reactivity of Gas Phase Ions
International Journal of Mass Spectrometry 240 (2005) 37–99 Review Theoretical methods that help understanding the structure and reactivity of gas phase ions J.M. Merceroa, J.M. Matxaina, X. Lopeza, D.M. Yorkb, A. Largoc, L.A. Erikssond,e, J.M. Ugaldea,∗ a Kimika Fakultatea, Euskal Herriko Unibertsitatea, P.K. 1072, 20080 Donostia, Euskadi, Spain b Department of Chemistry, University of Minnesota, 207 Pleasant St. SE, Minneapolis, MN 55455-0431, USA c Departamento de Qu´ımica-F´ısica, Universidad de Valladolid, Prado de la Magdalena, 47005 Valladolid, Spain d Department of Cell and Molecular Biology, Box 596, Uppsala University, 751 24 Uppsala, Sweden e Department of Natural Sciences, Orebro¨ University, 701 82 Orebro,¨ Sweden Received 27 May 2004; accepted 14 September 2004 Available online 25 November 2004 Abstract The methods of the quantum electronic structure theory are reviewed and their implementation for the gas phase chemistry emphasized. Ab initio molecular orbital theory, density functional theory, quantum Monte Carlo theory and the methods to calculate the rate of complex chemical reactions in the gas phase are considered. Relativistic effects, other than the spin–orbit coupling effects, have not been considered. Rather than write down the main equations without further comments on how they were obtained, we provide the reader with essentials of the background on which the theory has been developed and the equations derived. We committed ourselves to place equations in their own proper perspective, so that the reader can appreciate more profoundly the subtleties of the theory underlying the equations themselves. Finally, a number of examples that illustrate the application of the theory are presented and discussed. -

Introduction to Molecular Orbital Theory
Chapter 2: Molecular Structure and Bonding Bonding Theories 1. VSEPR Theory 2. Valence Bond theory (with hybridization) 3. Molecular Orbital Theory ( with molecualr orbitals) To date, we have looked at three different theories of molecular boning. They are the VSEPR Theory (with Lewis Dot Structures), the Valence Bond theory (with hybridization) and Molecular Orbital Theory. A good theory should predict physical and chemical properties of the molecule such as shape, bond energy, bond length, and bond angles.Because arguments based on atomic orbitals focus on the bonds formed between valence electrons on an atom, they are often said to involve a valence-bond theory. The valence-bond model can't adequately explain the fact that some molecules contains two equivalent bonds with a bond order between that of a single bond and a double bond. The best it can do is suggest that these molecules are mixtures, or hybrids, of the two Lewis structures that can be written for these molecules. This problem, and many others, can be overcome by using a more sophisticated model of bonding based on molecular orbitals. Molecular orbital theory is more powerful than valence-bond theory because the orbitals reflect the geometry of the molecule to which they are applied. But this power carries a significant cost in terms of the ease with which the model can be visualized. One model does not describe all the properties of molecular bonds. Each model desribes a set of properties better than the others. The final test for any theory is experimental data. Introduction to Molecular Orbital Theory The Molecular Orbital Theory does a good job of predicting elctronic spectra and paramagnetism, when VSEPR and the V-B Theories don't. -

Recent Progress in the Synthetic Assembly of 2-Cyclopentenones
REVIEW ▌1 Recentreview Progress in the Synthetic Assembly of 2-Cyclopentenones David2-Cyclopentenone J. Synthesis Aitken,*a Hendrik Eijsberg,a,b Angelo Frongia,b Jean Ollivier,a Pier Paolo Pirasb a Laboratoire de Synthèse Organique & Méthodologie, ICMMO (CNRS UMR 8182), Université Paris Sud, 15 rue Georges Clemenceau, 91045 Orsay cedex, France Fax +33(1)69156278; E-mail: [email protected] b Dipartimento di Scienze Chimiche e Geologiche, Università degli studi di Cagliari, Complesso Universitario di Monserrato, S.S. 554, Bivio per Sestu, 09042 Monserrato, Cagliari, Italy Received: 09.07.2013; Accepted after revision: 21.08.2013 considerable number of ways in which the target ring sys- Abstract: An overview of the most important synthetic strategies currently available for the preparation of cyclopent-2-enones is pre- tem can be created from acyclic precursors, in either inter- sented and illustrated with recent applications. molecular or intramolecular mode. Most of the possible disconnection strategies have been examined, and it is im- 1 Introduction portant to recognize that for any given target 2-cyclopen- 2 Multicomponent Ring Assembly tenone, there may be several convenient approaches 3 Cyclizations available. The main approaches for ring construction are 4 Transformations of Existing Cyclic Systems summarized graphically in Figure 1. 5 Miscellaneous Methods O (4+1) O O (3+2) (3+2) coupling 6 Conclusions 1 1 1 5 5 2 5 2 2 RCM Key words: cyclopentenones, cyclization, carbocycles, ring clo- (4+1) (3+2) Rautenstrauch 4 3 4 3 4 3 aldol-type annulation sure, rearrangement, annulation (2+2+1) PKR Nazarov (3+2) Figure 1 The main ring-construction strategies for 2-cyclopente- none synthesis, showing the atom connectivities made during ring as- 1 Introduction sembly (left and center) and cyclization approaches (right). -

3-MO Theory(U).Pptx
Molecular Orbital Theory Valence Bond Theory: Electrons are located in discrete pairs between specific atoms Molecular Orbital Theory: Electrons are located in the molecule, not held in discrete regions between two bonded atoms Thus the main difference between these theories is where the electrons are located, in valence bond theory we predict the electrons are always held between two bonded atoms and in molecular orbital theory the electrons are merely held “somewhere” in molecule Mathematically can represent molecule by a linear combination of atomic orbitals (LCAO) ΨMOL = c1 φ1 + c2 φ2 + c3 φ3 + cn φn Where Ψ2 = spatial distribution of electrons If the ΨMOL can be determined, then where the electrons are located can also be determined 66 Building Molecular Orbitals from Atomic Orbitals Similar to a wave function that can describe the regions of space where electrons reside on time average for an atom, when two (or more) atoms react to form new bonds, the region where the electrons reside in the new molecule are described by a new wave function This new wave function describes molecular orbitals instead of atomic orbitals Mathematically, these new molecular orbitals are simply a combination of the atomic wave functions (e.g LCAO) Hydrogen 1s H-H bonding atomic orbital molecular orbital 67 Building Molecular Orbitals from Atomic Orbitals An important consideration, however, is that the number of wave functions (molecular orbitals) resulting from the mixing process must equal the number of wave functions (atomic orbitals) used in the mixing -

Simple Molecular Orbital Theory Chapter 5
Simple Molecular Orbital Theory Chapter 5 Wednesday, October 7, 2015 Using Symmetry: Molecular Orbitals One approach to understanding the electronic structure of molecules is called Molecular Orbital Theory. • MO theory assumes that the valence electrons of the atoms within a molecule become the valence electrons of the entire molecule. • Molecular orbitals are constructed by taking linear combinations of the valence orbitals of atoms within the molecule. For example, consider H2: 1s + 1s + • Symmetry will allow us to treat more complex molecules by helping us to determine which AOs combine to make MOs LCAO MO Theory MO Math for Diatomic Molecules 1 2 A ------ B Each MO may be written as an LCAO: cc11 2 2 Since the probability density is given by the square of the wavefunction: probability of finding the ditto atom B overlap term, important electron close to atom A between the atoms MO Math for Diatomic Molecules 1 1 S The individual AOs are normalized: 100% probability of finding electron somewhere for each free atom MO Math for Homonuclear Diatomic Molecules For two identical AOs on identical atoms, the electrons are equally shared, so: 22 cc11 2 2 cc12 In other words: cc12 So we have two MOs from the two AOs: c ,1() 1 2 c ,1() 1 2 2 2 After normalization (setting d 1 and _ d 1 ): 1 1 () () [2(1 S )]1/2 12 [2(1 S )]1/2 12 where S is the overlap integral: 01 S LCAO MO Energy Diagram for H2 H2 molecule: two 1s atomic orbitals combine to make one bonding and one antibonding molecular orbital. -
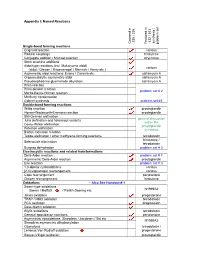
Appendix I: Named Reactions Single-Bond Forming Reactions Co
Appendix I: Named Reactions 235 / 335 432 / 533 synthesis / / synthesis Covered in Covered Featured in problem set problem Single-bond forming reactions Grignard reaction various Radical couplings hirstutene Conjugate addition / Michael reaction strychnine Stork enamine additions Aldol-type reactions (incl. Mukaiyama aldol) various (aldol / Claisen / Knoevenagel / Mannich / Henry etc.) Asymmetric aldol reactions: Evans / Carreira etc. saframycin A Organocatalytic asymmetric aldol saframycin A Pseudoephedrine glycinamide alkylation saframycin A Prins reaction Prins-pinacol reaction problem set # 2 Morita-Baylis-Hillman reaction McMurry condensation Gabriel synthesis problem set #3 Double-bond forming reactions Wittig reaction prostaglandin Horner-Wadsworth-Emmons reaction prostaglandin Still-Gennari olefination general discussion Julia olefination and heteroaryl variants within the Corey-Winter olefination prostaglandin Peterson olefination synthesis Barton extrusion reaction Tebbe olefination / other methylene-forming reactions tetrodotoxin hirstutene / Selenoxide elimination tetrodotoxin Burgess dehydration problem set # 3 Electrocyclic reactions and related transformations Diels-Alder reaction problem set # 1 Asymmetric Diels-Alder reaction prostaglandin Ene reaction problem set # 3 1,3-dipolar cycloadditions various [2,3] sigmatropic rearrangement various Cope rearrangement periplanone Claisen rearrangement hirstutene Oxidations – Also See Handout # 1 Swern-type oxidations (Swern / Moffatt / Parikh-Doering etc. N1999A2 Jones oxidation -

Elimination Reactions Are Described
Introduction In this module, different types of elimination reactions are described. From a practical standpoint, elimination reactions widely used for the generation of double and triple bonds in compounds from a saturated precursor molecule. The presence of a good leaving group is a prerequisite in most elimination reactions. Traditional classification of elimination reactions, in terms of the molecularity of the reaction is employed. How the changes in the nature of the substrate as well as reaction conditions affect the mechanism of elimination are subsequently discussed. The stereochemical requirements for elimination in a given substrate and its consequence in the product stereochemistry is emphasized. ELIMINATION REACTIONS Objective and Outline beta-eliminations E1, E2 and E1cB mechanisms Stereochemical considerations of these reactions Examples of E1, E2 and E1cB reactions Alpha eliminations and generation of carbene I. Basics Elimination reactions involve the loss of fragments or groups from a molecule to generate multiple bonds. A generalized equation is shown below for 1,2-elimination wherein the X and Y from two adjacent carbon atoms are removed, elimination C C C C -XY X Y Three major types of elimination reactions are: α-elimination: two atoms or groups are removed from the same atom. It is also known as 1,1-elimination. H R R C X C + HX R Both H and X are removed from carbon atom here R Carbene β-elimination: loss of atoms or groups on adjacent atoms. It is also H H known as 1,2- elimination. R C C R R HC CH R X H γ-elimination: loss of atoms or groups from the 1st and 3rd positions as shown below. -

UNIVERSITY of CALIFORNIA Santa Barbara Regiochemistry of Nitroso
UNIVERSITY OF CALIFORNIA Santa Barbara Regiochemistry of Nitroso Hetero Diels-Alder Reactions, Catechol Siderophore Analogues for Surface Wet Adhesion, and an Inquiry-Based Synthetic Organic Laboratory Course A dissertation submitted in partial satisfaction of the requirements for the degree Doctor of Philosophy in Chemistry by Robert Bradley Lewis Committee in charge: Professor Alison Butler, Co-Chair Professor Javier Read de Alaniz, Co-Chair Professor R. Daniel Little Dr. Morgan Gainer, Lecturer January 2018 The dissertation of Robert Bradley Lewis is approved. ____________________________________________ R. Daniel Little ____________________________________________ Morgan Gainer ____________________________________________ Javier Read de Alaniz, Committee Co-Chair ____________________________________________ Alison Butler, Committee Co-Chair January 2018 Regiochemistry of Nitroso Hetero Diels-Alder Reactions, Catechol Siderophore Analogues for Surface Wet Adhesion, and an Inquiry-Based Synthetic Organic Laboratory Course Copyright © 2018 by Robert Bradley Lewis iii Acknowledgements When I decided to pursue my PhD in chemistry, I had no idea how much of a monumental undertaking it would be. It has been a long and difficult journey, and this dissertation only gives a brief glimpse of what went into the process. Nothing contained in this dissertation would possible without the continued support of all my family, friends, and co-workers. First and foremost, I want to thank my advisors and mentors. Javier Read de Alaniz, you gave me my first home at UCSB. You were always patient and caring, and you fostered a wonderful group environment, both in and out of the lab. Alison Butler, you gave me my second home at UCSB. Your unbridled enthusiasm for science is infectious and I appreciate that you allowed me to explore my interest in education by taking a few quarters to teach a lecture course. -

2 Results and Discussion
22 2 Results and Discussion 2 Results and Discussion 2.1 Syntheses of Azoninones - Unsaturated Nine-Membered Ring Lactams 2.1.1 Syntheses of Vinyl pyrrolidines N-Vinyl pyrrolidines are important reactants for the preparation of azoninones by zwitterionic aza- Claisen rearrangement. They can be obtained by derivatisation reactions starting from chiral molecules such as L-proline and 2S,4R-4-hydroxyproline or by metal catalysed cyclisation reactions of N- substituted allenic amines.66 Since large amounts of vinyl pyrrolidines have to be synthetically available for the systematic examination of the aza-Claisen rearrangement and its application in a total synthesis, especially the ex-chiral pool synthesis of these precursors has been extensively investigated by our group.67 The following two schemes represent the synthesis of the vinyl pyrrolidines [6] and [11] that have been used in this work as precursors for the aza-Claisen rearrangement. This synthesis allows the preparation of 50-100g of vinyl pyrrolidines. HO HO HO BnCl, Et3N TBSCl, imid. AcCl, MeOH CH Cl CH2Cl2 2 2 N COOH CO Me 100% N CO2Me 88% N 2 95% H H Bn [1] [2] [3] TBSO TBSO 1) Oxalyl chloride TBSO DMSO, Et3N, CH2Cl2 DIBALH, THF 2) Ph3P=CH2, THF N CO2Me N 89% N 70% Bn Bn OH Bn [4] [5] [6] Scheme 19 Synthesis of vinyl pyrrolidine [6] Allylamine [6] was efficiently generated via a six-step sequence starting from trans-4-hydroxy-L-(-)- proline [1] the overall yield was about 50% (Scheme 19). After esterification,68 the N-benzyl group 66 (a) Huby, N. -

Application of Complex Aldol Reactions to the Total Synthesis of Phorboxazole B
J. Am. Chem. Soc. 2000, 122, 10033-10046 10033 Application of Complex Aldol Reactions to the Total Synthesis of Phorboxazole B David A. Evans,* Duke M. Fitch,1 Thomas E. Smith, and Victor J. Cee Contribution from the Department of Chemistry and Chemical Biology, HarVard UniVersity, Cambridge, Massachusetts 02138 ReceiVed June 29, 2000 Abstract: The synthesis of phorboxazole B has been accomplished in 27 linear steps and an overall yield of 12.6%. The absolute stereochemistry of the C4-C12,C33-C38, and C13-C19 fragments was established utilizing catalytic asymmetric aldol methodology, while the absolute stereochemistry of the C20-C32 fragment was derived from an auxiliary-based asymmetric aldol reaction. All remaining chirality was incorporated through internal asymmetric induction, with the exception of the C43 stereocenter which was derived from (R)-trityl glycidol. Key fragment couplings include a stereoselective double stereodifferentiating aldol reaction, a metalated oxazole alkylation, an oxazole-stabilized Wittig olefination, and a chelation-controlled addition of the fully elaborated alkenyl metal side chain. Introduction and HT29 (3.31 × 10-10 M), as well as leukemia CCRF-CBM (2.45 × 10-10 M), prostate cancer PC-3 (3.54 × 10-10 M), and Phorboxazoles A (1)andB(2) are marine natural products breast cancer MCF7 cell lines (5.62 × 10-10 M).2b In addition isolated from a recently discovered species of Indian Ocean to this impressive anticancer activity, phorboxazoles A and B sponge (genus Phorbas sp.) near Muiron Island, Western also exhibit potent in vitro antifungal activity against Candida Australia.2 These substances are representative of a new class albicans and Saccharomyces carlsbergensis at 0.1 µg/disk in of macrolides differing only in their C hydroxyl-bearing 13 the agar disk diffusion assay.2a stereocenters. -

Olefination Reactions Lecture Notes OTBS OPMB O
Olefination Reactions Lecture Notes OTBS OPMB O Key Reviews: O Me O Me XX Wittig Reaction K. C. Nicolaou and co-workers, Ann. 1997, 1283. Horner-Wadsworth-Emmons and Tebbe Olefinations S. E. Kelly, Comprehensive Org. Synth. 1991, Vol. 1, 729. Peterson Olefination D. J. Ager, Synthesis 1984, 384. Julia (Julia-Lythgoe) Olefination B. M. Trost, Bull. Chem. Soc. Jpn. 1988, 61, 107. Wittig Olefination: Background and Principles R1 n-BuLi, Ph P 3 O X LDA, R R1 LiHMDS PPh + 1 3 X Ph3P + R1 Ph3P pKa = 18-20 ylide when R = alkyl, H Ph Ph Ph Ph Ph R1 P Ph P -[Ph3P=O] O R1 O R strong bond 1 formation drives reaction oxaphosphatane betaine G. Wittig and G. Schollkopf, Chem. Ber. 1954, 87, 1318. Wittig Olefination: Background and Principles Stereoselectivity with non-stabilized ylides Me OMe Me Ph3P Ph3P Ph3P Me Ph3P Ph3P Not stable; must be made in situ and used immediately Wittig Olefination: Background and Principles Stereoselectivity with non-stabilized ylides Me OMe Me Ph3P Ph3P Ph3P Me Ph3P Ph3P Not stable; must be made in situ and used immediately Addition to carbonyl is an irreversible and concerted [2+2] cycloaddition such that the R groups on the aldehyde and the ylide are as far apart as possible PPh3 Ph3P O H H H O + Me Me Ph3P R O R H -[Ph3P=O] As the size of the R groups increases, selectivity for Z-alkene increases Me Nonpolar solvents favor initial addition Polar solvents favor the elimination Z-alkene Wittig Olefination: Background and Principles Stereoselectivity with stabilized ylides Me O O Ph P Ph3P 3 Ph3P Me OEt semistabilized Incredibly stable; not moisture sensitive, can be chromatographed Price for stability is lower reactivity: reacts well with aldehydes, slowly with ketones Wittig Olefination: Background and Principles Stereoselectivity with stabilized ylides Me O O Ph P Ph3P 3 Ph3P Me OEt semistabilized Incredibly stable; not moisture sensitive, can be chromatographed Price for stability is lower reactivity: reacts well with aldehydes, slowly with ketones Initial addition to carbonyl is reversible so the thermodynamic elimination product results.