Introduction to Atomic Nuclei
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Neutron Stars
Chandra X-Ray Observatory X-Ray Astronomy Field Guide Neutron Stars Ordinary matter, or the stuff we and everything around us is made of, consists largely of empty space. Even a rock is mostly empty space. This is because matter is made of atoms. An atom is a cloud of electrons orbiting around a nucleus composed of protons and neutrons. The nucleus contains more than 99.9 percent of the mass of an atom, yet it has a diameter of only 1/100,000 that of the electron cloud. The electrons themselves take up little space, but the pattern of their orbit defines the size of the atom, which is therefore 99.9999999999999% Chandra Image of Vela Pulsar open space! (NASA/PSU/G.Pavlov et al. What we perceive as painfully solid when we bump against a rock is really a hurly-burly of electrons moving through empty space so fast that we can't see—or feel—the emptiness. What would matter look like if it weren't empty, if we could crush the electron cloud down to the size of the nucleus? Suppose we could generate a force strong enough to crush all the emptiness out of a rock roughly the size of a football stadium. The rock would be squeezed down to the size of a grain of sand and would still weigh 4 million tons! Such extreme forces occur in nature when the central part of a massive star collapses to form a neutron star. The atoms are crushed completely, and the electrons are jammed inside the protons to form a star composed almost entirely of neutrons. -
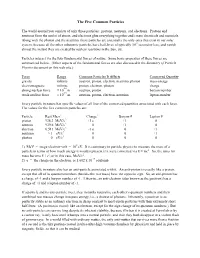
The Five Common Particles
The Five Common Particles The world around you consists of only three particles: protons, neutrons, and electrons. Protons and neutrons form the nuclei of atoms, and electrons glue everything together and create chemicals and materials. Along with the photon and the neutrino, these particles are essentially the only ones that exist in our solar system, because all the other subatomic particles have half-lives of typically 10-9 second or less, and vanish almost the instant they are created by nuclear reactions in the Sun, etc. Particles interact via the four fundamental forces of nature. Some basic properties of these forces are summarized below. (Other aspects of the fundamental forces are also discussed in the Summary of Particle Physics document on this web site.) Force Range Common Particles It Affects Conserved Quantity gravity infinite neutron, proton, electron, neutrino, photon mass-energy electromagnetic infinite proton, electron, photon charge -14 strong nuclear force ≈ 10 m neutron, proton baryon number -15 weak nuclear force ≈ 10 m neutron, proton, electron, neutrino lepton number Every particle in nature has specific values of all four of the conserved quantities associated with each force. The values for the five common particles are: Particle Rest Mass1 Charge2 Baryon # Lepton # proton 938.3 MeV/c2 +1 e +1 0 neutron 939.6 MeV/c2 0 +1 0 electron 0.511 MeV/c2 -1 e 0 +1 neutrino ≈ 1 eV/c2 0 0 +1 photon 0 eV/c2 0 0 0 1) MeV = mega-electron-volt = 106 eV. It is customary in particle physics to measure the mass of a particle in terms of how much energy it would represent if it were converted via E = mc2. -

Computing ATOMIC NUCLEI
UNIVERSAL NUCLEAR ENERGY DENSITY FUNCTIONAL Computing ATOMIC NUCLEI Petascale computing helps disentangle the nuclear puzzle. The goal of the Universal Nuclear Energy Density Functional (UNEDF) collaboration is to provide a comprehensive description of all nuclei and their reactions based on the most accurate knowledge of the nuclear interaction, the most reliable theoretical approaches, and the massive use of computer power. Science of Nuclei the Hamiltonian matrix. Coupled cluster (CC) Nuclei comprise 99.9% of all baryonic matter in techniques, which were formulated by nuclear sci- the Universe and are the fuel that burns in stars. entists in the 1950s, are essential techniques in The rather complex nature of the nuclear forces chemistry today and have recently been resurgent among protons and neutrons generates a broad in nuclear structure. Quantum Monte Carlo tech- range and diversity in the nuclear phenomena that niques dominate studies of phase transitions in can be observed. As shown during the last decade, spin systems and nuclei. These methods are used developing a comprehensive description of all to understand both the nuclear and electronic nuclei and their reactions requires theoretical and equations of state in condensed systems, and they experimental investigations of rare isotopes with are used to investigate the excitation spectra in unusual neutron-to-proton ratios. These nuclei nuclei, atoms, and molecules. are labeled exotic, or rare, because they are not When applied to systems with many active par- typically found on Earth. They are difficult to pro- ticles, ab initio and configuration interaction duce experimentally because they usually have methods present computational challenges as the extremely short lifetimes. -

B2.IV Nuclear and Particle Physics
B2.IV Nuclear and Particle Physics A.J. Barr February 13, 2014 ii Contents 1 Introduction 1 2 Nuclear 3 2.1 Structure of matter and energy scales . 3 2.2 Binding Energy . 4 2.2.1 Semi-empirical mass formula . 4 2.3 Decays and reactions . 8 2.3.1 Alpha Decays . 10 2.3.2 Beta decays . 13 2.4 Nuclear Scattering . 18 2.4.1 Cross sections . 18 2.4.2 Resonances and the Breit-Wigner formula . 19 2.4.3 Nuclear scattering and form factors . 22 2.5 Key points . 24 Appendices 25 2.A Natural units . 25 2.B Tools . 26 2.B.1 Decays and the Fermi Golden Rule . 26 2.B.2 Density of states . 26 2.B.3 Fermi G.R. example . 27 2.B.4 Lifetimes and decays . 27 2.B.5 The flux factor . 28 2.B.6 Luminosity . 28 2.C Shell Model § ............................. 29 2.D Gamma decays § ............................ 29 3 Hadrons 33 3.1 Introduction . 33 3.1.1 Pions . 33 3.1.2 Baryon number conservation . 34 3.1.3 Delta baryons . 35 3.2 Linear Accelerators . 36 iii CONTENTS CONTENTS 3.3 Symmetries . 36 3.3.1 Baryons . 37 3.3.2 Mesons . 37 3.3.3 Quark flow diagrams . 38 3.3.4 Strangeness . 39 3.3.5 Pseudoscalar octet . 40 3.3.6 Baryon octet . 40 3.4 Colour . 41 3.5 Heavier quarks . 43 3.6 Charmonium . 45 3.7 Hadron decays . 47 Appendices 48 3.A Isospin § ................................ 49 3.B Discovery of the Omega § ...................... -

TEK 8.5C: Periodic Table
Name: Teacher: Pd. Date: TEK 8.5C: Periodic Table TEK 8.5C: Interpret the arrangement of the Periodic Table, including groups and periods, to explain how properties are used to classify elements. Elements and the Periodic Table An element is a substance that cannot be separated into simpler substances by physical or chemical means. An element is already in its simplest form. The smallest piece of an element that still has the properties of that element is called an atom. An element is a pure substance, containing only one kind of atom. The Periodic Table of Elements is a list of all the elements that have been discovered and named, with each element listed in its own element square. Elements are represented on the Periodic Table by a one or two letter symbol, and its name, atomic number and atomic mass. The Periodic Table & Atomic Structure The elements are listed on the Periodic Table in atomic number order, starting at the upper left corner and then moving from the left to right and top to bottom, just as the words of a paragraph are read. The element’s atomic number is based on the number of protons in each atom of that element. In electrically neutral atoms, the atomic number also represents the number of electrons in each atom of that element. For example, the atomic number for neon (Ne) is 10, which means that each atom of neon has 10 protons and 10 electrons. Magnesium (Mg) has an atomic number of 12, which means it has 12 protons and 12 electrons. -

The R-Process Nucleosynthesis and Related Challenges
EPJ Web of Conferences 165, 01025 (2017) DOI: 10.1051/epjconf/201716501025 NPA8 2017 The r-process nucleosynthesis and related challenges Stephane Goriely1,, Andreas Bauswein2, Hans-Thomas Janka3, Oliver Just4, and Else Pllumbi3 1Institut d’Astronomie et d’Astrophysique, Université Libre de Bruxelles, CP 226, 1050 Brussels, Belgium 2Heidelberger Institut fr¨ Theoretische Studien, Schloss-Wolfsbrunnenweg 35, 69118 Heidelberg, Germany 3Max-Planck-Institut für Astrophysik, Postfach 1317, 85741 Garching, Germany 4Astrophysical Big Bang Laboratory, RIKEN, 2-1 Hirosawa, Wako, Saitama, 351-0198, Japan Abstract. The rapid neutron-capture process, or r-process, is known to be of fundamental importance for explaining the origin of approximately half of the A > 60 stable nuclei observed in nature. Recently, special attention has been paid to neutron star (NS) mergers following the confirmation by hydrodynamic simulations that a non-negligible amount of matter can be ejected and by nucleosynthesis calculations combined with the predicted astrophysical event rate that such a site can account for the majority of r-material in our Galaxy. We show here that the combined contribution of both the dynamical (prompt) ejecta expelled during binary NS or NS-black hole (BH) mergers and the neutrino and viscously driven outflows generated during the post-merger remnant evolution of relic BH-torus systems can lead to the production of r-process elements from mass number A > 90 up to actinides. The corresponding abundance distribution is found to reproduce the∼ solar distribution extremely well. It can also account for the elemental distributions observed in low-metallicity stars. However, major uncertainties still affect our under- standing of the composition of the ejected matter. -

Centripetal Force Is Balanced by the Circular Motion of the Elctron Causing the Centrifugal Force
STANDARD SC1 b. Construct an argument to support the claim that the proton (and not the neutron or electron) defines the element’s identity. c. Construct an explanation based on scientific evidence of the production of elements heavier than hydrogen by nuclear fusion. d. Construct an explanation that relates the relative abundance of isotopes of a particular element to the atomic mass of the element. First, we quickly review pre-requisite concepts One of the most curious observations with atoms is the fact that there are charged particles inside the atom and there is also constant spinning and Warm-up 1: List the name, charge, mass, and location of the three subatomic circling. How does atom remain stable under these conditions? Remember particles Opposite charges attract each other; Like charges repel each other. Your Particle Location Charge Mass in a.m.u. Task: Read the following information and consult with your teacher as STABILITY OF ATOMS needed, answer Warm-Up tasks 2 and 3 on Page 2. (3) Death spiral does not occur at all! This is because the centripetal force is balanced by the circular motion of the elctron causing the centrifugal force. The centrifugal force is the outward force from the center to the circumference of the circle. Electrons not only spin on their own axis, they are also in a constant circular motion around the nucleus. Despite this terrific movement, electrons are very stable. The stability of electrons mainly comes from the electrostatic forces of attraction between the nucleus and the electrons. The electrostatic forces are also known as Coulombic Forces of Attraction. -

Photofission Cross Sections of 238U and 235U from 5.0 Mev to 8.0 Mev Robert Andrew Anderl Iowa State University
Iowa State University Capstones, Theses and Retrospective Theses and Dissertations Dissertations 1972 Photofission cross sections of 238U and 235U from 5.0 MeV to 8.0 MeV Robert Andrew Anderl Iowa State University Follow this and additional works at: https://lib.dr.iastate.edu/rtd Part of the Nuclear Commons, and the Oil, Gas, and Energy Commons Recommended Citation Anderl, Robert Andrew, "Photofission cross sections of 238U and 235U from 5.0 MeV to 8.0 MeV " (1972). Retrospective Theses and Dissertations. 4715. https://lib.dr.iastate.edu/rtd/4715 This Dissertation is brought to you for free and open access by the Iowa State University Capstones, Theses and Dissertations at Iowa State University Digital Repository. It has been accepted for inclusion in Retrospective Theses and Dissertations by an authorized administrator of Iowa State University Digital Repository. For more information, please contact [email protected]. INFORMATION TO USERS This dissertation was produced from a microfilm copy of the original document. While the most advanced technological means to photograph and reproduce this document have been used, the quality is heavily dependent upon the quality of the original submitted. The following explanation of techniques is provided to help you understand markings or patterns which may appear on this reproduction, 1. The sign or "target" for pages apparently lacking from the document photographed is "Missing Page(s)". If it was possible to obtain the missing page(s) or section, they are spliced into the film along with adjacent pages. This may have necessitated cutting thru an image and duplicating adjacent pages to insure you complete continuity, 2. -

1 Lecture Notes in Nuclear Structure Physics B. Alex Brown November
1 Lecture Notes in Nuclear Structure Physics B. Alex Brown November 2005 National Superconducting Cyclotron Laboratory and Department of Physics and Astronomy Michigan State University, E. Lansing, MI 48824 CONTENTS 2 Contents 1 Nuclear masses 6 1.1 Masses and binding energies . 6 1.2 Q valuesandseparationenergies . 10 1.3 Theliquid-dropmodel .......................... 18 2 Rms charge radii 25 3 Charge densities and form factors 31 4 Overview of nuclear decays 40 4.1 Decaywidthsandlifetimes. 41 4.2 Alphaandclusterdecay ......................... 42 4.3 Betadecay................................. 51 4.3.1 BetadecayQvalues ....................... 52 4.3.2 Allowedbetadecay ........................ 53 4.3.3 Phase-space for allowed beta decay . 57 4.3.4 Weak-interaction coupling constants . 59 4.3.5 Doublebetadecay ........................ 59 4.4 Gammadecay............................... 61 4.4.1 Reduced transition probabilities for gamma decay . .... 62 4.4.2 Weisskopf units for gamma decay . 65 5 The Fermi gas model 68 6 Overview of the nuclear shell model 71 7 The one-body potential 77 7.1 Generalproperties ............................ 77 7.2 Theharmonic-oscillatorpotential . ... 79 7.3 Separation of intrinsic and center-of-mass motion . ....... 81 7.3.1 Thekineticenergy ........................ 81 7.3.2 Theharmonic-oscillator . 83 8 The Woods-Saxon potential 87 8.1 Generalform ............................... 87 8.2 Computer program for the Woods-Saxon potential . .... 91 8.2.1 Exampleforboundstates . 92 8.2.2 Changingthepotentialparameters . 93 8.2.3 Widthofanunboundstateresonance . 94 8.2.4 Width of an unbound state resonance at a fixed energy . 95 9 The general many-body problem for fermions 97 CONTENTS 3 10 Conserved quantum numbers 101 10.1 Angularmomentum. 101 10.2Parity .................................. -

Of the Periodic Table
of the Periodic Table teacher notes Give your students a visual introduction to the families of the periodic table! This product includes eight mini- posters, one for each of the element families on the main group of the periodic table: Alkali Metals, Alkaline Earth Metals, Boron/Aluminum Group (Icosagens), Carbon Group (Crystallogens), Nitrogen Group (Pnictogens), Oxygen Group (Chalcogens), Halogens, and Noble Gases. The mini-posters give overview information about the family as well as a visual of where on the periodic table the family is located and a diagram of an atom of that family highlighting the number of valence electrons. Also included is the student packet, which is broken into the eight families and asks for specific information that students will find on the mini-posters. The students are also directed to color each family with a specific color on the blank graphic organizer at the end of their packet and they go to the fantastic interactive table at www.periodictable.com to learn even more about the elements in each family. Furthermore, there is a section for students to conduct their own research on the element of hydrogen, which does not belong to a family. When I use this activity, I print two of each mini-poster in color (pages 8 through 15 of this file), laminate them, and lay them on a big table. I have students work in partners to read about each family, one at a time, and complete that section of the student packet (pages 16 through 21 of this file). When they finish, they bring the mini-poster back to the table for another group to use. -

1 PROF. CHHANDA SAMANTA, Phd PAPERS in PEER REVIEWED INTERNATIONAL JOURNALS: 1. C. Samanta, T. A. Schmitt, “Binding, Bonding A
PROF. CHHANDA SAMANTA, PhD PAPERS IN PEER REVIEWED INTERNATIONAL JOURNALS: 1. C. Samanta, T. A. Schmitt, “Binding, bonding and charge symmetry breaking in Λ- hypernuclei”, arXiv:1710.08036v2 [nucl-th] (to be published) 2. T. A. Schmitt, C. Samanta, “A-dependence of -bond and charge symmetry energies”, EPJ Web Conf. 182, 03012 (2018) 3. Chhanda Samanta, Superheavy Nuclei to Hypernuclei: A Tribute to Walter Greiner, EPJ Web Conf. 182, 02107 (2018) 4. C. Samanta with X Qiu, L Tang, C Chen, et al., “Direct measurements of the lifetime of medium-heavy hypernuclei”, Nucl. Phys. A973, 116 (2018); arXiv:1212.1133 [nucl-ex] 5. C. Samanta with with S. Mukhopadhyay, D. Atta, K. Imam, D. N. Basu, “Static and rotating hadronic stars mixed with self-interacting fermionic Asymmetric Dark Matter”, The European Physical Journal C.77:440 (2017); arXiv:1612.07093v1 6. C. Samanta with R. Honda, M. Agnello, J. K. Ahn et al, “Missing-mass spectroscopy with 6 − + 6 the Li(π ,K )X reaction to search for ΛH”, Phys. Rev. C 96, 014005 (2017); arXiv:1703.00623v2 [nucl-ex] 7. C. Samanta with T. Gogami, C. Chen, D. Kawama et al., “Spectroscopy of the neutron-rich 7 hypernucleus He from electron scattering”, Phys. Rev. C94, 021302(R) (2016); arXiv:1606.09157 8. C. Samanta with T. Gogami, C. Chen, D. Kawama,et al., ”High Resolution Spectroscopic 10 Study of ΛBe”, Phys. Rev. C93, 034314(2016); arXiv:1511.04801v1[nucl-ex] 9. C. Samanta with L. Tang, C. Chen, T. Gogami et al., “The experiments with the High 12 Resolution Kaon Spectrometer at JLab Hall C and the new spectroscopy of ΛB hypernuclei, Phys. -

Current Status of Equation of State in Nuclear Matter and Neutron Stars
Open Issues in Understanding Core Collapse Supernovae June 22-24, 2004 Current Status of Equation of State in Nuclear Matter and Neutron Stars J.R.Stone1,2, J.C. Miller1,3 and W.G.Newton1 1 Oxford University, Oxford, UK 2Physics Division, ORNL, Oak Ridge, TN 3 SISSA, Trieste, Italy Outline 1. General properties of EOS in nuclear matter 2. Classification according to models of N-N interaction 3. Examples of EOS – sensitivity to the choice of N-N interaction 4. Consequences for supernova simulations 5. Constraints on EOS 6. High density nuclear matter (HDNM) 7. New developments Equation of State is derived from a known dependence of energy per particle of a system on particle number density: EA/(==En) or F/AF(n) I. E ( or Boltzman free energy F = E-TS for system at finite temperature) is constructed in a form of effective energy functional (Hamiltonian, Lagrangian, DFT/EFT functional or an empirical form) II. An equilibrium state of matter is found at each density n by minimization of E (n) or F (n) III. All other related quantities like e.g. pressure P, incompressibility K or entropy s are calculated as derivatives of E or F at equilibrium: 2 ∂E ()n ∂F ()n Pn()= n sn()=− | ∂n ∂T nY, p ∂∂P()nnEE() ∂2 ()n Kn()==9 18n +9n2 ∂∂nn∂n2 IV. Use as input for model simulations (Very) schematic sequence of equilibrium phases of nuclear matter as a function of density: <~2x10-4fm-3 ~2x10-4 fm-3 ~0.06 fm-3 Nuclei in Nuclei in Neutron electron gas + ‘Pasta phase’ Electron gas ~0.1 fm-3 0.3-0.5 fm-3 >0.5 fm-3 Nucleons + n,p,e,µ heavy baryons Quarks ???