Using the Entropy Rate Balance to Determine the Heat Transfer and Work in an Internally Reversible, Polytropic, Steady State Flow Process
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
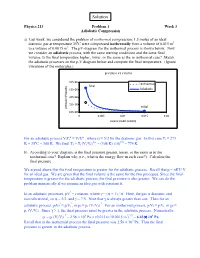
3-1 Adiabatic Compression
Solution Physics 213 Problem 1 Week 3 Adiabatic Compression a) Last week, we considered the problem of isothermal compression: 1.5 moles of an ideal diatomic gas at temperature 35oC were compressed isothermally from a volume of 0.015 m3 to a volume of 0.0015 m3. The pV-diagram for the isothermal process is shown below. Now we consider an adiabatic process, with the same starting conditions and the same final volume. Is the final temperature higher, lower, or the same as the in isothermal case? Sketch the adiabatic processes on the p-V diagram below and compute the final temperature. (Ignore vibrations of the molecules.) α α For an adiabatic process ViTi = VfTf , where α = 5/2 for the diatomic gas. In this case Ti = 273 o 1/α 2/5 K + 35 C = 308 K. We find Tf = Ti (Vi/Vf) = (308 K) (10) = 774 K. b) According to your diagram, is the final pressure greater, lesser, or the same as in the isothermal case? Explain why (i.e., what is the energy flow in each case?). Calculate the final pressure. We argued above that the final temperature is greater for the adiabatic process. Recall that p = nRT/ V for an ideal gas. We are given that the final volume is the same for the two processes. Since the final temperature is greater for the adiabatic process, the final pressure is also greater. We can do the problem numerically if we assume an idea gas with constant α. γ In an adiabatic processes, pV = constant, where γ = (α + 1) / α. -

Toward Quantifying the Climate Heat Engine: Solar Absorption and Terrestrial Emission Temperatures and Material Entropy Production
JUNE 2017 B A N N O N A N D L E E 1721 Toward Quantifying the Climate Heat Engine: Solar Absorption and Terrestrial Emission Temperatures and Material Entropy Production PETER R. BANNON AND SUKYOUNG LEE Department of Meteorology and Atmospheric Science, The Pennsylvania State University, University Park, Pennsylvania (Manuscript received 15 August 2016, in final form 22 February 2017) ABSTRACT A heat-engine analysis of a climate system requires the determination of the solar absorption temperature and the terrestrial emission temperature. These temperatures are entropically defined as the ratio of the energy exchanged to the entropy produced. The emission temperature, shown here to be greater than or equal to the effective emission temperature, is relatively well known. In contrast, the absorption temperature re- quires radiative transfer calculations for its determination and is poorly known. The maximum material (i.e., nonradiative) entropy production of a planet’s steady-state climate system is a function of the absorption and emission temperatures. Because a climate system does no work, the material entropy production measures the system’s activity. The sensitivity of this production to changes in the emission and absorption temperatures is quantified. If Earth’s albedo does not change, material entropy production would increase by about 5% per 1-K increase in absorption temperature. If the absorption temperature does not change, entropy production would decrease by about 4% for a 1% decrease in albedo. It is shown that, as a planet’s emission temperature becomes more uniform, its entropy production tends to increase. Conversely, as a planet’s absorption temperature or albedo becomes more uniform, its entropy production tends to decrease. -

Novel Hot Air Engine and Its Mathematical Model – Experimental Measurements and Numerical Analysis
POLLACK PERIODICA An International Journal for Engineering and Information Sciences DOI: 10.1556/606.2019.14.1.5 Vol. 14, No. 1, pp. 47–58 (2019) www.akademiai.com NOVEL HOT AIR ENGINE AND ITS MATHEMATICAL MODEL – EXPERIMENTAL MEASUREMENTS AND NUMERICAL ANALYSIS 1 Gyula KRAMER, 2 Gabor SZEPESI *, 3 Zoltán SIMÉNFALVI 1,2,3 Department of Chemical Machinery, Institute of Energy and Chemical Machinery University of Miskolc, Miskolc-Egyetemváros 3515, Hungary e-mail: [email protected], [email protected], [email protected] Received 11 December 2017; accepted 25 June 2018 Abstract: In the relevant literature there are many types of heat engines. One of those is the group of the so called hot air engines. This paper introduces their world, also introduces the new kind of machine that was developed and built at Department of Chemical Machinery, Institute of Energy and Chemical Machinery, University of Miskolc. Emphasizing the novelty of construction and the working principle are explained. Also the mathematical model of this new engine was prepared and compared to the real model of engine. Keywords: Hot, Air, Engine, Mathematical model 1. Introduction There are three types of volumetric heat engines: the internal combustion engines; steam engines; and hot air engines. The first one is well known, because it is on zenith nowadays. The steam machines are also well known, because their time has just passed, even the elder ones could see those in use. But the hot air engines are forgotten. Our aim is to consider that one. The history of hot air engines is 200 years old. -

Basic Thermodynamics-17ME33.Pdf
Module -1 Fundamental Concepts & Definitions & Work and Heat MODULE 1 Fundamental Concepts & Definitions Thermodynamics definition and scope, Microscopic and Macroscopic approaches. Some practical applications of engineering thermodynamic Systems, Characteristics of system boundary and control surface, examples. Thermodynamic properties; definition and units, intensive and extensive properties. Thermodynamic state, state point, state diagram, path and process, quasi-static process, cyclic and non-cyclic processes. Thermodynamic equilibrium; definition, mechanical equilibrium; diathermic wall, thermal equilibrium, chemical equilibrium, Zeroth law of thermodynamics, Temperature; concepts, scales, fixed points and measurements. Work and Heat Mechanics, definition of work and its limitations. Thermodynamic definition of work; examples, sign convention. Displacement work; as a part of a system boundary, as a whole of a system boundary, expressions for displacement work in various processes through p-v diagrams. Shaft work; Electrical work. Other types of work. Heat; definition, units and sign convention. 10 Hours 1st Hour Brain storming session on subject topics Thermodynamics definition and scope, Microscopic and Macroscopic approaches. Some practical applications of engineering thermodynamic Systems 2nd Hour Characteristics of system boundary and control surface, examples. Thermodynamic properties; definition and units, intensive and extensive properties. 3rd Hour Thermodynamic state, state point, state diagram, path and process, quasi-static -

Thermodynamics the Goal of Thermodynamics Is to Understand How Heat Can Be Converted to Work
Thermodynamics The goal of thermodynamics is to understand how heat can be converted to work Main lesson: Not all the heat energy can be converted to mechanical energy This is because heat energy comes with disorder (entropy), and overall disorder cannot decrease Temperature v T Random directions of velocity Higher temperature means higher velocities v Add up energy of all molecules: Internal Energy of gas U Mechanical energy: all atoms move in the same direction 1 Mv2 2 Statistical mechanics For one atom 1 1 1 1 1 1 3 E = mv2 + mv2 + mv2 = kT + kT + kT = kT h i h2 xi h2 yi h2 z i 2 2 2 2 Ideal gas: No Potential energy from attraction between atoms 3 U = NkT h i 2 v T Pressure v Pressure is caused because atoms T bounce off the wall Lx p − x px ∆px =2px 2L ∆t = x vx ∆p 2mv2 mv2 F = x = x = x ∆t 2Lx Lx ∆p 2mv2 mv2 F = x = x = x ∆t 2Lx Lx 1 mv2 =2 kT = kT h xi ⇥ 2 kT F = Lx Pressure v F kT 1 kT A = LyLz P = = = A Lx LyLz V NkT Many particles P = V Lx PV = NkT Volume V = LxLyLz Work Lx ∆V = A ∆Lx v A = LyLz Work done BY the gas ∆W = F ∆Lx We can write this as ∆W =(PA) ∆Lx = P ∆V This is useful because the body could have a generic shape Internal energy of gas decreases U U ∆W ! − Gas expands, work is done BY the gas P dW = P dV Work done is Area under curve V P Gas is pushed in, work is done ON the gas dW = P dV − Work done is negative of Area under curve V By convention, we use POSITIVE sign for work done BY the gas Getting work from Heat Gas expands, work is done BY the gas P dW = P dV V Volume in increases Internal energy decreases .. -

Appendix a Rate of Entropy Production and Kinetic Implications
Appendix A Rate of Entropy Production and Kinetic Implications According to the second law of thermodynamics, the entropy of an isolated system can never decrease on a macroscopic scale; it must remain constant when equilib- rium is achieved or increase due to spontaneous processes within the system. The ramifications of entropy production constitute the subject of irreversible thermody- namics. A number of interesting kinetic relations, which are beyond the domain of classical thermodynamics, can be derived by considering the rate of entropy pro- duction in an isolated system. The rate and mechanism of evolution of a system is often of major or of even greater interest in many problems than its equilibrium state, especially in the study of natural processes. The purpose of this Appendix is to develop some of the kinetic relations relating to chemical reaction, diffusion and heat conduction from consideration of the entropy production due to spontaneous processes in an isolated system. A.1 Rate of Entropy Production: Conjugate Flux and Force in Irreversible Processes In order to formally treat an irreversible process within the framework of thermo- dynamics, it is important to identify the force that is conjugate to the observed flux. To this end, let us start with the question: what is the force that drives heat flux by conduction (or heat diffusion) along a temperature gradient? The intuitive answer is: temperature gradient. But the answer is not entirely correct. To find out the exact force that drives diffusive heat flux, let us consider an isolated composite system that is divided into two subsystems, I and II, by a rigid diathermal wall, the subsystems being maintained at different but uniform temperatures of TI and TII, respectively. -

25 Kw Low-Temperature Stirling Engine for Heat Recovery, Solar, and Biomass Applications
25 kW Low-Temperature Stirling Engine for Heat Recovery, Solar, and Biomass Applications Lee SMITHa, Brian NUELa, Samuel P WEAVERa,*, Stefan BERKOWERa, Samuel C WEAVERb, Bill GROSSc aCool Energy, Inc, 5541 Central Avenue, Boulder CO 80301 bProton Power, Inc, 487 Sam Rayburn Parkway, Lenoir City TN 37771 cIdealab, 130 W. Union St, Pasadena CA 91103 *Corresponding author: [email protected] Keywords: Stirling engine, waste heat recovery, concentrating solar power, biomass power generation, low-temperature power generation, distributed generation ABSTRACT This paper covers the design, performance optimization, build, and test of a 25 kW Stirling engine that has demonstrated > 60% of the Carnot limit for thermal to electrical conversion efficiency at test conditions of 329 °C hot side temperature and 19 °C rejection temperature. These results were enabled by an engine design and construction that has minimal pressure drop in the gas flow path, thermal conduction losses that are limited by design, and which employs a novel rotary drive mechanism. Features of this engine design include high-surface- area heat exchangers, nitrogen as the working fluid, a single-acting alpha configuration, and a design target for operation between 150 °C and 400 °C. 1 1. INTRODUCTION Since 2006, Cool Energy, Inc. (CEI) has designed, fabricated, and tested five generations of low-temperature (150 °C to 400 °C) Stirling engines that drive internally integrated electric alternators. The fifth generation of engine built by Cool Energy is rated at 25 kW of electrical power output, and is trade-named the ThermoHeart® Engine. Sources of low-to-medium temperature thermal energy, such as internal combustion engine exhaust, industrial waste heat, flared gas, and small-scale solar heat, have relatively few methods available for conversion into more valuable electrical energy, and the thermal energy is usually wasted. -

The First Law of Thermodynamics Continued Pre-Reading: §19.5 Where We Are
Lecture 7 The first law of thermodynamics continued Pre-reading: §19.5 Where we are The pressure p, volume V, and temperature T are related by an equation of state. For an ideal gas, pV = nRT = NkT For an ideal gas, the temperature T is is a direct measure of the average kinetic energy of its 3 3 molecules: KE = nRT = NkT tr 2 2 2 3kT 3RT and vrms = (v )av = = r m r M p Where we are We define the internal energy of a system: UKEPE=+∑∑ interaction Random chaotic between atoms motion & molecules For an ideal gas, f UNkT= 2 i.e. the internal energy depends only on its temperature Where we are By considering adding heat to a fixed volume of an ideal gas, we showed f f Q = Nk∆T = nR∆T 2 2 and so, from the definition of heat capacity Q = nC∆T f we have that C = R for any ideal gas. V 2 Change in internal energy: ∆U = nCV ∆T Heat capacity of an ideal gas Now consider adding heat to an ideal gas at constant pressure. By definition, Q = nCp∆T and W = p∆V = nR∆T So from ∆U = Q W − we get nCV ∆T = nCp∆T nR∆T − or Cp = CV + R It takes greater heat input to raise the temperature of a gas a given amount at constant pressure than constant volume YF §19.4 Ratio of heat capacities Look at the ratio of these heat capacities: we have f C = R V 2 and f + 2 C = C + R = R p V 2 so C p γ = > 1 CV 3 For a monatomic gas, CV = R 3 5 2 so Cp = R + R = R 2 2 C 5 R 5 and γ = p = 2 = =1.67 C 3 R 3 YF §19.4 V 2 Problem An ideal gas is enclosed in a cylinder which has a movable piston. -

Thermodynamics of Manufacturing Processes—The Workpiece and the Machinery
inventions Article Thermodynamics of Manufacturing Processes—The Workpiece and the Machinery Jude A. Osara Mechanical Engineering Department, University of Texas at Austin, EnHeGi Research and Engineering, Austin, TX 78712, USA; [email protected] Received: 27 March 2019; Accepted: 10 May 2019; Published: 15 May 2019 Abstract: Considered the world’s largest industry, manufacturing transforms billions of raw materials into useful products. Like all real processes and systems, manufacturing processes and equipment are subject to the first and second laws of thermodynamics and can be modeled via thermodynamic formulations. This article presents a simple thermodynamic model of a manufacturing sub-process or task, assuming multiple tasks make up the entire process. For example, to manufacture a machined component such as an aluminum gear, tasks include cutting the original shaft into gear blanks of desired dimensions, machining the gear teeth, surfacing, etc. The formulations presented here, assessing the workpiece and the machinery via entropy generation, apply to hand-crafting. However, consistent isolation and measurement of human energy changes due to food intake and work output alone pose a significant challenge; hence, this discussion focuses on standardized product-forming processes typically via machine fabrication. Keywords: thermodynamics; manufacturing; product formation; entropy; Helmholtz energy; irreversibility 1. Introduction Industrial processes—manufacturing or servicing—involve one or more forms of electrical, mechanical, chemical (including nuclear), and thermal energy conversion processes. For a manufactured component, an interpretation of the first law of thermodynamics indicates that the internal energy content of the component is the energy that formed the product [1]. Cursorily, this sums all the work that goes into the manufacturing process from electrical to mechanical, chemical, and thermal power consumption by the manufacturing equipment. -

Supercritical CO 2 Cycles for Gas Turbine
Power Gen International December 8-10, Las Vegas, Nevada SUPERCRITICAL CO2 CYCLES FOR GAS TURBINE COMBINED CYCLE POWER PLANTS Timothy J. Held Chief Technology Officer Echogen Power Systems, LLC Akron, Ohio [email protected] ABSTRACT Supercritical carbon dioxide (sCO2) used as the working fluid in closed loop power conversion cycles offers significant advantages over steam and organic fluid based Rankine cycles. Echogen Power Systems LLC has developed several variants of sCO2 cycles that are optimized for bottoming and heat recovery applications. In contrast to cycles used in previous nuclear and CSP studies, these cycles are highly effective in extracting heat from a sensible thermal source such as gas turbine exhaust or industrial process waste heat, and then converting it to power. In this study, conceptual designs of sCO2 heat recovery systems are developed for gas turbine combined cycle (GTCC) power generation over a broad range of system sizes, ranging from distributed generation (~5MW) to utility scale (> 500MW). Advanced cycle simulation tools employing non-linear multivariate constrained optimization processes are combined with system and plant cost models to generate families of designs with different cycle topologies. The recently introduced EPS100 [1], the first commercial-scale sCO2 heat recovery engine, is used to validate the results of the cost and performance models. The results of the simulation process are shown as system installed cost as a function of power, which allows objective comparisons between different cycle architectures, and to other power generation technologies. Comparable system cost and performance studies for conventional steam-based GTCC are presented on the basis of GT-Pro™ simulations [2]. -

Removing the Mystery of Entropy and Thermodynamics – Part II Harvey S
Table IV. Example of audit of energy loss in collision (with a mass attached to the end of the spring). Removing the Mystery of Entropy and Thermodynamics – Part II Harvey S. Leff, Reed College, Portland, ORa,b Part II of this five-part series is focused on further clarification of entropy and thermodynamics. We emphasize that entropy is a state function with a numerical value for any substance in thermodynamic equilibrium with its surroundings. The inter- pretation of entropy as a “spreading function” is suggested by the Clausius algorithm. The Mayer-Joule principle is shown to be helpful in understanding entropy changes for pure work processes. Furthermore, the entropy change when a gas expands or is compressed, and when two gases are mixed, can be understood qualitatively in terms of spatial energy spreading. The question- answer format of Part I1 is continued, enumerating main results in Key Points 2.1-2.6. • What is the significance of entropy being a state Although the linear correlation in Fig. 1 is quite striking, the function? two points to be emphasized here are: (i) the entropy value In Part I, we showed that the entropy of a room temperature of each solid is dependent on the energy added to and stored solid can be calculated at standard temperature and pressure by it, and (ii) the amount of energy needed for the heating using heat capacity data from near absolute zero, denoted by T process from T = 0 + K to T = 298.15 K differs from solid to = 0+, to room temperature, Tf = 298.15 K. -

12/8 and 12/10/2010
PY105 C1 1. Help for Final exam has been posted on WebAssign. 2. The Final exam will be on Wednesday December 15th from 6-8 pm. First Law of Thermodynamics 3. You will take the exam in multiple rooms, divided as follows: SCI 107: Abbasi to Fasullo, as well as Khajah PHO 203: Flynn to Okuda, except for Khajah SCI B58: Ordonez to Zhang 1 2 Heat and Work done by a Gas Thermodynamics Initial: Consider a cylinder of ideal Thermodynamics is the study of systems involving gas at room temperature. Suppose the piston on top of energy in the form of heat and work. the cylinder is free to move vertically without friction. When the cylinder is placed in a container of hot water, heat Equilibrium: is transferred into the cylinder. Where does the heat energy go? Why does the volume increase? 3 4 The First Law of Thermodynamics The First Law of Thermodynamics The First Law is often written as: Some of the heat energy goes into raising the temperature of the gas (which is equivalent to raising the internal energy of the gas). The rest of it does work by raising the piston. ΔEQWint =− Conservation of energy leads to: QEW=Δ + int (the first law of thermodynamics) This form of the First Law says that the change in internal energy of a system is Q is the heat added to a system (or removed if it is negative) equal to the heat supplied to the system Eint is the internal energy of the system (the energy minus the work done by the system (usually associated with the motion of the atoms and/or molecules).