Tennessee Drug Statutes Chart
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Report of the International Narcotics Control Board for 2010
Report of the International Narcotics Control Board involving treatment for cocaine abuse accounted for 510. According to the 2009 AIDS Epidemic Update, 65 per cent of all cases involving treatment for published by the Joint United Nations Programme on substance abuse in 1998, and that figure decreased, in HIV/AIDS and WHO, an estimated 29 per cent of the relative terms, to 49 per cent in 2008. For the past more than 2 million Latin Americans who abuse drugs 10 years, cocaine has been the primary drug of abuse by injection are infected with HIV. HIV epidemics among persons treated for drug problems in the region. among such drug abusers in the region tend to be concentrated in the Southern Cone. It is estimated that 506. Demand for “crack” cocaine appears to be in Argentina alone, almost half of the persons who emerging in some countries in South America. In 2008, abuse drugs by injection are infected with HIV. seizures of “crack” cocaine were reported in Argentina, Brazil, Chile, Paraguay and Venezuela (Bolivarian Republic of). In the Bolivarian Republic of Venezuela, C. Asia lifetime prevalence of the abuse of “crack” cocaine among the population aged 15-70 is 11.9 per cent. In East and South-East Asia that country, about a quarter of the persons who received treatment for drug addiction were addicted to 1. Major developments “crack” cocaine. In 2010, the Government of Brazil launched its integrated plan to combat “crack” cocaine 511. In East and South-East Asia, progress in reducing and other drugs. opium production is under threat, owing to an upswing in opium poppy cultivation during the 2009 growing 507. -

Opioid Receptorsreceptors
OPIOIDOPIOID RECEPTORSRECEPTORS defined or “classical” types of opioid receptor µ,dk and . Alistair Corbett, Sandy McKnight and Graeme Genes encoding for these receptors have been cloned.5, Henderson 6,7,8 More recently, cDNA encoding an “orphan” receptor Dr Alistair Corbett is Lecturer in the School of was identified which has a high degree of homology to Biological and Biomedical Sciences, Glasgow the “classical” opioid receptors; on structural grounds Caledonian University, Cowcaddens Road, this receptor is an opioid receptor and has been named Glasgow G4 0BA, UK. ORL (opioid receptor-like).9 As would be predicted from 1 Dr Sandy McKnight is Associate Director, Parke- their known abilities to couple through pertussis toxin- Davis Neuroscience Research Centre, sensitive G-proteins, all of the cloned opioid receptors Cambridge University Forvie Site, Robinson possess the same general structure of an extracellular Way, Cambridge CB2 2QB, UK. N-terminal region, seven transmembrane domains and Professor Graeme Henderson is Professor of intracellular C-terminal tail structure. There is Pharmacology and Head of Department, pharmacological evidence for subtypes of each Department of Pharmacology, School of Medical receptor and other types of novel, less well- Sciences, University of Bristol, University Walk, characterised opioid receptors,eliz , , , , have also been Bristol BS8 1TD, UK. postulated. Thes -receptor, however, is no longer regarded as an opioid receptor. Introduction Receptor Subtypes Preparations of the opium poppy papaver somniferum m-Receptor subtypes have been used for many hundreds of years to relieve The MOR-1 gene, encoding for one form of them - pain. In 1803, Sertürner isolated a crystalline sample of receptor, shows approximately 50-70% homology to the main constituent alkaloid, morphine, which was later shown to be almost entirely responsible for the the genes encoding for thedk -(DOR-1), -(KOR-1) and orphan (ORL ) receptors. -

(Butorphanol Tartrate) Nasal Spray
NDA 19-890/S-017 Page 3 ® STADOL (butorphanol tartrate) Injection, USP STADOL NS® (butorphanol tartrate) Nasal Spray DESCRIPTION Butorphanol tartrate is a synthetically derived opioid agonist-antagonist analgesic of the phenanthrene series. The chemical name is (-)-17-(cyclobutylmethyl) morphinan-3, 14-diol [S- (R*,R*)] - 2,3 - dihydroxybutanedioate (1:1) (salt). The molecular formula is C21H29NO2,C4H6O6, which corresponds to a molecular weight of 477.55 and the following structural formula: Butorphanol tartrate is a white crystalline substance. The dose is expressed as the tartrate salt. One milligram of the salt is equivalent to 0.68 mg of the free base. The n-octanol/aqueous buffer partition coefficient of butorphanol is 180:1 at pH 7.5. STADOL (butorphanol tartrate) Injection, USP, is a sterile, parenteral, aqueous solution of butorphanol tartrate for intravenous or intramuscular administration. In addition to 1 or 2 mg of butorphanol tartrate, each mL of solution contains 3.3 mg of citric acid, 6.4 mg sodium citrate, and 6.4 mg sodium chloride, and 0.1 mg benzethonium chloride (in multiple dose vial only) as a preservative. NDA 19-890/S-017 Page 4 STADOL NS (butorphanol tartrate) Nasal Spray is an aqueous solution of butorphanol tartrate for administration as a metered spray to the nasal mucosa. Each bottle of STADOL NS contains 2.5 mL of a 10 mg/mL solution of butorphanol tartrate with sodium chloride, citric acid, and benzethonium chloride in purified water with sodium hydroxide and/or hydrochloric acid added to adjust the pH to 5.0. The pump reservoir must be fully primed (see PATIENT INSTRUCTIONS) prior to initial use. -

Phencyclidine: an Update
Phencyclidine: An Update U.S. DEPARTMENT OF HEALTH AND HUMAN SERVICES • Public Health Service • Alcohol, Drug Abuse and Mental Health Administration Phencyclidine: An Update Editor: Doris H. Clouet, Ph.D. Division of Preclinical Research National Institute on Drug Abuse and New York State Division of Substance Abuse Services NIDA Research Monograph 64 1986 DEPARTMENT OF HEALTH AND HUMAN SERVICES Public Health Service Alcohol, Drug Abuse, and Mental Health Administratlon National Institute on Drug Abuse 5600 Fishers Lane Rockville, Maryland 20657 For sale by the Superintendent of Documents, U.S. Government Printing Office Washington, DC 20402 NIDA Research Monographs are prepared by the research divisions of the National lnstitute on Drug Abuse and published by its Office of Science The primary objective of the series is to provide critical reviews of research problem areas and techniques, the content of state-of-the-art conferences, and integrative research reviews. its dual publication emphasis is rapid and targeted dissemination to the scientific and professional community. Editorial Advisors MARTIN W. ADLER, Ph.D. SIDNEY COHEN, M.D. Temple University School of Medicine Los Angeles, California Philadelphia, Pennsylvania SYDNEY ARCHER, Ph.D. MARY L. JACOBSON Rensselaer Polytechnic lnstitute National Federation of Parents for Troy, New York Drug Free Youth RICHARD E. BELLEVILLE, Ph.D. Omaha, Nebraska NB Associates, Health Sciences Rockville, Maryland REESE T. JONES, M.D. KARST J. BESTEMAN Langley Porter Neuropsychiatric lnstitute Alcohol and Drug Problems Association San Francisco, California of North America Washington, D.C. DENISE KANDEL, Ph.D GILBERT J. BOTV N, Ph.D. College of Physicians and Surgeons of Cornell University Medical College Columbia University New York, New York New York, New York JOSEPH V. -

Research Brief New Reports, Bills and Updates of Latest Research
Research Brief New reports, bills and updates of latest research Parliamentary Library and Information Service Department of Parliamentary Services ISSN 1836-7828 (Print) 1836-8050 (Online) Number 3 February 2014 Drugs, Poisons and Controlled Substances (Poppy Cultivation and Processing) Amendment Bill 2013 This Research Brief includes the following sections: Introduction .............................................................................................................. 1 1. Second Reading Speech ...................................................................................... 1 2. Background ........................................................................................................... 2 History of the Opium Poppy and the Poppy Industry ........................................................... 2 Development of the Tasmanian Poppy Industry ..................................................................... 3 Regulation of the Poppy Industry and the Thebaine Poppy .................................................. 5 The Tasmanian Poppy Industry Today and Processor Plans for Expansion ...................... 7 Expanding the Poppy Industry to Victoria ................................................................................ 8 3. The Bill ................................................................................................................... 9 4. Other Jurisdiction ............................................................................................. 11 Appendix 1. Description of Morphine, -

Drugs of Abuseon September Archived 13-10048 No
U.S. DEPARTMENT OF JUSTICE DRUG ENFORCEMENT ADMINISTRATION WWW.DEA.GOV 9, 2014 on September archived 13-10048 No. v. Stewart, in U.S. cited Drugs of2011 Abuse EDITION A DEA RESOURCE GUIDE V. Narcotics WHAT ARE NARCOTICS? Also known as “opioids,” the term "narcotic" comes from the Greek word for “stupor” and originally referred to a variety of substances that dulled the senses and relieved pain. Though some people still refer to all drugs as “narcot- ics,” today “narcotic” refers to opium, opium derivatives, and their semi-synthetic substitutes. A more current term for these drugs, with less uncertainty regarding its meaning, is “opioid.” Examples include the illicit drug heroin and pharmaceutical drugs like OxyContin®, Vicodin®, codeine, morphine, methadone and fentanyl. WHAT IS THEIR ORIGIN? The poppy papaver somniferum is the source for all natural opioids, whereas synthetic opioids are made entirely in a lab and include meperidine, fentanyl, and methadone. Semi-synthetic opioids are synthesized from naturally occurring opium products, such as morphine and codeine, and include heroin, oxycodone, hydrocodone, and hydromorphone. Teens can obtain narcotics from friends, family members, medicine cabinets, pharmacies, nursing 2014 homes, hospitals, hospices, doctors, and the Internet. 9, on September archived 13-10048 No. v. Stewart, in U.S. cited What are common street names? Street names for various narcotics/opioids include: ➔ Hillbilly Heroin, Lean or Purple Drank, OC, Ox, Oxy, Oxycotton, Sippin Syrup What are their forms? Narcotics/opioids come in various forms including: ➔ T ablets, capsules, skin patches, powder, chunks in varying colors (from white to shades of brown and black), liquid form for oral use and injection, syrups, suppositories, lollipops How are they abused? ➔ Narcotics/opioids can be swallowed, smoked, sniffed, or injected. -

Ketamine Homogeneous Enzyme Immunoassay (HEIA™)
Ketamine Homogeneous Enzyme Immunoassay (HEIA™) Exclusively from Immunalysis Formula: C13H16CINO Semi-Quantitative or Qualitative Testing Systematic Name: (RS)- 2- (2- chlorophenyl)- 2- (methylamino)cyclohexanone Accurate and reliable Brand Names: Ketanest®, Ketaset®, Ketalar® Ready to use About Ketamine: Ketamine is an anesthetic agent used in the United States since 1972 for veterinary and pediatric medicine. It is also used in the treatment of depression and postoperative pain management. However, in recent years it has gained popularity as a street drug used at clubs and raves due to its hallucinogenic effects. Administration: Oral; intravenous; intramuscular; insufflation Elimination: Ketamine metabolizes by N-demethylation to Norketamine and further dehydrogenates to Dehydronorketamine. After 72 hours of a single dose, 2.3% of Ketamine is unchanged, 1.6% is Norketamine, 16.2% is Dehydronorketamine, and 80% is hydroxylated derivatives of Ketamine.1,2 Abuse Potential: An overdose can cause unconsciousness and dangerously slowed breathing. 1) R. Baselt, Disposition of Toxic Drugs and Chemicals in Man, Fourth Edition, p. 412-414. 2) K. Moore, J.Skerov, B.Levine, and A.Jacobs, Urine Concentrations of Ketamine and Norketamine Following Illegal Consumption, J.Anal, Toxicol. 25: 583-588 (2001). Ketanest® is a registered trademark of Pfizer, Inc., Ketaset® is a trademark of ZOETIS W LLC., Ketalar® is a trademark of PAR STERILE PRODUCTS, LLC. Tel 909.482.0840 | Toll Free 888.664.8378 | Fax 909.482.0850 ISO13485:2003 www.immunalysis.com CERTIFIED -

Torbugesic® Orbugesic®
In horses, intravenous dosages of butorphanol Repeated administration of butorphanol at 1 mg/kg DOSAGE ranging from 0.05 to 0.4 mg/kg were shown to be (10 times the recommended dose) every four hours The recommended dosage in the horse is 0.1 mg 7. Popio, K.A. et al: “Hemodynamic and Respiratory eff ective in alleviating visceral and superfi cial pain for 48 hours caused constipation in one of two of butorphanol per kilogram of body weight Eff ects of Morphine and Butorphanol,” Clin. for at least 4 hours, as illustrated in the following horses. (0.05 mg/lb) by intravenous injection. This is Pharmacol. Ther. 23: 281–287, 1978. fi gure: equivalent to 5 mL of TORBUGESIC for each 1000 lbs 8. Robertson, J.T. and Muir, W.W.: “Cardiopulmonary Subacute Equine Studies body weight. Eff ects of Butorphanol Tartrate in Horses,” Am. J. Analgesic Eff ects of Butorphanol Given at Horses were found to tolerate butorphanol given The dose may be repeated within 3 to 4 hours Vet. Res. 42: 41–44, 1981. Various Dosages in Horses with Abdominal Pain intravenously at dosages of 0.1, 0.3 and 0.5 mg/kg but treatment should not exceed 48 hours. 9. Kalpravidh, M. et al: “Eff ects of Butorphanol, every 4 hours for 48 hours followed by once daily Pre-clinical model studies and clinical fi eld trials in Flunixin, Levorphanol, Morphine, Pentazocine injections for a total of 21 days. The only detectable horses demonstrate that the analgesic eff ects of and Xylazine in Ponies,” Am. -
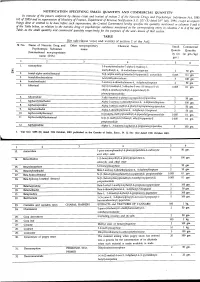
NOTIFICATION SPECIFYING SMALL QUANTITY and COMMERCIAL QUANTITYI Ly of the Pozoersconferred (Xxiiiil
NOTIFICATION SPECIFYING SMALL QUANTITY AND COMMERCIAL QUANTITYI ly of the pozoersconferred (xxiiiil . - lleyise by clansesfuiia) and of section2 of the Nnrcotic Drrtgsnnd pnlchotropic SrtbstancesAct, 19g5 (61 of 1985)and in supersessionof Ministry of Ftnnnce, (D Departmentof'Reuenue Noiification s.o.527 aotra i'6tt,1uli, tssi, exceptas respects things doneor omitted to be done .beforesuch supersession,the Cential Gooernmenihereby spectfiesthe qtnntitrl ,nlnt'ionedin coltnnnsS and 6 of the Tablebelow, in relationto.the narcoticdrug or psychotropicsubstnnce mentioned.ii tlr', ,1urrponding entnl in columns2 to 4 of thesaid Table,as the small quantitrl nnd commercialquantity respectiailyfor the purposesof the said clnuse:sof ttrit seciion. TABLE [Seesub-clause vii(a) and xxiii(a) of section 2 of the Act] Sl No. Name of Narcotic Drug and Other non-proprietary Chemical Name Small Commercial Psychotropic Substance name Quanti- Quantity (Intemational non-proprietary ity (in (in gm./kg.) name (INN) Acetorphine 3-0-acetyltetrahydro-7-alpha-(l-hydroxy-l- methylbutyt)-o, l4-endoetheno-onpavine $ 50 9.. 1\) Acetyl-alpha-methylfen N-[-(alpha-methylphenethyl)-4-piperidy]l acetanilide 0.1 g-. J. Acetyldihydrocodeine 100 4. Acetylmethadol 3-acetoxy-6-dimethylamino-4, 4 lheptane 50 gm. 5. Alfentanil -ethyl-4, N-[1-[2-( 5-dihydro-S-oxo-lH-tetrazol-t-yt) 01 gm. ethyll-4-(methoxymethyl)-4-piperidinyll -N- ylpropanamide Allyprodine 3-allyl Jmethyl-4-phenyl-4 Alpha-3-acetoxy-6-dimethylamino-4,Ld lheptane 100 gm Alpha-3-ethyl-l-methyl-4-phenyl-4-propionox 50 gm. 9.7' AlphamethadolArPnametnaool Alpha-6-dimethylamino-4, 4-diphenyl-3-heptanol 2 S0 gm. 10. Alphu-*"thylf"ntur,yl 11. -

Psycho Pharmacology © by Springer-Verlag 1976
Psychopharmacology 47, 65- 69 (1976) Psycho pharmacology © by Springer-Verlag 1976 Generalization of Morphine and Lysergic Acid Diethylamide (LSD) Stimulus Properties to Narcotic Analgesics I. D. HIRSCHHORN* and J. A. ROSECRANS Department of Pharmacology, Medical College of Virginia, Richmond, Virginia 23298, U.S.A. Abstract. The present investigation sought to deter- Physical dependence is not always associated with drug mine whether the stimulus properties of morphine craving and a high abuse liability. Some narcotic- and lysergic acid diethylamide (LSD) would gener- antagonist analgesics, such as cyclazocine and nalor- alize to several narcotic analgesics which vary in their phine, produce physical dependence with chronic subjective effects. Morphine and saline served as administration, but withdrawal does not result in drug discriminative stimuli for one group of rats in a 2-1ever seeking behavior (Martin et al., 1965; Martin and discrimination task. LSD and saline were discrimina- Gorodetzky, 1965). However, both cyclazocine and tive stimuli for a second group. Depression of one nalorphine have subjective side effects characterized lever in an operant chamber resulted in reinforcement by dysphoria and hallucinations (Haertzen, 1970) following the administration of morphine or LSD which render them unsuitable for therapeutic use. and the opposite lever was reinforced after saline. Pentazocine, a less potent antagonist of morphine After discriminated responding was stable, stimulus than cyclazocine and nalorphine, more closely resem- generalization tests with narcotic analgesics and bles morphine in its pharmacological effects and has antagonists showed that the stimulus properties of a somewhat greater incidence of non-medical use morphine generalized to methadone and meperidine, than antagonists of the nalorphine type (Paddock and partially to pentazocine, all of which produce etal., 1969). -

Narcotic Drugs Stupefiants Estupefacientes
E/INCB/1993/21Supp.6 INTERNATIONAL NARCOTICS CONTROL BOARD - VIENNA SUPPLEMENT No. 6 TO NARCOTIC DRUGS ESTIMATED WORLD REQUIREMENTS FOR 1994 STATISTICS FOR 1992 ESTIMATES UPDATED AS OF 30 JUNE 1994 ORGANE INTERNATIONAL DE CONTROLE DES STUPEFIANTS - VIENNE SUPPLEMENT N° 6 A r STUPEFIANTS EVALUATIONS DES BESOINS DU MONDE POUR 1994 STATISTIQUES POUR 1992 EVALUATIONS A JOUR AU 30 JUlN 1994 JUNTA INTERNACIONAL DE FISCALlZACION DE ESTUPEFACIENTES - VIENA SUPLEMENTO N.o 6 A ESTUPEFACIENTES PREVISIONES DE LAS NECESIDADES MUNDIALES PARA 1994 ESTADfsTICAS PARA 1992 PREVISIONES ACTUALlZADAS AL 30 DE JUNIO DE 1994 ~If..~~ ~ ~-tR UNITED NATIONS - NATIONS UNIES - NACIONES UNIDAS 1994 The updating of Table A is carried out by means of 12 monthly supplements. In order to facilitate the task of the exporting countries, the 12 supplements now report all the totals of the estimates and not only the amended data. In this way, each supplement cancels and replaces the published table in its entirety. In order to accelerate the transmission of the supplements to the competent national authorities, the 12 supplements will appear in English. Reading of these 12 supplements in French and Spanish may be facilitated by consulting the indexes of countries and territories and of narcotic drugs appearing in the annual publication. La mise El jour du tableau A s'effectue au moyen de douze supplements mensuels. Afin de faciliter la tache des pays exportateurs, les douze supplements contiennent tous les totaux des evaluations et non pas seulement les chiffres qui ont ete modifies. De celte maniere, chaque supplement annule et remplace entierement le tableau publie. -

ARK™ Ketamine Assay Package Insert
The ARK Ketamine Assay provides only a preliminary analytical test result. A more specific alternative chemical method must be used in order to obtain a confirmed positive analytical result. Gas Chromatography/Mass Spectrometry (GC/MS) or Liquid Chromatography/tandem Mass Spectrometry (LC-MS/MS) is the preferred confirmatory method. Clinical consideration and professional judgment should be exercised with any drug test result, particularly when the For Criminal Justice and Forensic Use Only preliminary test result is positive. 3 SUMMARY AND EXPLANATION OF THE TEST Ketamine ((+/-)-2-(2-chlorophenyl)-2-(methylamino)cyclohexanone) is a synthetic, non- RK™ Ketamine Assay barbiturate and rapid-acting general anesthetic that is indicated for use in both human and A veterinary surgical procedures.1,2 This ARK Diagnostics, Inc. package insert for the ARK Ketamine Assay must be read prior to use. Package insert instructions must be followed accordingly. The assay provides a simple Ketamine is a Schedule III substance under the United States Controlled Substances Act for and rapid analytical screening procedure for detecting ketamine in urine. Reliability of the its potential for abuse and risk of dependence. Ketamine is structurally and pharmacologically assay results cannot be guaranteed if there are any deviations from the instructions in this similar to phencyclidine (PCP), but is less potent, has a faster onset and shorter duration of package insert. action relative to PCP. Ketamine produces a variety of symptoms including, but not limited to anxiety, dysphoria, disorientation, insomnia, flashbacks, hallucinations, and psychotic episodes.1,3 CUSTOMER SERVICE Following administration in humans, ketamine is N-demethylated by liver microsomal ARK Diagnostics, Inc.