Hymenoptera: Formicidae), with Resurrection of the Genera Colobopsis and Dinomyrmex
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Unravelling the Diversity Behind the Ophiocordyceps Unilateralis (Ophiocordycipitaceae) Complex: Three New Species of Zombie-Ant Fungi from the Brazilian Amazon
Phytotaxa 220 (3): 224–238 ISSN 1179-3155 (print edition) www.mapress.com/phytotaxa/ PHYTOTAXA Copyright © 2015 Magnolia Press Article ISSN 1179-3163 (online edition) http://dx.doi.org/10.11646/phytotaxa.220.3.2 Unravelling the diversity behind the Ophiocordyceps unilateralis (Ophiocordycipitaceae) complex: Three new species of zombie-ant fungi from the Brazilian Amazon JOÃO P. M. ARAÚJO1*, HARRY C. EVANS2, DAVID M. GEISER3, WILLIAM P. MACKAY4 & DAVID P. HUGHES1, 5* 1 Department of Biology, Penn State University, University Park, Pennsylvania, United States of America. 2 CAB International, E-UK, Egham, Surrey, United Kingdom 3 Department of Plant Pathology, Penn State University, University Park, Pennsylvania, United States of America. 4 Department of Biological Sciences, University of Texas at El Paso, 500 West University Avenue, El Paso, Texas, United States of America. 5 Department of Entomology, Penn State University, University Park, Pennsylvania, United States of America. * email: [email protected]; [email protected] Abstract In tropical forests, one of the most commonly encountered relationships between parasites and insects is that between the fungus Ophiocordyceps (Ophiocordycipitaceae, Hypocreales, Ascomycota) and ants, especially within the tribe Campono- tini. Here, we describe three newly discovered host-specific species, Ophiocordyceps camponoti-atricipis, O. camponoti- bispinosi and O. camponoti-indiani, on Camponotus ants from the central Amazonian region of Brazil, which can readily be separated using morphological traits, in particular the shape and behavior of the ascospores. DNA sequence data support inclusion of these species within the Ophiocordyceps unilateralis complex. Introduction In tropical forests, social insects (ants, bees, termites and wasps) are the most abundant land-dwelling arthropods. -
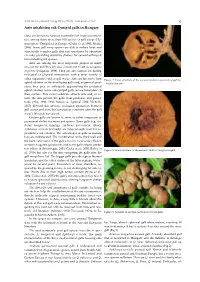
Ants Inhabiting Oak Cynipid Galls in Hungary
North-Western Journal of Zoology 2020, vol.16 (1) - Correspondence: Notes 95 Ants inhabiting oak Cynipid galls in Hungary Oaks are known to harbour extremely rich insect communi- ties, among them more than 100 species of gall wasps (Hy- menoptera: Cynipidae) in Europe (Csóka et al. 2005, Melika 2006). Some gall wasp species are able to induce large and structurally complex galls that can sometimes be abundant on oaks, providing attractive shelters for several arthropod taxa including ant species. Ants are among the most important players in many ecosystems and they are also considered to act as ecosystem engineers (Folgarait, 1998). They are also famous for having ecological or physical interactions with a great variety of other organisms, such as gall wasps. Ants are known to tend Figure 1. Inner structure of the asexual Andricus quercustozae gall in- aphid colonies on the developing galls and, as general pred- habited by ants. ators, they prey on arthropods approaching the protected aphid colonies. Some oak cynipid galls secrete honeydew on their surface. This sweet substrate attracts ants and, in re- turn, the ants protect the galls from predators and parasi- toids (Abe, 1988, 1992; Inouye & Agrawal 2004; Nicholls, 2017). Beyond this obvious ecological interaction between gall wasps and ants, this association continues after the gall wasp’s life cycle has ceased. Certain galls are known to serve as either temporary or permanent shelter for many ant species. Some galls (e.g. An- dricus hungaricus (Hartig), Andricus quercustozae (Bosc), Aphelonyx cerricola (Giraud)) are large enough even for re- productive ant colonies. The advantages of galls as nesting logs are multifaceted. -

Level 1 Fauna Survey of the Gruyere Gold Project Borefields (Harewood 2016)
GOLD ROAD RESOURCES LIMITED GRUYERE PROJECT EPA REFERRAL SUPPORTING DOCUMENT APPENDIX 5: LEVEL 1 FAUNA SURVEY OF THE GRUYERE GOLD PROJECT BOREFIELDS (HAREWOOD 2016) Gruyere EPA Ref Support Doc Final Rev 1.docx Fauna Assessment (Level 1) Gruyere Borefield Project Gold Road Resources Limited January 2016 Version 3 On behalf of: Gold Road Resources Limited C/- Botanica Consulting PO Box 2027 BOULDER WA 6432 T: 08 9093 0024 F: 08 9093 1381 Prepared by: Greg Harewood Zoologist PO Box 755 BUNBURY WA 6231 M: 0402 141 197 T/F: (08) 9725 0982 E: [email protected] GRUYERE BOREFIELD PROJECT –– GOLD ROAD RESOURCES LTD – FAUNA ASSESSMENT (L1) – JAN 2016 – V3 TABLE OF CONTENTS SUMMARY 1. INTRODUCTION .....................................................................................................1 2. SCOPE OF WORKS ...............................................................................................1 3. RELEVANT LEGISTALATION ................................................................................2 4. METHODS...............................................................................................................3 4.1 POTENTIAL VETEBRATE FAUNA INVENTORY - DESKTOP SURVEY ............. 3 4.1.1 Database Searches.......................................................................................3 4.1.2 Previous Fauna Surveys in the Area ............................................................3 4.1.3 Existing Publications .....................................................................................5 4.1.4 Fauna -

Morphology of the Mandibular Gland of the Ant Paraponera Clavata (Hymenoptera: Paraponerinae)
Received: 9 October 2018 Revised: 17 January 2019 Accepted: 2 February 2019 DOI: 10.1002/jemt.23242 RESEARCH ARTICLE Morphology of the mandibular gland of the ant Paraponera clavata (Hymenoptera: Paraponerinae) Thito Thomston Andrade1 | Wagner Gonzaga Gonçalves2 | José Eduardo Serrão2 | Luiza Carla Barbosa Martins1 1Programa de Pós-Graduação em Biodiversidade, Ambiente e Saúde, Abstract Departamento de Biologia e Química, The ant Paraponera clavata (Fabricius, 1775) is the only extant species of Paraponerinae and is Universidade Estadual do Maranhão, Caxias, widely distributed in Brazilian forests. Aspects of its biology are documented extensively in the Maranhão, Brazil literature; however, knowledge of P. clavata internal morphology, specifically of exocrine glands, 2Departamento de Biologia Geral, Universidade Federal de Viçosa, Viçosa, is restricted to the venom apparatus. The objective of this study was to describe the mandibular Minas Gerais, Brazil gland morphology of P. clavata workers. The mandibular gland is composed of a reservoir con- nected to a cluster of Type III secretory cells with cytoplasm rich in mitochondria and lipid drop- Correspondence lets, similar to that of other ants. Notably, the glandular secretion is rich in protein and has a Luiza Carla Barbosa Martins, Programa de Pós- Graduação em Biodiversidade, Ambiente e solid aspect. This is the first morphological description of the mandibular gland of P. clavata. Saúde, Departamento de Biologia e Química, Universidade Estadual do Maranhão, Caxias, Research Highlights Maranhão, Brazil. This study presents the morphological description of the mandibular gland of Paraponera clavata Email: [email protected] (Hymenoptera: Paraponerinae). Singular characteristics of the gland are described: the glandular Review Editor: George Perry secretion is rich in protein and has a solid aspect. -

Nutritional Ecology of the Carpenter Ant Camponotus Pennsylvanicus (De Geer): Macronutrient Preference and Particle Consumption
Nutritional Ecology of the Carpenter Ant Camponotus pennsylvanicus (De Geer): Macronutrient Preference and Particle Consumption Colleen A. Cannon Dissertation submitted to the Faculty of the Virginia Polytechnic Institute and State University in partial fulfillment of the requirements for the degree of Doctor of Philosophy in Entomology Richard D. Fell, Chairman Jeffrey R. Bloomquist Richard E. Keyel Charles Kugler Donald E. Mullins June 12, 1998 Blacksburg, Virginia Keywords: diet, feeding behavior, food, foraging, Formicidae Copyright 1998, Colleen A. Cannon Nutritional Ecology of the Carpenter Ant Camponotus pennsylvanicus (De Geer): Macronutrient Preference and Particle Consumption Colleen A. Cannon (ABSTRACT) The nutritional ecology of the black carpenter ant, Camponotus pennsylvanicus (De Geer) was investigated by examining macronutrient preference and particle consumption in foraging workers. The crops of foragers collected in the field were analyzed for macronutrient content at two-week intervals through the active season. Choice tests were conducted at similar intervals during the active season to determine preference within and between macronutrient groups. Isolated individuals and small social groups were fed fluorescent microspheres in the laboratory to establish the fate of particles ingested by workers of both castes. Under natural conditions, foragers chiefly collected carbohydrate and nitrogenous material. Carbohydrate predominated in the crop and consisted largely of simple sugars. A small amount of glycogen was present. Carbohydrate levels did not vary with time. Lipid levels in the crop were quite low. The level of nitrogen compounds in the crop was approximately half that of carbohydrate, and exhibited seasonal dependence. Peaks in nitrogen foraging occurred in June and September, months associated with the completion of brood rearing in Camponotus. -

Digging Deeper Into the Ecology of Subterranean Ants: Diversity and Niche Partitioning Across Two Continents
diversity Article Digging Deeper into the Ecology of Subterranean Ants: Diversity and Niche Partitioning across Two Continents Mickal Houadria * and Florian Menzel Institute of Organismic and Molecular Evolution, Johannes-Gutenberg-University Mainz, Hanns-Dieter-Hüsch-Weg 15, 55128 Mainz, Germany; [email protected] * Correspondence: [email protected] Abstract: Soil fauna is generally understudied compared to above-ground arthropods, and ants are no exception. Here, we compared a primary and a secondary forest each on two continents using four different sampling methods. Winkler sampling, pitfalls, and four types of above- and below-ground baits (dead, crushed insects; melezitose; living termites; living mealworms/grasshoppers) were applied on four plots (4 × 4 grid points) on each site. Although less diverse than Winkler samples and pitfalls, subterranean baits provided a remarkable ant community. Our baiting system provided a large dataset to systematically quantify strata and dietary specialisation in tropical rainforest ants. Compared to above-ground baits, 10–28% of the species at subterranean baits were overall more common (or unique to) below ground, indicating a fauna that was truly specialised to this stratum. Species turnover was particularly high in the primary forests, both concerning above-ground and subterranean baits and between grid points within a site. This suggests that secondary forests are more impoverished, especially concerning their subterranean fauna. Although subterranean ants rarely displayed specific preferences for a bait type, they were in general more specialised than above-ground ants; this was true for entire communities, but also for the same species if they foraged in both strata. Citation: Houadria, M.; Menzel, F. -

Easychair Preprint Ant Microbial Endosymbionts and the Emergent
EasyChair Preprint № 1661 Ant microbial endosymbionts and the emergent properties of social groups Alessio Sclocco, Shirlyn Jia Yun Ong and Serafino Teseo EasyChair preprints are intended for rapid dissemination of research results and are integrated with the rest of EasyChair. October 14, 2019 Ant microbial endosymbionts and the emergent properties of social groups Alessio Sclocco1, Shirlyn Jia Yun Ong2 and Serafino Teseo2† 1Netherlands eScience Center, Amsterdam, The Netherlands (E-mail: [email protected]) 2School of Biological Sciences, Nanyang Technological University, Singapore (Tel: +65 63167088; E-mail: [email protected]) Abstract: In the last fifteen years, research on animal models has provided advances on how gut symbiotic microbes affect behavior and its underlying neurophysiology. However, most studies on the gut microbiota only take into exam individual behavior without considering social dynamics. Contrarily, animals and humans live in complex societies where they constantly adjust physiology and behavior to social interactions. To improve our understanding of how microbes and hosts interact and produce functional individual, social and collective phenotypes, we need to broaden our experimental approaches to a group-level dimension. The ideal models for this purpose are social animals living in stable symbioses with microbes, such as eusocial insects. In our research, we investigate Camponotus carpenter ants and their obligate bacterial symbiont Blochmannia from a behavioral ecology perspective. We aim to create ant colonies including differential proportions of bacteria-free individuals by suppressing Blochmannia with antibiotics. Then, using a machine learning-based video tracking system, we will study network features and group-level behavior of such experimental colonies. Keywords: Social Evolution; Microbiota-Gut-Brain Axis; Ants; Group-Level Behavior; Machine Learning; Real Time Data Analysis 1. -

James K. Wetterer
James K. Wetterer Wilkes Honors College, Florida Atlantic University 5353 Parkside Drive, Jupiter, FL 33458 Phone: (561) 799-8648; FAX: (561) 799-8602; e-mail: [email protected] EDUCATION UNIVERSITY OF WASHINGTON, Seattle, WA, 9/83 - 8/88 Ph.D., Zoology: Ecology and Evolution; Advisor: Gordon H. Orians. MICHIGAN STATE UNIVERSITY, East Lansing, MI, 9/81 - 9/83 M.S., Zoology: Ecology; Advisors: Earl E. Werner and Donald J. Hall. CORNELL UNIVERSITY, Ithaca, NY, 9/76 - 5/79 A.B., Biology: Ecology and Systematics. UNIVERSITÉ DE PARIS III, France, 1/78 - 5/78 Semester abroad: courses in theater, literature, and history of art. WORK EXPERIENCE FLORIDA ATLANTIC UNIVERSITY, Wilkes Honors College 8/04 - present: Professor 7/98 - 7/04: Associate Professor Teaching: Biodiversity, Principles of Ecology, Behavioral Ecology, Human Ecology, Environmental Studies, Tropical Ecology, Field Biology, Life Science, and Scientific Writing 9/03 - 1/04 & 5/04 - 8/04: Fulbright Scholar; Ants of Trinidad and Tobago COLUMBIA UNIVERSITY, Department of Earth and Environmental Science 7/96 - 6/98: Assistant Professor Teaching: Community Ecology, Behavioral Ecology, and Tropical Ecology WHEATON COLLEGE, Department of Biology 8/94 - 6/96: Visiting Assistant Professor Teaching: General Ecology and Introductory Biology HARVARD UNIVERSITY, Museum of Comparative Zoology 8/91- 6/94: Post-doctoral Fellow; Behavior, ecology, and evolution of fungus-growing ants Advisors: Edward O. Wilson, Naomi Pierce, and Richard Lewontin 9/95 - 1/96: Teaching: Ethology PRINCETON UNIVERSITY, Department of Ecology and Evolutionary Biology 7/89 - 7/91: Research Associate; Ecology and evolution of leaf-cutting ants Advisor: Stephen Hubbell 1/91 - 5/91: Teaching: Tropical Ecology, Introduction to the Scientific Method VANDERBILT UNIVERSITY, Department of Psychology 9/88 - 7/89: Post-doctoral Fellow; Visual psychophysics of fish and horseshoe crabs Advisor: Maureen K. -

THE TRUE ARMY ANTS of the INDO-AUSTRALIAN AREA (Hymenoptera: Formicidae: Dorylinae)
Pacific Insects 6 (3) : 427483 November 10, 1964 THE TRUE ARMY ANTS OF THE INDO-AUSTRALIAN AREA (Hymenoptera: Formicidae: Dorylinae) By Edward O. Wilson BIOLOGICAL LABORATORIES, HARVARD UNIVERSITY, CAMBRIDGE, MASS., U. S. A. Abstract: All of the known Indo-Australian species of Dorylinae, 4 in Dorylus and 34 in Aenictus, are included in this revision. Eight of the Aenictus species are described as new: artipus, chapmani, doryloides, exilis, huonicus, nganduensis, philiporum and schneirlai. Phylo genetic and numerical analyses resulted in the discarding of two extant subgenera of Aenictus (Typhlatta and Paraenictus) and the loose clustering of the species into 5 informal " groups" within the unified genus Aenictus. A consistency test for phylogenetic characters is discussed. The African and Indo-Australian doryline species are compared, and available information in the biology of the Indo-Australian species is summarized. The " true " army ants are defined here as equivalent to the subfamily Dorylinae. Not included are species of Ponerinae which have developed legionary behavior independently (see Wilson, E. O., 1958, Evolution 12: 24-31) or the subfamily Leptanillinae, which is very distinct and may be independent in origin. The Dorylinae are not as well developed in the Indo-Australian area as in Africa and the New World tropics. Dorylus itself, which includes the famous driver ants, is centered in Africa and sends only four species into tropical Asia. Of these, the most widespread reaches only to Java and the Celebes. Aenictus, on the other hand, is at least as strongly developed in tropical Asia and New Guinea as it is in Africa, with 34 species being known from the former regions and only about 15 from Africa. -

Borowiec Et Al-2020 Ants – Phylogeny and Classification
A Ants: Phylogeny and 1758 when the Swedish botanist Carl von Linné Classification published the tenth edition of his catalog of all plant and animal species known at the time. Marek L. Borowiec1, Corrie S. Moreau2 and Among the approximately 4,200 animals that he Christian Rabeling3 included were 17 species of ants. The succeeding 1University of Idaho, Moscow, ID, USA two and a half centuries have seen tremendous 2Departments of Entomology and Ecology & progress in the theory and practice of biological Evolutionary Biology, Cornell University, Ithaca, classification. Here we provide a summary of the NY, USA current state of phylogenetic and systematic 3Social Insect Research Group, Arizona State research on the ants. University, Tempe, AZ, USA Ants Within the Hymenoptera Tree of Ants are the most ubiquitous and ecologically Life dominant insects on the face of our Earth. This is believed to be due in large part to the cooperation Ants belong to the order Hymenoptera, which also allowed by their sociality. At the time of writing, includes wasps and bees. ▶ Eusociality, or true about 13,500 ant species are described and sociality, evolved multiple times within the named, classified into 334 genera that make up order, with ants as by far the most widespread, 17 subfamilies (Fig. 1). This diversity makes the abundant, and species-rich lineage of eusocial ants the world’s by far the most speciose group of animals. Within the Hymenoptera, ants are part eusocial insects, but ants are not only diverse in of the ▶ Aculeata, the clade in which the ovipos- terms of numbers of species. -

The Neotropical Genus Brachymyrmex Mayr, 1868 (Hymenoptera: Formicidae) in Argentina
Acta Zoológica Mexicana (n.s.) 20(1): 273-285 (2004) THE NEOTROPICAL GENUS BRACHYMYRMEX MAYR, 1868 (HYMENOPTERA: FORMICIDAE) IN ARGENTINA. REDESCRIPTION OF THE TYPE SPECIES, B. PATAGONICUS MAYR,1868; B. BRUCHI FOREL, 1912 AND B. OCULATUS SANTSCHI, 1919 Estela M. QUIRÁN1, Juan J. MARTÍNEZ 1 y Axel O. BACHMANN 2 1 Facultad de Ciencias Exactas y Naturales, UNLPam, Uruguay 151, 6300 Santa Rosa, La Pampa, ARGENTINA [email protected] ; [email protected] 2 Facultad de Ciencias Exactas y Naturales, UBA. Pabellón II – 4º Piso. Ciudad Universitaria. 1428 Capital Federal, ARGENTINA [email protected] RESUMEN El género Brachymyrmex es originario de la Región Neotropical donde incluye aproximadamente 30 especies, de las cuales 26 se han citado para la Argentina. Fue establecido para B. patagonicus (Mayr 1868). Santschi (1923) hizo la única revisión sistemática completa del género existente hasta la fecha, pero restan ambigüedades en la identificación de algunas especies. En este trabajo se redescriben la especie tipo del género, B. patagonicus, como asimismo B. bruchi y B. oculatus. Se acompañan las descripciones de las diferentes castas (obrera, reina y macho) con ilustraciones de los principales caracteres morfológicos utilizados en la identificación. Se designa lectotipo para B. oculatus. El material estudiado pertenece al Museo Argentino de Ciencias Naturales “B. Rivadavia”, al Naturhistorisches Museum Wien, y al Museo de La Plata. Palabras Clave: Brachymyrmex, sistemática, taxonomía, diagnosis. ABSTRACT Of the 30 Neotropical species of Brachymyrmex, 26 live in Argentina. This genus was originally proposed by G. Mayr to for the Argentinian B. patagonicus. Santschi published the only systematic revision presently available, but several species cannot be yet easily separated or named. -

Manipulating Tropical Fire Ants to Reduce the Coffee Berry Borer
Ecological Entomology (2014), DOI: 10.1111/een.12139 Manipulating tropical fire ants to reduce the coffee berry borer WARING TRIBLE1 and R O N C A R R O L L 2 1The Rockefeller University, New York , New York, U.S.A. and 2Odum School of Ecology, University of Georgia, Athens, Georgia, U.S.A. Abstract. 1. The coffee berry borer Hypothenemus hampei (Coleoptera, Curculion- idae) (Ferrari) is the most important pest of coffee production worldwide. 2. The hypothesis that the tropical !re ant, Solenopsis geminata Westwood, indirectly protects the coffee berry borer by suppressing other ant species that are the coffee berry borer’s primary predators was tested. 3. It was found that removing S. geminata from coffee plots signi!cantly increased the disappearance of adult coffee berry borer beetles from coffee berries compared with control plots. An average of 6% of beetles disappeared from plots with S. geminata whereas 23% of beetles disappeared from plots from which S. geminata was removed. This pattern was observed on two shade coffee farms with marked differences in ant species composition, one in the rainforest in central Costa Rica and one in the cloudforest in northwest Costa Rica. 4. If the results of this small-scale study can be replicated on the farm level, then S. geminata suppression may represent a new management technique for the coffee berry borer throughout Central and South America. Key words. Biological control, coffee berry borer, !re ant, Formicidae, Hypothenemus hampei, Solenopsis. Introduction coffee berries in the soil, and soil-dwelling ants may thus play an important role in reducing dry season coffee berry borer Coffee is the second most important commodity in the world by populations and minimising future outbreaks (Armbrecht & value, and the coffee berry borer is a beetle that constitutes the Gallego, 2007; Jaramillo et al.,2007;Chapmanet al.,2009; most important pest in coffee production worldwide (Damon, Vélez et al., 2006).