Testing Peatland Water-Table Depth Transfer Functions Using High-Resolution Hydrological Monitoring Data
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Natural Heritage Program List of Rare Plant Species of North Carolina 2012
Natural Heritage Program List of Rare Plant Species of North Carolina 2012 Edited by Laura E. Gadd, Botanist John T. Finnegan, Information Systems Manager North Carolina Natural Heritage Program Office of Conservation, Planning, and Community Affairs N.C. Department of Environment and Natural Resources 1601 MSC, Raleigh, NC 27699-1601 Natural Heritage Program List of Rare Plant Species of North Carolina 2012 Edited by Laura E. Gadd, Botanist John T. Finnegan, Information Systems Manager North Carolina Natural Heritage Program Office of Conservation, Planning, and Community Affairs N.C. Department of Environment and Natural Resources 1601 MSC, Raleigh, NC 27699-1601 www.ncnhp.org NATURAL HERITAGE PROGRAM LIST OF THE RARE PLANTS OF NORTH CAROLINA 2012 Edition Edited by Laura E. Gadd, Botanist and John Finnegan, Information Systems Manager North Carolina Natural Heritage Program, Office of Conservation, Planning, and Community Affairs Department of Environment and Natural Resources, 1601 MSC, Raleigh, NC 27699-1601 www.ncnhp.org Table of Contents LIST FORMAT ......................................................................................................................................................................... 3 NORTH CAROLINA RARE PLANT LIST ......................................................................................................................... 10 NORTH CAROLINA PLANT WATCH LIST ..................................................................................................................... 71 Watch Category -

A-231 Forested Peatland Management in Southeast
15TH INTERNATIONAL PEAT CONGRESS 2016 Abstract No: A-231 FORESTED PEATLAND MANAGEMENT IN SOUTHEAST VIRGINIA AND NORTHEAST NORTH CAROLINA, USA Frederic C. Wurster1, Sara Ward2, and Christine Pickens3 1 U.S. Fish and Wildlife Service, Great Dismal Swamp National Wildlife Refuge, Suffolk, VA, USA 2 U.S. Fish and Wildlife Service, Raleigh Field Office, Raleigh, NC, USA 3 The Nature Conservancy, Kill Devil Hills, NC, USA *Corresponding author: [email protected] SUMMARY The U.S. Fish and Wildlife Service manage 130,000 ha of forested peatlands in southeast Virginia and northeast North Carolina, U.S.A. at three national wildlife refuges: Pocosin Lakes, Alligator River, and Great Dismal Swamp. The refuges contain extensive networks of roads and drainage canals; the legacy of past logging and farming activities. Canals have contributed to peat subsidence and facilitated the conversion of wetland forest communities to upland forest communities. During drought periods, drained peat is prone to wildfires that can burn for several months and pose health risks to neighboring communities. Refuge managers are working to restore historic forest communities and stop peat loss through hydrologic restoration, forest management, and fire management. Following four separate severe peat fires on refuges in 2008 and 2011, emphasis has been placed on improving water control capability to support habitat restoration and fire suppression activities. Keywords: Pocosin, forested peatland, peatland restoration, controlled drainage, national wildlife refuge INTRODUCTION The South Atlantic Coastal plain of the United States is home to extensive forested wetlands on peat soils known as pocosins (Richardson, 1991). Also described as evergreen (or southeastern) shrub bogs, the pocosin vegetation community consists of an open-pine canopy underlain by a dense shrub layer. -

Pocosin Breeding Bird Fauna
P0c0sinbreeding bird fauna Widely recognized for their unique botanical nature, little study has been done on pocosin avifauna. David S. Lee systemsand/or adjacent to estuarine from an Algonquin Indian term systems.Such mixed areas often pro- HE"poquosin"WORD POCOSIN and is oneORIGINATED of the few vide a rich mosaic of wetland habitats Algonquin words used by European involvingbroad zonesof transitionand settlers. Pocosin habitats are defined complex successionalpatterns. Exten- vath difficulty since considerablecon- siveareas called pocosins by naturalists fusion persistsin the use of the term. and environmentalistsare, in fact, often Nevertheless,it is obviouslycritical to composedof swamp forest, hardwood definethese habits accuratelyif mean- or evergreenshrub bog have been used forest, and marshes.Although there lngful habitat comparisonsare to be to describepocosin vegetation types. seemsto be no comprehensivebotanical made. Tooker (1899) provided detailed The term "bay" is particularly con- definition of pocosinmost researchers discussionon the origin, meaning,and fusing becauseit refers to a number agreethat pocosinsare characterizedby usageof the word, and somerecent au- of moderately advanced successional Pond Pine and denseevergreen shrub thors have credited him as translating stages of Southeastern wetlands that vegetationgrowing on deep peat or pocosin as "swamp on a hill." While support several speciesof bay trees sandypeat soilswith protractedhydro- th•s is an interestinginterpretation of (Sweetbay,Redbay, and Loblolly-bay), periods. the word, it was never defined as such whereasthe term Carolina bay, partly Although pocosins are currently by Tooker.In tracingout the earlyusage named for the presenceof bay trees,re- communities of major interest to en- of the term, both.by Native Americans fers to elliptical depressionsthat may vironmentalistsand are widely recog- and early settlers,Tooker found that support pocosin vegetation. -

WRRI Project Nos. 5007Wand 50071 August 1984
(N. C. CEIP Report No. 41) HYDROLOG IC AND WATER QUALITY IMPACTS OF PEAT MINING IN NORTH CAROLINA -L n Jd- J. D. R. W. R. G. Gregory, Skaggs, J1 ~roadheady~ R. H. ~ulbreath," J. R. Bailey," and T. L. Foutzda' 9: Department of Forestry Department of Biological and Agricultural EngineeringJ,* North Carolina State University Raleigh, NC 27695 The work upon which this publication is based was supported by (1) a Coastal Energy Impact Program grant provided by the North Carolina Coastal Management Program through funds authorized by the Coastal Zone Management Act of 1972, as amended, and administered by the Off ice of Coastal Management, National Oceanic and Atmospheric Administration; and (2) by the North Carolina Agricultural Research Service in cooperation with First Colony Farms, Inc. and Peat Methanol Associates. Project administration was provided by the Water Resources Research ~nstituteof The University of North Carolina. NOAA Grants No. NA-79-AA-D-CZ097 and NA-80-AA-D-CZ149 WRRI Project Nos. 5007Wand 50071 August 1984 ACKNOWLEDGMENTS Major support for this research was provided by The National Oceanic and Atmospheric Administration through The Coastal Energy Impact Program and the North Carolina Office of Coastal Management. The support and assistance of James F. Smith, Coordinator North Carolina Coastal Energy Impact Program is gratefully acknowledged. The study was conducted at First Colony Farms, Creswell, North Carolina. We thank Andy Allen and Steve Barnes whose staff installed the flashboard riser structures, constructed and maintained the stilling ponds and provided other assistance in the field work. Additional support was provided by The North Carolina Agricultural Research Service in the form of faculty time. -

Video: the Riparian Zone
Introduction to Wetlands VIDEO: Freshwater Wetlands Adapted from: Freshwater Wetlands-Life at the Waterworks. Educational Media Corporation / North Carolina State Museum of Natural Sciences. Grade Level: Basic ACADEMIC STANDARDS (ENVIRONMENT & ECOLOGY): 7th Grade 4.1.7.D. Explain and describe characteristics of a wetland. Duration: 40 minutes - Identify specific characteristics of wetland plants and soils. - Recognize the common types of plants and animals. Setting: Classroom - Describe different types of wetlands. - Describe the different functions of a wetland. 4.1.7.E. Describe the impact of watersheds and wetlands on people. Summary: Students watch a video - Explain the impact of watersheds and wetlands in flood control, wildlife about different types of freshwater habitats and pollution abatement. wetlands, their soils, flora, and fauna - Explain the influence of flooding on wetlands. and complete a worksheet. 10th Grade 4.1.10.D. Describe the multiple functions of wetlands. Objectives: Students will become - Describe wetlands in terms of their effects (e. g., habitat, flood, buffer zones, familiar with soils, flora, fauna, and prevention areas, nurseries, food production areas). - Explain how a wetland influences water quality, wildlife and water retention. specific threats and issues related to - Analyze wetlands through their indicators (e. g., soils, plants, hydrology). bogs, marshes, pocosins, savannas, 4.1.10.E. Identify and describe natural and human events on watersheds and river floodplains, and seasonal wetlands. wetlands. - Describe how natural events affect a watershed (e. g., drought, floods). - Identify the effects of humans and human events on watersheds. Vocabulary: 12th Grade Hydric soils, hydrophytic vegetation 4.1.12.D. Analyze the complex and diverse ecosystems of wetlands. -

Volume 32, Issue 26 Virginia Register of Regulations August 22, 2016 3443 PUBLICATION SCHEDULE and DEADLINES
VOL. 32 ISS. 26 PUBLISHED EVERY OTHER WEEK BY THE VIRGINIA CODE COMMISSION AUGUST 22, 2016 VOL TABLE OF CONTENTS Register Information Page ......................................................................................................................................... 3443 Publication Schedule and Deadlines ....................................................................................................................... 3444 Regulations ....................................................................................................................................................................... 3445 1VAC30-105. Regulations Banning Concealed Firearms in Offices Owned or Occupied by Executive Branch Agencies (Proposed) ............................................................................................................................. 3445 2VAC5-685. Regulations Governing Pesticide Applicator Certification under Authority of Virginia Pesticide Control Act (Final) ............................................................................................................................... 3448 6VAC20-230. Regulations Relating to Special Conservator of the Peace (Final) ................................................................. 3455 8VAC20-440. Regulations Governing the Employment of Professional Personnel (Proposed) ............................................ 3457 8VAC20-441. Regulations Governing the Employment of Professional Personnel (Proposed) ............................................ 3457 9VAC25-260. -
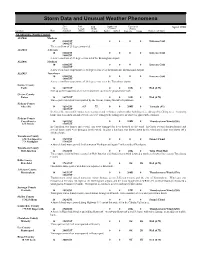
Storm Data and Unusual Weather Phenomena
Storm Data and Unusual Weather Phenomena Time Path Path Number of Estimated April 1996 Local/ Length Width Persons Damage Location Date Standard (Miles) (Yards) Killed Injured Property Crops Character of Storm ALABAMA, North Central ALZ006 Madison 07 0100CST 0 0 0 0 Extreme Cold 1800CST The record low of 29 degrees was tied. ALZ024 Jefferson 10 0100CST 0 0 0 0 Extreme Cold 1800CST A new record low of 29 degrees was set at the Birmingham airport. ALZ006 Madison 10 0100CST 0 0 0 0 Extreme Cold 1800CST A new record low temperature of 30 degrees was set at the Huntsville International Airport. ALZ023 Tuscaloosa 10 0100CST 0 0 0 0 Extreme Cold 1800CST A new record low temperature of 30 degrees was set at the Tuscaloosa airport. Sumter County York 14 1627CST 0 0 10K 0 Hail (0.75) Hail up to three-quarters of an inch in diameter covered the ground near York. Greene County Eutaw 14 1627CST 0 0 10K 0 Hail (0.75) Three-quarter inch hail was reported by the Greene County Sheriff's Department. Pickens County Aliceville 14 1638CST 0.5 75 0 0 200K 0 Tornado (F1) 1642CST In Aliceville, two mobile homes were destroyed and 12 houses and two other buildings were damaged by falling trees. A nursing home roof was taken off and several cars were damaged by falling trees in what was apparently a tornado. Pickens County Carrollton to 14 1642CST 0 0 100K 0 Thunderstorm Wind (G56) 6 N Gordo 1705CST In Carrollton two homes and several cars were damaged by trees downed by the wind. -

Dichotomous Keys and Mapping Codes for Wetland Landscape Position, Landform, Water Flow Path, and Waterbody Type Descriptors: Version 2.0
Cover photo by R.W. Tiner. Dichotomous Keys and Mapping Codes for Wetland Landscape Position, Landform, Water Flow Path, and Waterbody Type Descriptors: Version 2.0 Ralph W. Tiner Regional Wetland Coordinator U.S. Fish and Wildlife Service National Wetlands Inventory Project Northeast Region 300 Westgate Center Drive Hadley, MA 01035 August 2011 This report should be cited as: Tiner, R.W. 2011. Dichotomous Keys and Mapping Codes for Wetland Landscape Position, Landform, Water Flow Path, and Waterbody Type Descriptors: Version 2.0. U.S. Fish and Wildlife Service, National Wetlands Inventory Program, Northeast Region, Hadley, MA. 51 pp. Table of Contents Page Section 1. Introduction 1 Need for New Descriptors 1 Background on Development of Keys 2 Use of the Keys 3 Uses of Enhanced Digital Database 3 Organization of this Report 4 Section 2. Wetland Keys 5 Key A-1: Key to Wetland Landscape Position 8 Key B-1: Key to Inland Landforms 13 Key C-1: Key to Coastal Landforms 16 Key D-1: Key to Water Flow Paths 18 Section 3. Waterbody Keys 22 Key A-2: Key to Major Waterbody Type 23 Key B-2: Key to River/Stream Gradient and Other Modifiers Key 25 Key C-2: Key to Lakes 26 Key D-2: Key to Oceans and Marine Embayments 27 Key E-2: Key to Estuaries 28 Key F-2: Key to Water Flow Paths 29 Key G-2: Key to Estuarine Hydrologic Circulation Types 30 Section 4. Coding System for LLWW Descriptors 31 Codes for Wetlands 31 Landscape Position 31 Lotic Gradient 31 Lentic Type 32 Estuary Type 32 Inland Landform 33 Coastal Landform 35 Water Flow Path 36 Other Modifiers 36 Codes for Waterbodies (Deepwater Habitats and Ponds) 38 Waterbody Type 38 Water Flow Path 42 Estuarine Hydrologic Circulation Type 42 Other Modifiers 43 Section 5. -

Environmental and Anthropogenic Controls Over Bacterial Communities in Wetland Soils
Environmental and anthropogenic controls over bacterial communities in wetland soils Wyatt H. Hartmana,1, Curtis J. Richardsona, Rytas Vilgalysb, and Gregory L. Brulandc aDuke University Wetland Center, Nicholas School of the Environment, Duke University, Durham, NC 27708; bBiology Department, Duke University, Durham, NC 27708; and cDepartment of Natural Resources and Environmental Management, University of Hawai’i at Manoa, Honolulu, HI 96822 Communicated by William L. Chameides, Duke University, Durham, NC, September 29, 2008 (received for review October 12, 2007) Soil bacteria regulate wetland biogeochemical processes, yet little specific phylogenetic groups of microbes affected by restoration is known about controls over their distribution and abundance. have not yet been determined in either of these systems. Eu- Bacteria in North Carolina swamps and bogs differ greatly from trophication and productivity gradients appear to be the primary Florida Everglades fens, where communities studied were unex- determinants of microbial community composition in freshwater pectedly similar along a nutrient enrichment gradient. Bacterial aquatic ecosystems (17, 18). To capture the range of likely composition and diversity corresponded strongly with soil pH, land controls over uncultured bacterial communities across freshwa- use, and restoration status, but less to nutrient concentrations, and ter wetland types, we chose sites representing a range of soil not with wetland type or soil carbon. Surprisingly, wetland resto- chemistry and land uses, including reference wetlands, agricul- ration decreased bacterial diversity, a response opposite to that in tural and restored wetlands, and sites along a nutrient enrich- terrestrial ecosystems. Community level patterns were underlain ment gradient. by responses of a few taxa, especially the Acidobacteria and The sites we selected represented a range of land uses Proteobacteria, suggesting promise for bacterial indicators of res- encompassing natural, disturbed, and restored conditions across toration and trophic status. -

Restoration and Enhancement Site Mapping for the North Carolina Coastal Plain
NCDENR Division of Coastal Management GIS Data Guidance Document GIS Potential Restoration and Enhancement Site Mapping for the North Carolina Coastal Plain Background Much of the North Carolina Coastal Plain is occupied by wetlands, which, in many areas, comprise 50 percent or more of the landscape. These wetlands are of great ecological importance, in part because they occupy so much of the landscape and are a significant component of virtually all coastal ecosystems. They are also important because of their relationships to coastal water quality, estuarine productivity, wildlife habitat, and the overall character of the coastal area. Historically, approximately 50 percent of the original wetlands of the coastal area have been drained and converted to other land uses (Hefner and Brown, 1985; Dahl, 1990; DEM, 1991). Increasing human alteration of the landscape continues to threaten the natural functions of wetlands. Alteration of wetlands compromises their capacity to function and, therefore, compromises their value. Recognizing the functions of wetlands and the values of these functions to society, many natural resource permitting and management agencies have placed a high priority on the protection and restoration of wetlands and riparian areas. An increasing number of state and federal agencies have developed river basin or watershed level wetland and riparian area restoration plans. Environmental organizations are involved in a wide variety of projects emphasizing wetland and riparian area restoration. Although many of the philosophical and technical issues surrounding ecological restoration have yet to be resolved, it is increasingly practiced and often mandated as part of environmental regulatory programs. Unavoidable fill or discharge in wetlands is often accompanied by a regulatory requirement to compensate for the resulting losses in wetland functions. -

PMSD Wetlands Curriculum
WWEETTLLAANNDDSS CCUURRRRIICCUULLUUMM POCONO MOUNTAIN SCHOOL DISTRICT ACKNOWLEDGMENTS This curriculum was funded through a grant from Pennsylvania’s Growing Greener Program administered through the Pennsylvania Department of Environmental Protection. The grant was awarded to the Tobyhanna Creek/Tunkhannock Creek Watershed Association to support ongoing watershed resource protection. The views herein are those of the author(s) and do not necessarily reflect the views of the Pennsylvania Department of Environmental Protection. Special appreciation is extended to the Pocono Mountain School District, especially Mr. Thomas Knorr, Science Supervisor, and Dr. David Krauser, Superintendent of Schools for their support of and commitment to this project. Project partners responsible for this project include: a Pocono Mountain School District a Tobyhanna Creek/Tunkhannock Creek Watershed Association a Pennsylvania Department of Environmental Protection a The Nature Conservancy Science Office a Monroe County Planning Commission a F. X. Browne, Inc. TABLE OF CONTENTS PAGE INTRODUCTION 1 WETLANDS – WHAT ARE THY EXACTLY? 2 WETLAND TYPES 4 WETLANDS CLASSIFICATION 7 PENNSYLVANIA WETLANDS 13 THE THREE H’S: HYDROLOGY, HYDRIC SOILS, AND HYDROPHYTES 15 ARE WETLANDS IMPORTANT? 19 THE FUNCTIONS AND VALUES OF WETLANDS 20 WETLANDS PROTECTION 22 DETERMINING THE WETLANDS BOUNDARY 26 TOBYHANNA CREEK/TUNKANNOCK CREEK WATERSHED ASSOCIATION 28 REFERENCES 30 HOMEWORK ASSIGNMENT #1 – TEST YOUR WETLAND IQ: WHAT DO YOU KNOW ABOUT WETLANDS????? HOMEWORK ASSIGNMENT #2 – USING PLANT IDENTIFICATION GUIDES HOMEWORK ASSIGNMENT #3 – POSITION PAPER FIELD STUDY #1 – FORESTED WETLANDS FIELD STUDY #2 – THE BOG APPENDIX A – PLANT PHOTOGRAPHS – BOG SITE APPENDIX B – PLANT KEYS – BOG SITE APPENDIX C – WETLANDS DELINEATION FIELD DATA SHEETS APPENDIX D – BOG SITE FIELD DATA SHEETS APPENDIX E – GLOSSARY OF TERMS INTRODUCTION Wetlands have not always been a subject of study. -

Pocosin Lakes National Wildlife Refuge
Pocosin Lakes National Wildlife Refuge Comprehensive Conservation Plan U.S. Department of the Interior Fish and Wildlife Service Southeast Region September 2007 COMPREHENSIVE CONSERVATION PLAN POCOSIN LAKES NATIONAL WILDLIFE REFUGE U.S. Department of the Interior Fish and Wildlife Service Southeast Region Atlanta, Georgia 30345 November 2007 TABLE OF CONTENTS COMPREHENSIVE CONSERVATION PLAN EXECUTIVE SUMMARY ....................................................................................................................... 1 I. BACKGROUND ................................................................................................................................. 1 Introduction ................................................................................................................................... 1 Purpose and Need For The Plan .................................................................................................. 1 U.S. Fish and Wildlife Service ...................................................................................................... 2 National Wildlife Refuge System .................................................................................................. 2 Legal Policy Context ..................................................................................................................... 3 National Conservation Plans and Initiatives .................................................................................3 Relationship To State Partners .....................................................................................................4