Analytical Method Development & Validation for Simultaneous
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

New Developments in Prokinetic Therapy for Gastric Motility Disorders
REVIEW published: 24 August 2021 doi: 10.3389/fphar.2021.711500 New Developments in Prokinetic Therapy for Gastric Motility Disorders Michael Camilleri* and Jessica Atieh Clinical Enteric Neuroscience Translational and Epidemiological Research (CENTER), Division of Gastroenterology and Hepatology, Mayo Clinic, Rochester, MN, United States Prokinetic agents amplify and coordinate the gastrointestinal muscular contractions to facilitate the transit of intra-luminal content. Following the institution of dietary recommendations, prokinetics are the first medications whose goal is to improve gastric emptying and relieve symptoms of gastroparesis. The recommended use of metoclopramide, the only currently approved medication for gastroparesis in the United States, is for a duration of less than 3 months, due to the risk of reversible or irreversible extrapyramidal tremors. Domperidone, a dopamine D2 receptor antagonist, is available for prescription through the FDA’s program for Expanded Access to Investigational Drugs. Macrolides are used off label and are associated with tachyphylaxis and variable duration of efficacy. Aprepitant relieves some symptoms of gastroparesis. There are newer agents in the pipeline targeting diverse gastric (fundic, antral and pyloric) motor functions, including novel serotonergic 5-HT4 agonists, dopaminergic D2/3 antagonists, neurokinin NK1 antagonists, and ghrelin agonist. Novel Edited by: targets with potential to improve gastric motor functions include the pylorus, macrophage/ Jan Tack, inflammatory function, oxidative -

Prokinetics and Ghrelin for the Management of Cancer Cachexia Syndrome
85 Review Article Prokinetics and ghrelin for the management of cancer cachexia syndrome Jimi S. Malik, Sriram Yennurajalingam Department of Palliative Care, Rehabilitation and Integrative Medicine, The University of Texas MD Anderson Cancer Center, Houston, TX, USA Contributions: (I) Conception and design: All authors; (II) Administrative support: S Yennurajalingam; (III) Provision of study materials or patients: S Yennurajalingam; (IV) Collection and assembly of data: All authors; (V) Data analysis and interpretation: All authors; (VI) Manuscript writing: All authors; (VII) Final approval of manuscript: All authors. Correspondence to: Sriram Yennurajalingam, MD. Department of Palliative Care, Rehabilitation, and Integrative Medicine, The University of Texas MD Anderson Cancer Center, 1515 Holcombe Blvd., Unit 1414, Houston, TX 77030, USA. Email: [email protected]. Abstract: Cancer cachexia (CC) is one of the most distressing syndromes for both patients and their families. CC can have an impact on patient reported quality of life and overall survival. It is often associated with symptoms such as fatigue, depressed mood, early satiety, and anorexia. Prokinetic agents have been found to improve chronic nausea and early satiety associated with CC. Among the prokinetic agents, metoclopramide is one of the best studied medications. The role of the other prokinetic agents, such as domperidone, erythromycin, haloperidol, levosulpiride, tegaserod, cisapride, mosapride, renzapride, and prucalopride is unclear for use in cachectic cancer patients due to their side effect profile and limited efficacy studies in cancer patients. There has been an increased interest in the use of ghrelin-receptor agonists for the treatment of CC. Anamorelin HCl is a highly selective, novel ghrelin receptor agonist. -

International Journal of Medicine and Pharmaceutical Research
Wayal Sunil Anil et al, IJMPR, 2019, 7(6): 180-183 CODEN (USA): IJCPNH | ISSN: 2321-2624 International Journal of Medicine and Pharmaceutical Research Journal Home Page: www.pharmaresearchlibrary.com/ijmpr R E S E A R C H A R T I C L E Formulation and In-vitro Evaluation of Alosetron Oral Thin Films Wayal Sunil Anil1, G.S. Valluri2, Gampa Vijay Kumar3 Department of Pharmacy, KGR Institute of Technology and Management, Rampally, Kesara, Medchal, Telangana, India. A B S T R A C T Alosetron, is a 5-HT3 antagonist used for the management of severe diarrhea-predominant irritable bowel syndrome. In present study oral thin films of Alosetron were developed to have a faster on set of action. The oralthin films were developed by using polymers Guar gum, Pullulan and PVP K30.Oral thin films were prepared by employing solvent casting method. Propylene glycol was selected as permeation enhancer and plasticizer. Drug excipient compatibility studies were carried out by using FTIR, and it was observed that there were no interactions. Formulations were prepared with the varying concentrations polymers ranging from F1-F9, and all the formulations were evaluated for various physical parameters Physical appearance, Weight variation, Thickness, Folding endurance, Tensile strength, Drug content, Moisture uptake, Moisture content and all the results were found to be were found to be within the pharmacopeial limits, in-vitro drug release studies by using USP dissolution Apparatus Type II. Among all the 9 formulations F4 formulation which contain Pullulan 10mg and shown 97.06% cumulative drug release within 30 min. -

Comprehensive Pgx Report for 1 / 31 Examples of Different Levels of Evidence for Pgx Snps
Comprehensive PGx report for PERSONAL DETAILS Advanced Diagnostics Laboratory LLC CLIA:31D2149403 Phone: Fax: PATIENT DOB Address: 1030 North Kings Highway Suite 304 Cherry Hill, NJ 08034 GENDER FEMALE Website: http://advanceddiagnosticslaboratory.com/ SPECIMEN TYPE Oral Fluid LABORATORY INFORMATION ORDERING PHYSICIAN ACCESSION NUMBER 100344 FACILITY COLLECTION DATE 08/10/2020 RECEIVED DATE 08/14/2020 REPORT GENERATED 09/08/2020 LABORATORY DIRECTOR Dr. Jeanine Chiaffarano Current Patient Medication Clonidine (Catapres, Kapvay) The personalized pharmacogenomics profile of this patient reveals intermediate CYP2D6-mediated metabolism, extensive CYP1A2-mediated metabolism, and extensive CYP3A5-mediated metabolism. For further details, please find supporting evidence in this report or on websites such as www.pharmgkb.org or www.fda.gov. Losartan (Cozaar) The personalized pharmacogenomics profile of this patient reveals extensive CYP2C9-mediated metabolism, extensive CYP3A4-mediated metabolism, and extensive CYP3A5-mediated metabolism. For further details, please find supporting evidence in this report or on websites such as www.pharmgkb.org or www.fda.gov. Diltiazem (Cardizem, Tiazac) The personalized pharmacogenomics profile of this patient reveals extensive CYP3A4-mediated metabolism, intermediate CYP2C19-mediated metabolism, and extensive CYP3A5- mediated metabolism. For further details, please find supporting evidence in this report or on websites such as www.pharmgkb.org or www.fda.gov. Labetalol (Normodyne, Trandate) The personalized pharmacogenomics profile of this patient reveals intermediate CYP2D6-mediated metabolism, and intermediate CYP2C19-mediated metabolism. For further details, please find supporting evidence in this report or on websites such as www.pharmgkb.org or www.fda.gov. Mycophenolate mofetil (Myfortic, CellCept) The personalized pharmacogenomics profile of this patient reveals extensive CYP3A4-mediated metabolism, extensive CYP3A5-mediated metabolism, and extensive CYP2C8-mediated metabolism. -

Classification of Medicinal Drugs and Driving: Co-Ordination and Synthesis Report
Project No. TREN-05-FP6TR-S07.61320-518404-DRUID DRUID Driving under the Influence of Drugs, Alcohol and Medicines Integrated Project 1.6. Sustainable Development, Global Change and Ecosystem 1.6.2: Sustainable Surface Transport 6th Framework Programme Deliverable 4.4.1 Classification of medicinal drugs and driving: Co-ordination and synthesis report. Due date of deliverable: 21.07.2011 Actual submission date: 21.07.2011 Revision date: 21.07.2011 Start date of project: 15.10.2006 Duration: 48 months Organisation name of lead contractor for this deliverable: UVA Revision 0.0 Project co-funded by the European Commission within the Sixth Framework Programme (2002-2006) Dissemination Level PU Public PP Restricted to other programme participants (including the Commission x Services) RE Restricted to a group specified by the consortium (including the Commission Services) CO Confidential, only for members of the consortium (including the Commission Services) DRUID 6th Framework Programme Deliverable D.4.4.1 Classification of medicinal drugs and driving: Co-ordination and synthesis report. Page 1 of 243 Classification of medicinal drugs and driving: Co-ordination and synthesis report. Authors Trinidad Gómez-Talegón, Inmaculada Fierro, M. Carmen Del Río, F. Javier Álvarez (UVa, University of Valladolid, Spain) Partners - Silvia Ravera, Susana Monteiro, Han de Gier (RUGPha, University of Groningen, the Netherlands) - Gertrude Van der Linden, Sara-Ann Legrand, Kristof Pil, Alain Verstraete (UGent, Ghent University, Belgium) - Michel Mallaret, Charles Mercier-Guyon, Isabelle Mercier-Guyon (UGren, University of Grenoble, Centre Regional de Pharmacovigilance, France) - Katerina Touliou (CERT-HIT, Centre for Research and Technology Hellas, Greece) - Michael Hei βing (BASt, Bundesanstalt für Straßenwesen, Germany). -

CAS Number Index
2334 CAS Number Index CAS # Page Name CAS # Page Name CAS # Page Name 50-00-0 905 Formaldehyde 56-81-5 967 Glycerol 61-90-5 1135 Leucine 50-02-2 596 Dexamethasone 56-85-9 963 Glutamine 62-44-2 1640 Phenacetin 50-06-6 1654 Phenobarbital 57-00-1 514 Creatine 62-46-4 1166 α-Lipoic acid 50-11-3 1288 Metharbital 57-22-7 2229 Vincristine 62-53-3 131 Aniline 50-12-4 1245 Mephenytoin 57-24-9 1950 Strychnine 62-73-7 626 Dichlorvos 50-23-7 1017 Hydrocortisone 57-27-2 1428 Morphine 63-05-8 127 Androstenedione 50-24-8 1739 Prednisolone 57-41-0 1672 Phenytoin 63-25-2 335 Carbaryl 50-29-3 569 DDT 57-42-1 1239 Meperidine 63-75-2 142 Arecoline 50-33-9 1666 Phenylbutazone 57-43-2 108 Amobarbital 64-04-0 1648 Phenethylamine 50-34-0 1770 Propantheline bromide 57-44-3 191 Barbital 64-13-1 1308 p-Methoxyamphetamine 50-35-1 2054 Thalidomide 57-47-6 1683 Physostigmine 64-17-5 784 Ethanol 50-36-2 497 Cocaine 57-53-4 1249 Meprobamate 64-18-6 909 Formic acid 50-37-3 1197 Lysergic acid diethylamide 57-55-6 1782 Propylene glycol 64-77-7 2104 Tolbutamide 50-44-2 1253 6-Mercaptopurine 57-66-9 1751 Probenecid 64-86-8 506 Colchicine 50-47-5 589 Desipramine 57-74-9 398 Chlordane 65-23-6 1802 Pyridoxine 50-48-6 103 Amitriptyline 57-92-1 1947 Streptomycin 65-29-2 931 Gallamine 50-49-7 1053 Imipramine 57-94-3 2179 Tubocurarine chloride 65-45-2 1888 Salicylamide 50-52-2 2071 Thioridazine 57-96-5 1966 Sulfinpyrazone 65-49-6 98 p-Aminosalicylic acid 50-53-3 426 Chlorpromazine 58-00-4 138 Apomorphine 66-76-2 632 Dicumarol 50-55-5 1841 Reserpine 58-05-9 1136 Leucovorin 66-79-5 -
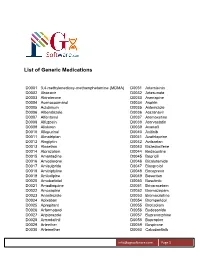
List of Generic Medications
List of Generic Medications D0001 3,4-methylenedioxy-methamphetamine (MDMA) D0031 Artemisinin D0002 Abacavir D0032 Artesunate D0003 Abiraterone D0033 Asenapine D0004 Acenocoumarol D0034 Aspirin D0005 Aclidinium D0035 Astemizole D0006 Albendazole D0036 Atazanavir D0007 Alfentanyl D0037 Atomoxetine D0008 Alfuzosin D0038 Atorvastatin D0009 Aliskiren D0039 Avanafil D0010 Allopurinol D0040 Axitinib D0011 Almotriptan D0041 Azathioprine D0012 Alogliptin D0042 Azilsartan D0013 Alosetron D0043 Bazedoxifene D0014 Alprazolam D0044 Bedaquiline D0015 Amantadine D0045 Bepridil D0016 Amiodarone D0046 Bicalutamide D0017 Amisulpride D0047 Bisoprolol D0018 Amitriptyline D0048 Boceprevir D0019 Amlodipine D0049 Bosentan D0020 Amobarbital D0050 Bosutinib D0021 Amodiaquine D0051 Brivaracetam D0022 Amoxapine D0052 Bromazepam D0023 Anastrozole D0053 Bromocriptine D0024 Apixaban D0054 Bromperidol D0025 Aprepitant D0055 Brotizolam D0026 Arformoterol D0056 Budesonide D0027 Aripiprazole D0057 Buprenorphine D0028 Armodafinil D0058 Bupropion D0029 Arteether D0059 Buspirone D0030 Artemether D0060 Cabozantinib [email protected] Page 1 List of Generic Medications D0061 Canagliflozin D0091 Clofarabine D0062 Cannabidiol (CBD) D0092 Clofibrate D0063 Cannabinol (CBN) D0093 Clomipramine D0064 Captopril D0094 Clonazepam D0065 Carbamazepine D0095 Clonidine D0066 Carisoprodol D0096 Clopidogrel D0067 Carmustine D0097 Clorazepate D0068 Carvedilol D0098 Clozapine D0069 Celecoxib D0099 Cocaine D0070 Ceritinib D0100 Codeine D0071 Cerivastatin D0101 Colchicine D0072 Cetirizine -

Gastroparesis: 2014
GASTROINTESTINAL MOTILITY AND FUNCTIONAL BOWEL DISORDERS, SERIES #1 Richard W. McCallum, MD, FACP, FRACP (Aust), FACG Status of Pharmacologic Management of Gastroparesis: 2014 Richard W. McCallum Joseph Sunny, Jr. Gastroparesis is characterized by delayed gastric emptying without mechanical obstruction of the gastric outlet or small intestine. The main etiologies are diabetes, idiopathic and post- gastric and esophageal surgical settings. The management of gastroparesis is challenging due to a limited number of medications and patients often have symptoms, which are refractory to available medications. This article reviews current treatment options for gastroparesis including adverse events and limitations as well as future directions in pharmacologic research. INTRODUCTION astroparesis is a syndrome characterized by documented gastroparesis are increasing.2 Physicians delayed emptying of gastric contents without have both medical and surgical approaches for these Gmechanical obstruction of the stomach, pylorus or patients (See Figure 1). Medical therapy includes both small bowel. Patients can present with nausea, vomiting, prokinetics and antiemetics (See Table 1 and Table 2). postprandial fullness, early satiety, pressure, fullness The gastroparesis population will grow as diabetes and abdominal distension. In addition, abdominal pain increases and new therapies will be required. What located in the epigastrium, and distinguished from the do we know about the size of the gastroparetic term discomfort, is increasingly being recognized population? According to a study from the Mayo Clinic as an important symptom. The main etiologies of group surveying Olmsted County in Minnesota, the gastroparesis are diabetes, idiopathic, and post gastric risk of gastroparesis in Type 1 diabetes mellitus was and esophageal surgeries.1 Hospitalizations from significantly greater than for Type 2. -

A Four-Country Comparison of Healthcare Systems, Implementation
Neurogastroenterology & Motility Neurogastroenterol Motil (2014) 26, 1368–1385 doi: 10.1111/nmo.12402 REVIEW ARTICLE A four-country comparison of healthcare systems, implementation of diagnostic criteria, and treatment availability for functional gastrointestinal disorders A report of the Rome Foundation Working Team on cross-cultural, multinational research M. SCHMULSON,* E. CORAZZIARI,† U. C. GHOSHAL,‡ S.-J. MYUNG,§ C. D. GERSON,¶ E. M. M. QUIGLEY,** K.-A. GWEE†† & A. D. SPERBER‡‡ *Laboratorio de Hıgado, Pancreas y Motilidad (HIPAM)-Department of Experimental Medicine, Faculty of Medicine-Universidad Nacional Autonoma de Mexico (UNAM). Hospital General de Mexico, Mexico City, Mexico †Gastroenterologia A, Department of Internal Medicine and Medical Specialties, University La Sapienza, Rome, Italy ‡Department of Gastroenterology, Sanjay Gandhi Postgraduate Institute of Medical Science, Lucknow, India §Department of Gastroenterology, University of Ulsan College of Medicine, Asan Medical Center, Seoul, Korea ¶Division of Gastroenterology, Mount Sinai School of Medicine, New York, NY, USA **Division of Gastroenterology and Hepatology, Houston Methodist Hospital and Weill Cornell Medical College, Houston, TX, USA ††Department of Medicine, Yong Loo Lin School of Medicine, National University of Singapore, Singapore, Singapore ‡‡Faculty of Health Sciences, Ben-Gurion University of the Negev, Beer-Sheva, Israel Key Messages • This report identified seven key issues related to healthcare provision that may impact how patients with FGIDs are investigated, diagnosed and managed. • Variations in healthcare provision around the world in patients with FGIDs have not been reviewed. • We compared four countries that are geographically and culturally diverse, and exhibit differences in the healthcare coverage provided to their population: Italy, South Korea, India and Mexico. • Since there is a paucity of publications relating to the issues covered in this report, some of the findings are based on the authors’ personal perspectives, press reports and other published sources. -

The Use of Stems in the Selection of International Nonproprietary Names (INN) for Pharmaceutical Substances
WHO/PSM/QSM/2006.3 The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances 2006 Programme on International Nonproprietary Names (INN) Quality Assurance and Safety: Medicines Medicines Policy and Standards The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances FORMER DOCUMENT NUMBER: WHO/PHARM S/NOM 15 © World Health Organization 2006 All rights reserved. Publications of the World Health Organization can be obtained from WHO Press, World Health Organization, 20 Avenue Appia, 1211 Geneva 27, Switzerland (tel.: +41 22 791 3264; fax: +41 22 791 4857; e-mail: [email protected]). Requests for permission to reproduce or translate WHO publications – whether for sale or for noncommercial distribution – should be addressed to WHO Press, at the above address (fax: +41 22 791 4806; e-mail: [email protected]). The designations employed and the presentation of the material in this publication do not imply the expression of any opinion whatsoever on the part of the World Health Organization concerning the legal status of any country, territory, city or area or of its authorities, or concerning the delimitation of its frontiers or boundaries. Dotted lines on maps represent approximate border lines for which there may not yet be full agreement. The mention of specific companies or of certain manufacturers’ products does not imply that they are endorsed or recommended by the World Health Organization in preference to others of a similar nature that are not mentioned. Errors and omissions excepted, the names of proprietary products are distinguished by initial capital letters. -

Patent Application Publication ( 10 ) Pub . No . : US 2019 / 0192440 A1
US 20190192440A1 (19 ) United States (12 ) Patent Application Publication ( 10) Pub . No. : US 2019 /0192440 A1 LI (43 ) Pub . Date : Jun . 27 , 2019 ( 54 ) ORAL DRUG DOSAGE FORM COMPRISING Publication Classification DRUG IN THE FORM OF NANOPARTICLES (51 ) Int . CI. A61K 9 / 20 (2006 .01 ) ( 71 ) Applicant: Triastek , Inc. , Nanjing ( CN ) A61K 9 /00 ( 2006 . 01) A61K 31/ 192 ( 2006 .01 ) (72 ) Inventor : Xiaoling LI , Dublin , CA (US ) A61K 9 / 24 ( 2006 .01 ) ( 52 ) U . S . CI. ( 21 ) Appl. No. : 16 /289 ,499 CPC . .. .. A61K 9 /2031 (2013 . 01 ) ; A61K 9 /0065 ( 22 ) Filed : Feb . 28 , 2019 (2013 .01 ) ; A61K 9 / 209 ( 2013 .01 ) ; A61K 9 /2027 ( 2013 .01 ) ; A61K 31/ 192 ( 2013. 01 ) ; Related U . S . Application Data A61K 9 /2072 ( 2013 .01 ) (63 ) Continuation of application No. 16 /028 ,305 , filed on Jul. 5 , 2018 , now Pat . No . 10 , 258 ,575 , which is a (57 ) ABSTRACT continuation of application No . 15 / 173 ,596 , filed on The present disclosure provides a stable solid pharmaceuti Jun . 3 , 2016 . cal dosage form for oral administration . The dosage form (60 ) Provisional application No . 62 /313 ,092 , filed on Mar. includes a substrate that forms at least one compartment and 24 , 2016 , provisional application No . 62 / 296 , 087 , a drug content loaded into the compartment. The dosage filed on Feb . 17 , 2016 , provisional application No . form is so designed that the active pharmaceutical ingredient 62 / 170, 645 , filed on Jun . 3 , 2015 . of the drug content is released in a controlled manner. Patent Application Publication Jun . 27 , 2019 Sheet 1 of 20 US 2019 /0192440 A1 FIG . -

A Systematic Review of the Effectiveness of Antianxiety and Antidepressive Agents for Functional Dyspepsia
doi: 10.2169/internalmedicine.9099-17 Intern Med 56: 3127-3133, 2017 http://internmed.jp 【 ORIGINAL ARTICLE 】 A Systematic Review of the Effectiveness of Antianxiety and Antidepressive Agents for Functional Dyspepsia Mariko Hojo 1, Akihito Nagahara 2, Daisuke Asaoka 1, Yuji Shimada 2, Hitoshi Sasaki 3, Kohei Matsumoto 1, Tsutomu Takeda 1, Hiroya Ueyama 1, Kenshi Matsumoto 1 and Sumio Watanabe 1 Abstract: Objective Functional dyspepsia (FD) is defined as persistent or recurrent pain or discomfort centered in the upper abdomen without organic disease. Psychosocial factors have been proposed as an important element in the pathophysiology of FD. Therefore, psychotropic agents having antianxiety or antidepressive action are ex- pected to alleviate FD. We previously reported on the treatment of FD using such agents in a systematic re- view, wherein the effectiveness of the agents on FD was suggested, although there were several limitations. We searched for articles on this subject after our systematic review and re-reviewed them systematically. Methods Articles were searched for in MEDLINE from 2003 to 2014 using terms related to antianxiety or antidepressive agents. Clinical studies in which the effectiveness of such agents was clearly stated were se- lected from the retrieved articles. The newly selected and previously selected studies were combined, and sta- tistical analyses were carried out. Results Nine studies were selected. Five of the studies indicated a significant symptomatic improvement us- ing psychotropic drugs. A statistical analysis suggested a significant treatment effect of psychotropic agents having antianxiety or antidepressive action [pooled relative risk (PRR), 0.72; 95% confidence interval (95% CI), 0.52-0.99; p=0.0406] but did not show a significant benefit of treatment with agents having an antide- pressive action alone (PRR, 0.63; 95% CI, 0.38-1.03; p=0.0665).