A Place That Radiates Great Science
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Hitler and Heisenberg
The Wall Street Journal December 27-28, 2014 Book Review “Serving the Reich” by Philip Ball, Chicago, $30, 303 pages Hitler and Heisenberg Werner Heisenberg thought that once Germany had won, the ‘good Germans’ would get rid of the Nazis. Fizzing The museum inside the former basement tavern where in early 1945 German scientists built a small nuclear reactor. Associated Press by Jeremy Bernstein The German scientists who remained in Germany throughout the Nazi period can be divided into three main groups. At one end were the anti-Nazis—people who not only despised the regime but tried to do something about it. They were very few. Two that come to mind were Fritz Strassmann and Max von Laue. Strassmann was a radiochemist—a specialist in radioactive materials—who was blacklisted from university jobs by the Society of German Chemists in 1933 for his anti-Nazi views. He was fortunate to get a half-pay job at the Kaiser Wilhelm Institute for Chemistry, which got some of its funding from industry. He and his wife (they had a 3-year-old son) hid a Jewish friend in their apartment for months. With the radiochemist Otto Hahn, he performed the 1938 experiment in which fission was observed for the first time. Von Laue won the Nobel Prize in physics in 1914. Soon after Einstein published his 1905 paper on relativity, von Laue went to Switzerland to see him. They remained friends for life. Under the Nazis, Einstein’s work was denounced as “Jewish science.” When a number of von Laue’s colleagues agreed to use relativity but attribute it to some Aryan, he refused. -

What Use Is Chemistry?
2 Inspirational chemistry What use is chemistry? Index 1.1 1 sheet This activity is based on a Sunday Times article by Sir Harry Kroto, a Nobel prize winning chemist who discovered a new allotrope of carbon – buckminsterfullerene or ‘bucky balls’. The article appeared on November 28, 2004 and is reproduced overleaf as a background for teachers. The aim is to introduce students to the scope of modern chemistry and the impact that it has on their lives, even in areas that they may not think of as related to chemistry. An alternative exercise for more able students would be to research what was used before chemical scientists had produced a particular new product or material (eg silk or wool stockings before nylon, leather footballs before synthetics, grated carbolic soap before shampoo) and then to write about the difference it would make to their lives if they did not have the modern product. Students will need: ■ Plenty of old magazines and catalogues (Argos catalogues are good as virtually everything in them would not exist without modern chemistry) ■ Large sheets of sugar paper ■ Glue and scissors. It works well if students produce the poster in groups, but then do the written work by themselves. The activity could be set for homework. Inspirational chemistry 3 What use is chemistry? Some years ago I was delighted chemistry-related industries make a to receive an honorary degree £5 billion profit on a £50 billion from Exeter University turnover, the apparent government recognising my contributions to inaction over the looming disaster chemistry – especially the is scarcely credible. -

Goverdhan Mehta Chemistry - a 21St Century Science for Global Sustainability: Is It Future Ready?
Goverdhan Mehta Chemistry - A 21st Century Science for Global Sustainability: Is it future ready? Goverdhan Mehta A ‘selfie’ with theSchool chemical of Chemistry world…… University of Hyderabad National Geophysical Research Institute, CSIR Foundation Day, Sept. 27, 2019 Introducing Chemistry through the Lens of Earth's Systems: What Role Can Systems Thinking Play in Developing Chemically and Environmentally Literate Citizens? J. Kornfeld, S. Stokoe. J. Chemical Education 2019, 96, 2910-2917 A bouquet of ‘matters’ that matter New symbols Passion Sustainability Legacies Ethics & values Responsible Connections Systems Directions Humility Ideas & icons Inspirations Un mélange de beaucoup de choses “Chemistry ought not to be for chemists alone” - Miguel de Unamuno ‘…Life, Universe and Everything’ Chemistry – a source of happiness…. Chem -Connectome ‘...I feel sorry for people who don’t know anything about chemistry. They are missing an important source of happiness....’ - Linus Pauling 1901-1994 S.A. Matlin, G. Mehta, H. Hopf. Chemistry Embraced by All. Science 2015, 347, 1179 Chemistry is in everything. and everything is in it, it is the basis of life, without it we wouldn't exist. Green tea has ~ 200 chemicals Coffee has ~ 1000 chemicals Wine has >1000 chemicals Light cigarette ~ 4000 chemicals Chemistry is ubiquitous/omnipresent Chemistry – Tracing the roots and to the present BCE Art & craft of mixing substances A giant knowledge leap Alchemy to modern science Evidence based science Discipline in a Table - systematization Mendeleev’s Periodic Law ‘Molecularization’ of chemical matter 20th Century A century of evolutionary march of chemistry 20h Century Value added products from almost anything “Utility science” and everything Molecular understanding of life processes and “Core Science” chemical matter Interdisciplinarity in forefront “Integrative Science” Resource stressed planet “Sustainability Science” 21st Century S. -

The Lock-And-Key Analogy in 20Th Century Biochemistry
From: Rebecca Mertens The Construction of Analogy-Based Research Programs The Lock-and-Key Analogy in 20th Century Biochemistry April 2019, 224 p., pb., ill. 34,99 € (DE), 978-3-8376-4442-5 E-Book: PDF: 34,99 € (DE), ISBN 978-3-8394-4442-9 When the German chemist Emil Fischer presented his lock-and-key hypothesis in 1899, his analogy to describe the molecular relationship between enzymes and substrates quickly gained vast influence and provided future generations of scientists with a tool to investigate the relation between chemical structure and biological specificity. Rebecca Mertens explains the appeal of the lock-and-key analogy by its role in model building and in the construction of long-term, cross-generational research programs. She argues that a crucial feature of these research programs, namely ascertaining the continuity of core ideas and concepts, is provided by a certain way of analogy-based modelling. Rebecca Mertens (PhD), born in 1984, is a postdoctoral researcher in the history and philosophy of science at the University of Bielefeld, Germany. She works on the role of analogies, models and forms of comparison in the history of molecular genetics and is a member of the collaborative research program "Practices of ComparisonÚ Ordering and Changing the World". During her graduate and doctoral studies, she was a visiting scholar at the École Normale Supérieure in Paris and a visiting graduate fellow at the Minnesota Center for Philosophy of Science. For further information: www.transcript-verlag.de/en/978-3-8376-4442-5 © 2019 -

Ein Bisher Unbekanntes Zeitzeugnis
N. T. M. 16 (2008) 103–115 0036-6978/08/010103–13 OUND DOI 10.1007/S00048-007-0277-7 © 2008 BIRKHÄUSER VERLAG, BASEL F & OST /L UNDSTÜCK Ein bisher unbekanntes Zeitzeugnis. F Otto Warburgs Tagebuchnotizen von Februar–April 1945 Kärin Nickelsen Warburg’s diary notes February–April 1945 Personal notes in the form of a diary, written by the German cell biologist Otto Warburg in the weeks before the collapse of Germany’s Third Reich (Feb.-April 1945), were found at the end of one of his labo- ratory notebooks, which are preserved in the Archives of the Berlin-Brandenburg Academy of Sciences and Humanities. These notes are unique in form and content: there seem to be no other surviving diary notes of this type. Written from a very personal perspective, they provide a detailed account of how Warburg experienced local events in this decisive and turbulent period. Furthermore, they clearly demonstrate Warburg’s embittered attitude towards some of his long-standing collaborators, whom he suspected of having denounced him to the Nazi authorities. In this paper, Warburg’s notes are fully transcribed and integrated into the historical context. Keywords: Otto Warburg, Kaiser-Wilhelm-Society, National Socialism, diary notes Schlüsselwörter: Otto Warburg, Kaiser-Wilhelm-Gesellschaft, Nationalsozialismus, Tagebuchnotizen | downloaded: 29.9.2021 Otto Warburg (1883–1970) ist in der Wissenschaftsgeschichte vor allem bekannt für seine bahnbrechenden Arbeiten zur Zellatmung, für die ihm 1931 der Nobelpreis verliehen wurde.1 In anderen Kontexten wiederum ist er bekannt als einer der ganz wenigen Wissenschaftler jüdischer Abstam- mung, die während der gesamten Zeit des Nationalsozialismus in Amt und Würden blieben: Nicht nur wurde Warburg nicht deportiert, er blieb bis 1945 Direktor des Kaiser-Wilhelm-Instituts (KWI) für Zellphysiologie in Berlin-Dahlem.2 Unter anderem stand er eigenen Angaben zufolge bis 1937 unter dem persönlichen Schutz von Friedrich Glum, damals Generaldirektor der Kaiser-Wilhelm-Gesellschaft (Werner 1991: 284). -

Chartered Status Charteredeverything You Need Tostatus Know Everything You Need to Know
Chartered Status CharteredEverything you need toStatus know Everything you need to know www.rsc.org/cchem www.rsc.org/cchem ‘The best of any profession is always chartered’ The RSC would like to thank its members (pictured top to bottom) Ben Greener, Pfizer, Elaine Baxter, Procter & Gamble, and Richard Sleeman, Mass Spec Analytical Ltd, for their participation and support . Chartered Status | 1 Contents About chartered status 3 Why become chartered? 3 What skills and experience do I need? 3 The professional attributes for a Chartered Chemist 5 Supporting you throughout the programme yThe Professional Development Programme 5 yThe Direct Programme 7 How to apply 7 Achieving Chartered Scientist status 8 Revalidation 8 The next step 8 Application form 9 2 | Chartered Status ‘Having a professionally recognised qualification will build my external credibility’ Elaine Baxter BSc PhD MRSC Procter & Gamble Elaine Baxter is a Senior Scientist at Procter & Gamble (P&G). Since joining the company, she has had roles in formulation, process and technology development in skin and shaving science. She graduated in 2001, before completing a PhD on synthetic inorganic chemistry of platinum dyes with applications in solar cells. Elaine is currently working towards Chartered Chemist status through the Professional Development Programme. Why do you want to achieve Chartered Chemist status? My role involves science communication with people such as dermatologists, academics and the media; having a professionally recognised qualification will build my external credibility with these professionals. How do you feel the programme has worked for you? Working towards achieving the attributes required for the CChem award has presented me with opportunities to share my industry knowledge and help others. -

Pauling-Linus.Pdf
NATIONAL ACADEMY OF SCIENCES L I N U S C A R L P A U L I N G 1901—1994 A Biographical Memoir by J A C K D. D UNITZ Any opinions expressed in this memoir are those of the author(s) and do not necessarily reflect the views of the National Academy of Sciences. Biographical Memoir COPYRIGHT 1997 NATIONAL ACADEMIES PRESS WASHINGTON D.C. LINUS CARL PAULING February 28, 1901–August 19, 1994 BY JACK D. DUNITZ INUS CARL PAULING was born in Portland, Oregon, on LFebruary 28, 1901, and died at his ranch at Big Sur, California, on August 19, 1994. In 1922 he married Ava Helen Miller (died 1981), who bore him four children: Linus Carl, Peter Jeffress, Linda Helen (Kamb), and Edward Crellin. Pauling is widely considered the greatest chemist of this century. Most scientists create a niche for themselves, an area where they feel secure, but Pauling had an enormously wide range of scientific interests: quantum mechanics, crys- tallography, mineralogy, structural chemistry, anesthesia, immunology, medicine, evolution. In all these fields and especially in the border regions between them, he saw where the problems lay, and, backed by his speedy assimilation of the essential facts and by his prodigious memory, he made distinctive and decisive contributions. He is best known, perhaps, for his insights into chemical bonding, for the discovery of the principal elements of protein secondary structure, the alpha-helix and the beta-sheet, and for the first identification of a molecular disease (sickle-cell ane- mia), but there are a multitude of other important contri- This biographical memoir was prepared for publication by both The Royal Society of London and the National Academy of Sciences of the United States of America. -

Basic Research in the Max Planck Society Science Policy in the Federal Republic of Germany, 1945–1970
Chapter 5 Basic Research in the Max Planck Society Science Policy in the Federal Republic of Germany, 1945–1970 Carola Sachse ላሌ The Max Planck Society for the Advancement of Science (MPG, Max- Planck-Gesellschaft zur Förderung der Wissenschaften) is commonly consid- ered a stronghold of basic research in the German research landscape. When it was reestablished in 1948 in the Western zones of occupation, the society adopted the defi ning part of its predecessor’s name, the Kaiser Wilhelm So- ciety for the Advancement of Science (KWG, Kaiser-Wilhelm-Gesellschaft zur Förderung der Wissenschaften), which had been founded in 1911. In contrast to its organizational counterpart, the Fraunhofer Society for the Ad- vancement of Applied Research (Fraunhofer-Gesellschaft zur Förderung der angewandten Forschung), whose distinctive purpose has been incorporated into its name since its foundation in 1949 (Trischler and vom Bruch 1999), the MPG did not include the term “basic research” in its full name, nor does the term appear in any of its statutes of the last one hundred years. The offi - cial documents of the MPG instead refer to “scientifi c research” in the broad German sense of wissenschaftliche Forschung and combine it in varying formu- lations with the terms “freedom” and “independence.”1 Both of these terms are cornerstones in what became known as Harnack- Prinzip (Harnack principle). Named after Adolf von Harnack, the initiator and fi rst and longtime serving president of the KWG, this principle included two main aspects: fi rst, institutes should be founded only if a man was avail- able (women were not considered) for the post of managing director “who has This open access library edition is supported by the University of Bonn. -

Named Organic Reactions (U – Z)
Dr. John Andraos, http://www.careerchem.com/NAMED/Named-Rxns(U-Z).pdf 1 NAMED ORGANIC REACTIONS (U – Z) © Dr. John Andraos, 2000 - 2014 Department of Chemistry, York University 4700 Keele Street, Toronto, ONTARIO M3J 1P3, CANADA For suggestions, corrections, additional information, and comments please send e-mails to [email protected] http://www.chem.yorku.ca/NAMED/ Sakae Uemura Japanese, b. Japan Uemura oxidation Nishimura, T.; Onoue, T.; Ohe, K.; Uemura S. Tetrahedron Lett. 1998 , 39 , 6011 Nishimura, T.; Onoue, T.; Ohe, K.; Uemura S. J. Org. Chem. 1999 , 64 , 6750 Biographical references: Ivar Karl Ugi 5 September 1930 – 29 September 2005 German, b. Kuressaare, Estland, Germany Ugi condensation Ugi, I.; Meyr, R.; Fetzer, U.; Steinbrueckner, C. Angew. Chem. 1959 , 71 , 386 Urban, R.; Ugi, I., Angew. Chem. Int. Engl. Ed . 1975 , 14 , 61 Biographical references: Chemical Research Faculties: An International Directory 1996 , American Chemical Society: Washington, D.C., 1996 Wer ist Wer? Das Deutsche Who’s Who 1999/2000 , Schmidt-Römhild: Lübeck, 1999 Lemmen, P.; Fontain, E.; Bauer, J. Angew. Chem. Int. Ed. 2006 , 45 , 193 Fritz Ullmann 2 July 1875 - 17 March 1939 German, b. Fürth, Germany Ullmann reaction Ullmann, F. Ann. Chem . 1904 , 332 , 38 Dr. John Andraos, http://www.careerchem.com/NAMED/Named-Rxns(U-Z).pdf 2 Biographical references: Meyer, K.H., Helv. Chim. Acta 1940 , 23 , 93 Upjohn reaction Van Rheenen, V.; Kelly, R.C.; Cha, D.Y. Tetrahedron Lett. 1976 , 1973 A. Verley French, b. ? Meerwein-Pondorff-Verley reduction see Hans Leberecht Meerwein Biographical references: Victor Villiger 1 September 1868 - 1934 Swiss, b. -

Otto Hahn Otto Hahn
R.N. 70269/98 Postal Registration No.: DL-SW-1/4082/15-17 ISSN : 0972-169X Date of posting: 26-27 of advance month Date of publication: 24 of advance month May 2017 Vol. 19 No. 8 Rs. 5.00 Otto Hahn Discoverer of Nuclear Fission Editorial: Consolidating 35 science communication activities in our country Otto Hahn: Discoverer of 34 Nuclear Fission Keep Your Eyes Healthy 31 Phenol: A Serious 30 Environmental Threat Accidental Discoveries in 28 Medical Science Cures for haemorrhoids— 24 Simple treatments and Surgeries Recent developments 21 in science and technology 36 Editorial Consolidating science communication activities in our country Dr. R. Gopichandran It is well known that the National Council of Science Museums of India’s leadership in science technology and innovation (STI) across the Ministry of Culture, Government of India, the National Institute the bilateral and multilateral framework also. The news feature service of Science Communication and Information Resources (NISCAIR) and the portal activity have well defined action plans to reach out to of CSIR, the National Council for Science and Technology fellow institutions and citizens with suitably embellished platform Communication (NCSTC) of the Department of Science and and opportunities for all to deliver together. Technology (DST), Government of India and Vigyan Prasar, also While these are interesting and extremely important, especially of DST, have been carrying out excellent science communication because they respond to the call to upscale and value add science activities over the years. It cannot be denied that the reach has been and technology communication, it is equally important to document quite significant collectively. -
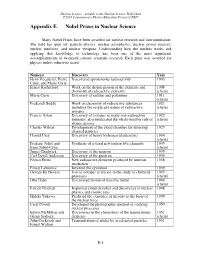
Appendix E Nobel Prizes in Nuclear Science
Nuclear Science—A Guide to the Nuclear Science Wall Chart ©2018 Contemporary Physics Education Project (CPEP) Appendix E Nobel Prizes in Nuclear Science Many Nobel Prizes have been awarded for nuclear research and instrumentation. The field has spun off: particle physics, nuclear astrophysics, nuclear power reactors, nuclear medicine, and nuclear weapons. Understanding how the nucleus works and applying that knowledge to technology has been one of the most significant accomplishments of twentieth century scientific research. Each prize was awarded for physics unless otherwise noted. Name(s) Discovery Year Henri Becquerel, Pierre Discovered spontaneous radioactivity 1903 Curie, and Marie Curie Ernest Rutherford Work on the disintegration of the elements and 1908 chemistry of radioactive elements (chem) Marie Curie Discovery of radium and polonium 1911 (chem) Frederick Soddy Work on chemistry of radioactive substances 1921 including the origin and nature of radioactive (chem) isotopes Francis Aston Discovery of isotopes in many non-radioactive 1922 elements, also enunciated the whole-number rule of (chem) atomic masses Charles Wilson Development of the cloud chamber for detecting 1927 charged particles Harold Urey Discovery of heavy hydrogen (deuterium) 1934 (chem) Frederic Joliot and Synthesis of several new radioactive elements 1935 Irene Joliot-Curie (chem) James Chadwick Discovery of the neutron 1935 Carl David Anderson Discovery of the positron 1936 Enrico Fermi New radioactive elements produced by neutron 1938 irradiation Ernest Lawrence -

Antibodies with Some Bite Antibodies Have Yet to Be Determined, It Is Reasonable to Suppose That the Ester Or David E
-~-------------------------------------N8NSANDVIEWS----------------_N_A_TU_R_E_V-O_L_._32_5_22_J_A_N_U_A_RY__ 19~87 Enzyme catalysis kinetics and is subject to competitive inhibition. Although the detailed chemical mechanisms of these catalytic Antibodies with some bite antibodies have yet to be determined, it is reasonable to suppose that the ester or David E. Hansen carbamate is strained towards a tetra hedral geometry on binding, thus facilitat THE spectacular specificities and rate William Jencks', more directly, stated in ing the attack of the hydroxide ion. Fur enhancements of enzymes are without 1969 thermore, the Scripps group has already equal among all known catalysts. It is no If complementarity between active site and found that their antibody can act via both wonder then that the design of new en transition state contributes significantly to nucleophilic and general base catalysis, zymes has long been a goal of biochemists. enzyme catalysis, it should be possible to syn depending on the substrate used. Now two independent groups, one led by thesize an enzyme by constructing a binding The fact that this approach succeeded at Alfonso Tramontano and Richard Lerner site. One way to do this is to prepare an anti all gives great hope to those attempting to of the Scripps Clinic and Research Foun body to a haptenic group which resembles the isolate even more efficient antibody cat dation', and the other by Peter Schultz of transition state of a given reaction. The com alysts by this and by other strategies. In the University of California at Berkeley\ bining sites of such antibodies should be com addition, it may be possible to modify have taken steps towards this goal by plementary to the transition state and should cause an acceleration by forcing bound sub existing catalytic antibodies.