Excitatory and Inhibitory Synaptic Placement and Functional Implications
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
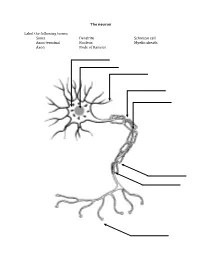
The Neuron Label the Following Terms: Soma Axon Terminal Axon Dendrite
The neuron Label the following terms: Soma Dendrite Schwaan cell Axon terminal Nucleus Myelin sheath Axon Node of Ranvier Neuron Vocabulary You must know the definitions of these terms 1. Synaptic Cleft 2. Neuron 3. Impulse 4. Sensory Neuron 5. Motor Neuron 6. Interneuron 7. Body (Soma) 8. Dendrite 9. Axon 10. Action Potential 11. Myelin Sheath (Myelin) 12. Afferent Neuron 13. Threshold 14. Neurotransmitter 15. Efferent Neurons 16. Axon Terminal 17. Stimulus 18. Refractory Period 19. Schwann 20. Nodes of Ranvier 21. Acetylcholine STEPS IN THE ACTION POTENTIAL 1. The presynaptic neuron sends neurotransmitters to postsynaptic neuron. 2. Neurotransmitters bind to receptors on the postsynaptic cell. - This action will either excite or inhibit the postsynaptic cell. - The soma becomes more positive. 3. The positive charge reaches the axon hillock. - Once the threshold of excitation is reached the neuron will fire an action potential. 4. Na+ channels open and Na+ is forced into the cell by the concentration gradient and the electrical gradient. - The neuron begins to depolarize. 5. The K+ channels open and K+ is forced out of the cell by the concentration gradient and the electrical gradient. - The neuron is depolarized. 6. The Na+ channels close at the peak of the action potential. - The neuron starts to repolarize. 7. The K+ channels close, but they close slowly and K+ leaks out. 8. The terminal buttons release neurotransmitter to the postsynaptic neuron. 9. The resting potential is overshot and the neuron falls to a -90mV (hyperpolarizes). - The neuron continues to repolarize. 10. The neuron returns to resting potential. The Synapse Label the following terms: Pre-synaptic membrane Neurotransmitters Post-synaptic membrane Synaptic cleft Vesicle Post-synaptic receptors . -

Resting and Active Properties of Pyramidal Neurons in Subiculum and CA1 of Rat Hippocampus
Resting and Active Properties of Pyramidal Neurons in Subiculum and CA1 of Rat Hippocampus NATHAN P. STAFF, HAE-YOON JUNG, TARA THIAGARAJAN, MICHAEL YAO, AND NELSON SPRUSTON Department of Neurobiology and Physiology, Institute for Neuroscience, Northwestern University, Evanston, Illinois 60208 Received 22 March 2000; accepted in final form 8 August 2000 Staff, Nathan P., Hae-Yoon Jung, Tara Thiagarajan, Michael region (Chrobak and Buzsaki 1996; Colling et al. 1998). Sub- Yao, and Nelson Spruston. Resting and active properties of pyrami- iculum has been shown to be the major output center of the dal neurons in subiculum and CA1 of rat hippocampus. J Neuro- hippocampus, to which it belongs, sending parallel projections physiol 84: 2398–2408, 2000. Action potentials are the end product of synaptic integration, a process influenced by resting and active neu- from various subicular regions to different cortical and subcor- ronal membrane properties. Diversity in these properties contributes tical areas (Naber and Witter 1998). Functionally, the principal to specialized mechanisms of synaptic integration and action potential neurons of the subiculum appear to be “place cells,” similar to firing, which are likely to be of functional significance within neural those of CA1 (Sharp and Green 1994); and in a human mag- circuits. In the hippocampus, the majority of subicular pyramidal netic resonance imaging (MRI) study, subiculum was shown to neurons fire high-frequency bursts of action potentials, whereas CA1 be an area involved in memory retrieval (Gabrieli et al. 1997). pyramidal neurons exhibit regular spiking behavior when subjected to Therefore both CA1 and subiculum have been implicated in direct somatic current injection. -

A Guide to Glutamate Receptors
A guide to glutamate receptors 1 Contents Glutamate receptors . 4 Ionotropic glutamate receptors . 4 - Structure ........................................................................................................... 4 - Function ............................................................................................................ 5 - AMPA receptors ................................................................................................. 6 - NMDA receptors ................................................................................................. 6 - Kainate receptors ............................................................................................... 6 Metabotropic glutamate receptors . 8 - Structure ........................................................................................................... 8 - Function ............................................................................................................ 9 - Group I: mGlu1 and mGlu5. .9 - Group II: mGlu2 and mGlu3 ................................................................................. 10 - Group III: mGlu4, mGlu6, mGlu7 and mGlu8 ............................................................ 10 Protocols and webinars . 11 - Protocols ......................................................................................................... 11 - Webinars ......................................................................................................... 12 References and further reading . 13 Excitatory synapse pathway -

Neuromodulators and Long-Term Synaptic Plasticity in Learning and Memory: a Steered-Glutamatergic Perspective
brain sciences Review Neuromodulators and Long-Term Synaptic Plasticity in Learning and Memory: A Steered-Glutamatergic Perspective Amjad H. Bazzari * and H. Rheinallt Parri School of Life and Health Sciences, Aston University, Birmingham B4 7ET, UK; [email protected] * Correspondence: [email protected]; Tel.: +44-(0)1212044186 Received: 7 October 2019; Accepted: 29 October 2019; Published: 31 October 2019 Abstract: The molecular pathways underlying the induction and maintenance of long-term synaptic plasticity have been extensively investigated revealing various mechanisms by which neurons control their synaptic strength. The dynamic nature of neuronal connections combined with plasticity-mediated long-lasting structural and functional alterations provide valuable insights into neuronal encoding processes as molecular substrates of not only learning and memory but potentially other sensory, motor and behavioural functions that reflect previous experience. However, one key element receiving little attention in the study of synaptic plasticity is the role of neuromodulators, which are known to orchestrate neuronal activity on brain-wide, network and synaptic scales. We aim to review current evidence on the mechanisms by which certain modulators, namely dopamine, acetylcholine, noradrenaline and serotonin, control synaptic plasticity induction through corresponding metabotropic receptors in a pathway-specific manner. Lastly, we propose that neuromodulators control plasticity outcomes through steering glutamatergic transmission, thereby gating its induction and maintenance. Keywords: neuromodulators; synaptic plasticity; learning; memory; LTP; LTD; GPCR; astrocytes 1. Introduction A huge emphasis has been put into discovering the molecular pathways that govern synaptic plasticity induction since it was first discovered [1], which markedly improved our understanding of the functional aspects of plasticity while introducing a surprisingly tremendous complexity due to numerous mechanisms involved despite sharing common “glutamatergic” mediators [2]. -

Dendritic Spikes and Their Influence on Extracellular Calcium Signaling
Dendritic Spikes and Their Influence on Extracellular Calcium Signaling MICHAEL C. WIEST,1 DAVID M. EAGLEMAN,2 RICHARD D. KING,1 AND P. READ MONTAGUE1 1Division of Neuroscience, Center for Theoretical Neuroscience, Baylor College of Medicine, Houston, Texas 77030; and 2Sloan Center for Theoretical Neurobiology, The Salk Institute, La Jolla, California 92037 Wiest, Michael C., David M. Eagleman, Richard D. King, and P. trical or neurotransmitter stimulation (Benninger et al. 1980; Read Montague. Dendritic spikes and their influence on extracellular Heinemann et al. 1990; Lucke et al. 1995; Nicholson et al. calcium signaling. J. Neurophysiol. 83: 1329–1337, 2000. Extracel- 1978; Pumain and Heinemann 1985; Stanton and Heine- lular calcium is critical for many neural functions, including neuro- mann 1986) (see Fig. 1). Given that many important com- transmission, cell adhesion, and neural plasticity. Experiments have putational processes function on millisecond time scales and shown that normal neural activity is associated with changes in extracellular calcium, which has motivated recent computational work submicron spatial scales, we were led to ask how electrical that employs such fluctuations in an information-bearing role. This events at these smaller scales affect the external calcium possibility suggests that a new style of computing is taking place in level. the mammalian brain in addition to current ‘circuit’ models that use In the mammalian brain, action potentials can propagate into only neurons and connections. Previous computational models of the dendrites of cortical and hippocampal neurons (Stuart and rapid external calcium changes used only rough approximations of Sakmann 1994). These spikes cause large influxes of calcium calcium channel dynamics to compute the expected calcium decre- from the extracellular space (Helmchen et al. -

Crayfish Escape Behavior and Central Synapses
Crayfish Escape Behavior and Central Synapses. III. Electrical Junctions and Dendrite Spikes in Fast Flexor Motoneurons ROBERT S. ZUCKER Department of Biological Sciences and Program in the Neurological Sciences, Stanford University, Stanford, California 94305 THE LATERAL giant fiber is the decision fiber and fast flexor motoneurons are located in for a behavioral response in crayfish in- the dorsal neuropil of each abdominal gan- volving rapid tail flexion in response to glion. Dye injections and anatomical recon- abdominal tactile stimulation (27). Each structions of ‘identified motoneurons reveal lateral giant impulse is followed by a rapid that only those motoneurons which have tail flexion or tail flip, and when the giant dentritic branches in contact with a giant fiber does not fire in response to such stim- fiber are activated by that giant fiber (12, uli, the movement does not occur. 21). All of tl ie fast flexor motoneurons are The afferent limb of the reflex exciting excited by the ipsilateral giant; all but F4, the lateral giant has been described (28), F5 and F7 are also excited by the contra- and the habituation of the behavior has latkral giant (21 a). been explained in terms of the properties The excitation of these motoneurons, of this part of the neural circuit (29). seen as depolarizing potentials in their The properties of the neuromuscular somata, does not always reach threshold for junctions between the fast flexor motoneu- generating a spike which propagates out the rons and the phasic flexor muscles have also axon to the periphery (14). Indeed, only t,he been described (13). -

Plp-Positive Progenitor Cells Give Rise to Bergmann Glia in the Cerebellum
Citation: Cell Death and Disease (2013) 4, e546; doi:10.1038/cddis.2013.74 OPEN & 2013 Macmillan Publishers Limited All rights reserved 2041-4889/13 www.nature.com/cddis Olig2/Plp-positive progenitor cells give rise to Bergmann glia in the cerebellum S-H Chung1, F Guo2, P Jiang1, DE Pleasure2,3 and W Deng*,1,3,4 NG2 (nerve/glial antigen2)-expressing cells represent the largest population of postnatal progenitors in the central nervous system and have been classified as oligodendroglial progenitor cells, but the fate and function of these cells remain incompletely characterized. Previous studies have focused on characterizing these progenitors in the postnatal and adult subventricular zone and on analyzing the cellular and physiological properties of these cells in white and gray matter regions in the forebrain. In the present study, we examine the types of neural progeny generated by NG2 progenitors in the cerebellum by employing genetic fate mapping techniques using inducible Cre–Lox systems in vivo with two different mouse lines, the Plp-Cre-ERT2/Rosa26-EYFP and Olig2-Cre-ERT2/Rosa26-EYFP double-transgenic mice. Our data indicate that Olig2/Plp-positive NG2 cells display multipotential properties, primarily give rise to oligodendroglia but, surprisingly, also generate Bergmann glia, which are specialized glial cells in the cerebellum. The NG2 þ cells also give rise to astrocytes, but not neurons. In addition, we show that glutamate signaling is involved in distinct NG2 þ cell-fate/differentiation pathways and plays a role in the normal development of Bergmann glia. We also show an increase of cerebellar oligodendroglial lineage cells in response to hypoxic–ischemic injury, but the ability of NG2 þ cells to give rise to Bergmann glia and astrocytes remains unchanged. -

Control of Synapse Number by Glia Erik M
R EPORTS RGCs (8). Similar results were obtained un- der both conditions. After 2 weeks in culture, Control of Synapse Number we used whole-cell patch-clamp recording to measure the significant enhancement of spon- by Glia taneous synaptic activity by astrocytes (Fig. 1, A and B) (9). In the absence of astroglia, Erik M. Ullian,* Stephanie K. Sapperstein, Karen S. Christopherson, little synaptic activity occurred even when Ben A. Barres RGCs were cultured with their tectal target cells (6). This difference in synaptic activity Although astrocytes constitute nearly half of the cells in our brain, their persisted even after a month in culture, indi- function is a long-standing neurobiological mystery. Here we show by quantal cating that it is not accounted for by a matu- analyses, FM1-43 imaging, immunostaining, and electron microscopy that few rational delay. To examine whether glia reg- synapses form in the absence of glial cells and that the few synapses that do ulate synaptic activity by enhancing postsyn- form are functionally immature. Astrocytes increase the number of mature, aptic responsiveness, we measured the re- functional synapses on central nervous system (CNS) neurons by sevenfold and sponse of RGCs to pulses of L-glutamate are required for synaptic maintenance in vitro. We also show that most syn- applied to the cell somas. Regardless of the apses are generated concurrently with the development of glia in vivo. These presence of glia, RGCs responded with in- data demonstrate a previously unknown function for glia in inducing and ward currents (Fig. 1C) that were blocked by stabilizing CNS synapses, show that CNS synapse number can be profoundly glutamate receptor antagonists (10). -

Review Article Mechanisms of Cerebrovascular Autoregulation and Spreading Depolarization-Induced Autoregulatory Failure: a Literature Review
Int J Clin Exp Med 2016;9(8):15058-15065 www.ijcem.com /ISSN:1940-5901/IJCEM0026645 Review Article Mechanisms of cerebrovascular autoregulation and spreading depolarization-induced autoregulatory failure: a literature review Gang Yuan1*, Bingxue Qi2*, Qi Luo1 1Department of Neurosurgery, The First Hospital of Jilin University, Changchun, China; 2Department of Endocrinology, Jilin Province People’s Hospital, Changchun, China. *Equal contributors. Received February 25, 2016; Accepted June 4, 2016; Epub August 15, 2016; Published August 30, 2016 Abstract: Cerebrovascular autoregulation maintains brain hemostasis via regulating cerebral flow when blood pres- sure fluctuation occurs. Monitoring autoregulation can be achieved by transcranial Doppler ultrasonography, the pressure reactivity index (PRx) can serve as a secondary index of vascular deterioration, and outcome and prognosis are assessed by the low-frequency PRx. Although great changes in arterial blood pressure (ABP) occur, complex neu- rogenic, myogenic, endothelial, and metabolic mechanisms are involved to maintain the flow within its narrow limits. The steady association between ABP and cerebral blood flow (CBF) reflects static cerebral autoregulation (CA). Spreading depolarization (SD) is a sustained depolarization of neurons with concomitant pronounced breakdown of ion gradients, which originates in patients with brain ischemia, hemorrhage, trauma, and migraine. It is character- ized by the propagation of an extracellular negative potential, followed by an increase in O2 and glucose consump- tion. Immediately after SD, CA is transiently impaired but is restored after 35 min. This process initiates a cascade of pathophysiological mechanisms, leading to neuronal damage and loss if consecutive events are evoked. The clini- cal application of CA in regulating CBF is to dilate the cerebral arteries as a compensatory mechanism during low blood pressure, thus protecting the brain from ischemia. -

Area-Specific Synapse Structure in Branched Posterior Nucleus Axons Reveals a New Level of Complexity in Thalamocortical Networks
The Journal of Neuroscience, March 25, 2020 • 40(13):2663–2679 • 2663 Systems/Circuits Area-Specific Synapse Structure in Branched Posterior Nucleus Axons Reveals a New Level of Complexity in Thalamocortical Networks Javier Rodriguez-Moreno,1 Cesar Porrero,1 Astrid Rollenhagen,2 Mario Rubio-Teves,1 Diana Casas-Torremocha,1 X Lidia Alonso-Nanclares,3 Rachida Yakoubi,2 XAndrea Santuy,3 XAngel Merchan-Pe´rez,3,5,6 XJavier DeFelipe,3,4,5 Joachim H.R. Lu¨bke,2,7,8* and XFrancisco Clasca1* 1Department of Anatomy and Neuroscience, School of Medicine, Auto´noma de Madrid University, 28029 Madrid, Spain, 2Institute of Neuroscience and Medicine INM-10, Research Centre Ju¨lich GmbH, 52425 Ju¨lich, Germany, 3Laboratorio Cajal de Circuitos Corticales, Centro de Tecnología Biome´dica, Universidad Polite´cnica de Madrid, Pozuelo de Alarco´n, 28223 Madrid, Spain, 4Instituto Cajal, Consejo Superior de Investigaciones Científicas, Arce 37 28002, Madrid, Spain, 5CIBERNED, Centro de Investigacio´n Biome´dica en Red de Enfermedades Neurodegenerativas, 28031 Madrid, Spain, 6Departamento de Arquitectura y Tecnología de Sistemas Informa´ticos, Universidad Polite´cnica de Madrid. Boadilla del Monte, 28660 Madrid, Spain, 7Department of Psychiatry, Psychotherapy and Psychosomatics, Medical Faculty RWTH University Hospital Aachen, 52074 Aachen, Germany, and 8JARA-Translational Brain Medicine, 52425 Ju¨lich-Aachen, Germany Thalamocortical posterior nucleus (Po) axons innervating the vibrissal somatosensory (S1) and motor (MC) cortices are key links in the brain neuronal network that allows rodents to explore the environment whisking with their motile snout vibrissae. Here, using fine-scale high-end 3D electron microscopy, we demonstrate in adult male C57BL/6 wild-type mice marked differences between MC versus S1 Po synapses in (1) bouton and active zone size, (2) neurotransmitter vesicle pool size, (3) distribution of mitochondria around synapses, and (4) proportion of synapses established on dendritic spines and dendritic shafts. -

The Interplay Between Neurons and Glia in Synapse Development And
Available online at www.sciencedirect.com ScienceDirect The interplay between neurons and glia in synapse development and plasticity Jeff A Stogsdill and Cagla Eroglu In the brain, the formation of complex neuronal networks and regulate distinct aspects of synaptic development and amenable to experience-dependent remodeling is complicated circuit connectivity. by the diversity of neurons and synapse types. The establishment of a functional brain depends not only on The intricate communication between neurons and glia neurons, but also non-neuronal glial cells. Glia are in and their cooperative roles in synapse formation are now continuous bi-directional communication with neurons to direct coming to light due in large part to advances in genetic the formation and refinement of synaptic connectivity. This and imaging tools. This article will examine the progress article reviews important findings, which uncovered cellular made in our understanding of the role of mammalian and molecular aspects of the neuron–glia cross-talk that perisynaptic glia (astrocytes and microglia) in synapse govern the formation and remodeling of synapses and circuits. development, maturation, and plasticity since the previ- In vivo evidence demonstrating the critical interplay between ous Current Opinion article [1]. An integration of past and neurons and glia will be the major focus. Additional attention new findings of glial control of synapse development and will be given to how aberrant communication between neurons plasticity is tabulated in Box 1. and glia may contribute to neural pathologies. Address Glia control the formation of synaptic circuits Department of Cell Biology, Duke University Medical Center, Durham, In the CNS, glial cells are in tight association with NC 27710, USA synapses in all brain regions [2]. -

PRESYNAPTIC Α2δ SUBUNITS ARE KEY ORGANIZERS of GLUTAMATERGIC SYNAPSES Clemens L
bioRxiv preprint doi: https://doi.org/10.1101/826016; this version posted October 31, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. PRESYNAPTIC α2δ SUBUNITS ARE KEY ORGANIZERS OF GLUTAMATERGIC SYNAPSES Clemens L. Schöpf1, Stefanie Geisler1, Ruslan I. Stanika1, Marta Campiglio1, Walter A. Kaufmann3, Benedikt Nimmervoll1, Bettina Schlick1, Ryuichi Shigemoto3, and Gerald J. Obermair1,2* From Division of Physiology, Medical University Innsbruck, A-6020 Innsbruck1, Division Physiology, Karl Landsteiner University of Health Sciences, A-3500 Krems2, and Institute of Science and Technology Austria, A-3400 Klosterneuburg3, Austria. Running head: Presynaptic α2δ subunits organize synapses Address correspondence to: *Gerald J. Obermair, Univ.-Prof. Dr., Phone: +43-2732-72090490, E-mail: [email protected], [email protected] In nerve cells the genes encoding for α2δ the auxiliary β and α2δ subunits. α2δ subunits, the subunits of voltage-gated calcium channels targets of the widely prescribed anti-epileptic and (VGCCs) have been linked to synaptic functions anti-allodynic drugs gabapentin and pregabalin, are and neurological disease. Here we show that α2δ membrane-anchored extracellular glycoproteins, subunits are essential for the formation and which modulate VGCC trafficking and calcium organization of glutamatergic synapses. Using a currents (Arikkath and Campbell, 2003; Dolphin, cellular α2δ subunit triple loss-of-function model, 2013; Geisler et al., 2015; Obermair et al., 2008; we demonstrate a failure in presynaptic Zamponi et al., 2015).