Advanced Materials Modelling and Characterization.Pdf
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

CESMIX: Center for the Exascale Simulation of Materials in Extreme Environments
CESMIX: Center for the Exascale Simulation of Materials in Extreme Environments Project Overview Youssef Marzouk MIT PSAAP-3 Team 18 August 2020 The CESMIX team • Our team integrates expertise in quantum chemistry, atomistic simulation, materials science; hypersonic flow; validation & uncertainty quantification; numerical algorithms; parallel computing, programming languages, compilers, and software performance engineering 1 Project objectives • Exascale simulation of materials in extreme environments • In particular: ultrahigh temperature ceramics in hypersonic flows – Complex materials, e.g., metal diborides – Extreme aerothermal and chemical loading – Predict materials degradation and damage (oxidation, melting, ablation), capturing the central role of surfaces and interfaces • New predictive simulation paradigms and new CS tools for the exascale 2 Broad relevance • Intense current interest in reentry vehicles and hypersonic flight – A national priority! – Materials technologies are a key limiting factor • Material properties are of cross-cutting importance: – Oxidation rates – Thermo-mechanical properties: thermal expansion, creep, fracture – Melting and ablation – Void formation • New systems being proposed and fabricated (e.g., metallic high-entropy alloys) • May have relevance to materials aging • Yet extreme environments are largely inaccessible in the laboratory – Predictive simulation is an essential path… 3 Demonstration problem: specifics • Aerosurfaces of a hypersonic vehicle… • Hafnium diboride (HfB2) promises necessary temperature -

Popular GPU-Accelerated Applications
LIFE & MATERIALS SCIENCES GPU-ACCELERATED APPLICATIONS | CATALOG | AUG 12 LIFE & MATERIALS SCIENCES APPLICATIONS CATALOG Application Description Supported Features Expected Multi-GPU Release Status Speed Up* Support Bioinformatics BarraCUDA Sequence mapping software Alignment of short sequencing 6-10x Yes Available now reads Version 0.6.2 CUDASW++ Open source software for Smith-Waterman Parallel search of Smith- 10-50x Yes Available now protein database searches on GPUs Waterman database Version 2.0.8 CUSHAW Parallelized short read aligner Parallel, accurate long read 10x Yes Available now aligner - gapped alignments to Version 1.0.40 large genomes CATALOG GPU-BLAST Local search with fast k-tuple heuristic Protein alignment according to 3-4x Single Only Available now blastp, multi cpu threads Version 2.2.26 GPU-HMMER Parallelized local and global search with Parallel local and global search 60-100x Yes Available now profile Hidden Markov models of Hidden Markov Models Version 2.3.2 mCUDA-MEME Ultrafast scalable motif discovery algorithm Scalable motif discovery 4-10x Yes Available now based on MEME algorithm based on MEME Version 3.0.12 MUMmerGPU A high-throughput DNA sequence alignment Aligns multiple query sequences 3-10x Yes Available now LIFE & MATERIALS& LIFE SCIENCES APPLICATIONS program against reference sequence in Version 2 parallel SeqNFind A GPU Accelerated Sequence Analysis Toolset HW & SW for reference 400x Yes Available now assembly, blast, SW, HMM, de novo assembly UGENE Opensource Smith-Waterman for SSE/CUDA, Fast short -
Biovia Materials Studio Visualizer Datasheet
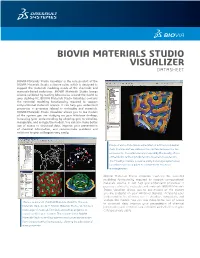
BIOVIA MATERIALS STUDIO VISUALIZER DATASHEET BIOVIA Materials Studio Visualizer is the core product of the BIOVIA Materials Studio software suite, which is designed to support the materials modeling needs of the chemicals and materials-based industries. BIOVIA Materials Studio brings science validated by leading laboratories around the world to your desktop PC. BIOVIA Materials Studio Visualizer contains the essential modeling functionality required to support computational materials science. It can help you understand properties or processes related to molecules and materials. BIOVIA Materials Studio Visualizer allows you to see models of the system you are studying on your Windows desktop, increasing your understanding by allowing you to visualize, manipulate, and analyze the models. You can also make better use of access to structural data, improve your presentation of chemical information, and communicate problems and solutions to your colleagues very easily. Image of early-stage phase segregation in a diblock copolymer melt. The blue surface indicates the interface between the two components. The volume is colormapped by the density of one of the blocks, red being high density, blue being low-density. The MesoDyn module is used to study these large systems over long-times such as required to observe these structural rearrangements. BIOVIA Materials Studio Visualizer contains the essential modeling functionality required to support computational materials science. It can help you understand properties or processes related to molecules and materials. BIOVIA Materials Studio Visualizer allows you to see models of the system you are studying on your Windows desktop, increasing your understanding by allowing you to visualize, manipulate, and analyze the models. -

Download Author Version (PDF)
PCCP Accepted Manuscript This is an Accepted Manuscript, which has been through the Royal Society of Chemistry peer review process and has been accepted for publication. Accepted Manuscripts are published online shortly after acceptance, before technical editing, formatting and proof reading. Using this free service, authors can make their results available to the community, in citable form, before we publish the edited article. We will replace this Accepted Manuscript with the edited and formatted Advance Article as soon as it is available. You can find more information about Accepted Manuscripts in the Information for Authors. Please note that technical editing may introduce minor changes to the text and/or graphics, which may alter content. The journal’s standard Terms & Conditions and the Ethical guidelines still apply. In no event shall the Royal Society of Chemistry be held responsible for any errors or omissions in this Accepted Manuscript or any consequences arising from the use of any information it contains. www.rsc.org/pccp Page 1 of 11 PhysicalPlease Chemistry do not adjust Chemical margins Physics PCCP PAPER Effect of nanosize on surface properties of NiO nanoparticles for adsorption of Quinolin-65 ab a a Received 00th January 20xx, Nedal N. Marei, Nashaat N. Nassar* and Gerardo Vitale Accepted 00th January 20xx Using Quinolin-65 (Q-65) as a model-adsorbing compound for polar heavy hydrocarbons, the nanosize effect of NiO Manuscript DOI: 10.1039/x0xx00000x nanoparticles on adsorption of Q-65 was investigated. Different-sized NiO nanoparticles with sizes between 5 and 80 nm were prepared by controlled thermal dehydroxylation of Ni(OH)2. -

Supporting Information
Electronic Supplementary Material (ESI) for Chemical Science. This journal is © The Royal Society of Chemistry 2015 Supporting Information A Single Crystalline Porphyrinic Titanium Metal−Organic Framework Shuai Yuana†, Tian-Fu Liua†, Dawei Fenga, Jian Tiana, Kecheng Wanga, Junsheng Qina, a a a a b Qiang Zhang , Ying-Pin Chen , Mathieu Bosch , Lanfang Zou , Simon J. Teat, Scott J. c a Dalgarno and Hong-Cai Zhou * a Department of Chemistry, Texas A&M University, College Station, Texas 77842-3012, USA b Advanced Light Source, Lawrence Berkeley National Laboratory Berkeley, CA 94720, USA c Institute of Chemical Sciences, Heriot-Watt University Riccarton, Edinburgh EH14 4AS, U.K. † Equal contribution to this work *To whom correspondence should be addressed. Email: [email protected] Tel: +1 (979) 845-4034; Fax: +1 (979) 845-1595 S1 Contents S1. Ligand Synthesis..............................................................................................................3 S2. Syntheses of PCN-22.......................................................................................................5 S3. X-ray Crystallography .....................................................................................................6 S4. Topological Analyses ......................................................................................................9 S5. N2 Sorption Isotherm .....................................................................................................10 S6. Simulation of the Accessible Surface Area ...................................................................12 -

Copyrighted Material
PART I METHODS COPYRIGHTED MATERIAL CHAPTER 1 Overview of Thermochemistry and Its Application to Reaction Kinetics ELKE GOOS Institute of Combustion Technology, German Aerospace Center (DLR), Stuttgart, Germany ALEXANDER BURCAT Faculty of Aerospace Engineering, Technion - Israel Institute of Technology, Haifa, Israel 1.1 HISTORY OF THERMOCHEMISTRY Thermochemistry deals with energy and enthalpy changes accompanying chemical reactions and phase transformations and gives a first estimate of whether a given reaction can occur. To our knowledge, the field of thermochemistry started with the experiments done by Malhard and Le Chatelier [1] with gunpowder and explosives. The first of their two papers of 1883 starts with the sentence: “All combustion is accompanied by the release of heat that increases the temperature of the burned bodies.” In 1897, Berthelot [2], who also experimented with explosives, published his two-volume monograph Thermochimie in which he summed up 40 years of calori- metric studies. The first textbook, to our knowledge, that clearly explained the principles of thermochemical properties was authored by Lewis and Randall [3] in 1923. Thermochemical data, actually heats of formation, were gathered, evaluated, and published for the first time in the seven-volume book International Critical Tables of Numerical Data, Physics, Chemistry and Technology [4] during 1926–1930 (and the additional index in 1933). In 1932, the American Chemical Society (ACS) monograph No. 60 The Free Energy of Some Organic Compounds [5] appeared. Rate Constant Calculation for Thermal Reactions: Methods and Applications, Edited by Herbert DaCosta and Maohong Fan. Ó 2012 John Wiley & Sons, Inc. Published 2012 by John Wiley & Sons, Inc. 3 4 OVERVIEW OF THERMOCHEMISTRY AND ITS APPLICATION TO REACTION KINETICS In 1936 was published The Thermochemistry of the Chemical Substances [6] where the authors Bichowsky and Rossini attempted to standardize the available data and published them at a common temperature of 18C (291K) and pressure of 1 atm. -

Appendix a a List of the Program and Data Files on the Disk*
Appendix A A List of the Program and Data Files on the Disk* 2dnrnr Simulates a IH COSY two-dimensional NMR plot (5.5). Abc Calculates principal moments of inertia for linear and non linear molecules (4.5). Acetate Calculates the pH in an acetate buffer (3.2). Anderko Solves the Anderko-Pitzer nonideal gas law (1.2). Aorbital Calculates and plots H atomic orbitals in two dimensions (4.3). Atpl Calculates and plots the apparent standard Gibbs energy I:irGo 1 for the ATP hydrolysis reaction at various pMg's and a specified pH and ionic strength (3.3). Atp2 Calculates and plots the apparent standard Gibbs energy I:irGo 1 for the ATP hydrolysis reaction at various pH's and a specified pMg and ionic strength (3.3). Beattie Solves the Beattie-Bridgeman nonideal gas law (1.2). Beyer Calculates and plots the density of states N(E) and the sum of states G(E) for a molecule's vibrational modes using the Beyer-Swinehart direct-count algorithm (11.4). Biochern Calculates and plots the apparent standard Gibbs energy I:irGo 1 for the F -+ M biochemical reaction at various pH's (3.3). Biokin Calculates and plots extracellular glucose and insulin con centrations as functions of time (10.4). Branch Calculates and plots H, 0, OH and 02 concentrations for the branching catalytic cycle that drives the 2H2 + 02 -+ 2H20 reaction (10.2). Brussels Calculates and plots concentrations of intermediates in the autocatalytic Brusselator model (11.5). Butler Calculates and plots the Butler-Volmer and Tafel equations relating electrode current and overpotential (11.6). -

Reactive Molecular Dynamics Study of the Thermal Decomposition of Phenolic Resins
Article Reactive Molecular Dynamics Study of the Thermal Decomposition of Phenolic Resins Marcus Purse 1, Grace Edmund 1, Stephen Hall 1, Brendan Howlin 1,* , Ian Hamerton 2 and Stephen Till 3 1 Department of Chemistry, Faculty of Engineering and Physical Sciences, University of Surrey, Guilford, Surrey GU2 7XH, UK; [email protected] (M.P.); [email protected] (G.E.); [email protected] (S.H.) 2 Bristol Composites Institute (ACCIS), Department of Aerospace Engineering, School of Civil, Aerospace, and Mechanical Engineering, University of Bristol, Bristol BS8 1TR, UK; [email protected] 3 Defence Science and Technology Laboratory, Porton Down, Salisbury SP4 0JQ, UK; [email protected] * Correspondence: [email protected]; Tel.: +44-1483-686-248 Received: 6 March 2019; Accepted: 23 March 2019; Published: 28 March 2019 Abstract: The thermal decomposition of polyphenolic resins was studied by reactive molecular dynamics (RMD) simulation at elevated temperatures. Atomistic models of the polyphenolic resins to be used in the RMD were constructed using an automatic method which calls routines from the software package Materials Studio. In order to validate the models, simulated densities and heat capacities were compared with experimental values. The most suitable combination of force field and thermostat for this system was the Forcite force field with the Nosé–Hoover thermostat, which gave values of heat capacity closest to those of the experimental values. Simulated densities approached a final density of 1.05–1.08 g/cm3 which compared favorably with the experimental values of 1.16–1.21 g/cm3 for phenol-formaldehyde resins. -

Dmol Guide to Select a Dmol3 Task 1
DMOL3 GUIDE MATERIALS STUDIO 8.0 Copyright Notice ©2014 Dassault Systèmes. All rights reserved. 3DEXPERIENCE, the Compass icon and the 3DS logo, CATIA, SOLIDWORKS, ENOVIA, DELMIA, SIMULIA, GEOVIA, EXALEAD, 3D VIA, BIOVIA and NETVIBES are commercial trademarks or registered trademarks of Dassault Systèmes or its subsidiaries in the U.S. and/or other countries. All other trademarks are owned by their respective owners. Use of any Dassault Systèmes or its subsidiaries trademarks is subject to their express written approval. Acknowledgments and References To print photographs or files of computational results (figures and/or data) obtained using BIOVIA software, acknowledge the source in an appropriate format. For example: "Computational results obtained using software programs from Dassault Systèmes Biovia Corp.. The ab initio calculations were performed with the DMol3 program, and graphical displays generated with Materials Studio." BIOVIA may grant permission to republish or reprint its copyrighted materials. Requests should be submitted to BIOVIA Support, either through electronic mail to [email protected], or in writing to: BIOVIA Support 5005 Wateridge Vista Drive, San Diego, CA 92121 USA Contents DMol3 1 Setting up a molecular dynamics calculation20 Introduction 1 Choosing an ensemble 21 Further Information 1 Defining the time step 21 Tasks in DMol3 2 Defining the thermostat control 21 Energy 3 Constraints during dynamics 21 Setting up the calculation 3 Setting up a transition state calculation 22 Dynamics 4 Which method to use? -

Kepler Gpus and NVIDIA's Life and Material Science
LIFE AND MATERIAL SCIENCES Mark Berger; [email protected] Founded 1993 Invented GPU 1999 – Computer Graphics Visual Computing, Supercomputing, Cloud & Mobile Computing NVIDIA - Core Technologies and Brands GPU Mobile Cloud ® ® GeForce Tegra GRID Quadro® , Tesla® Accelerated Computing Multi-core plus Many-cores GPU Accelerator CPU Optimized for Many Optimized for Parallel Tasks Serial Tasks 3-10X+ Comp Thruput 7X Memory Bandwidth 5x Energy Efficiency How GPU Acceleration Works Application Code Compute-Intensive Functions Rest of Sequential 5% of Code CPU Code GPU CPU + GPUs : Two Year Heart Beat 32 Volta Stacked DRAM 16 Maxwell Unified Virtual Memory 8 Kepler Dynamic Parallelism 4 Fermi 2 FP64 DP GFLOPS GFLOPS per DP Watt 1 Tesla 0.5 CUDA 2008 2010 2012 2014 Kepler Features Make GPU Coding Easier Hyper-Q Dynamic Parallelism Speedup Legacy MPI Apps Less Back-Forth, Simpler Code FERMI 1 Work Queue CPU Fermi GPU CPU Kepler GPU KEPLER 32 Concurrent Work Queues Developer Momentum Continues to Grow 100M 430M CUDA –Capable GPUs CUDA-Capable GPUs 150K 1.6M CUDA Downloads CUDA Downloads 1 50 Supercomputer Supercomputers 60 640 University Courses University Courses 4,000 37,000 Academic Papers Academic Papers 2008 2013 Explosive Growth of GPU Accelerated Apps # of Apps Top Scientific Apps 200 61% Increase Molecular AMBER LAMMPS CHARMM NAMD Dynamics GROMACS DL_POLY 150 Quantum QMCPACK Gaussian 40% Increase Quantum Espresso NWChem Chemistry GAMESS-US VASP CAM-SE 100 Climate & COSMO NIM GEOS-5 Weather WRF Chroma GTS 50 Physics Denovo ENZO GTC MILC ANSYS Mechanical ANSYS Fluent 0 CAE MSC Nastran OpenFOAM 2010 2011 2012 SIMULIA Abaqus LS-DYNA Accelerated, In Development NVIDIA GPU Life Science Focus Molecular Dynamics: All codes are available AMBER, CHARMM, DESMOND, DL_POLY, GROMACS, LAMMPS, NAMD Great multi-GPU performance GPU codes: ACEMD, HOOMD-Blue Focus: scaling to large numbers of GPUs Quantum Chemistry: key codes ported or optimizing Active GPU acceleration projects: VASP, NWChem, Gaussian, GAMESS, ABINIT, Quantum Espresso, BigDFT, CP2K, GPAW, etc. -

Clefs CEA N°60
No. 60 clefsSummer 2011 Chemistry is everywhere No. 60 - Summer 2011 clefs Chemistry is everywhere www.cea.fr No. 60 Summer 2011 clefs Chemistry is everywhere Chemistry 2 Foreword, by Valérie Cabuil is everywhere I. NUCLEAR CHEMISTRY Clefs CEA No. 60 – SUMMER 2011 4 Introduction, by Stéphane Sarrade Main cover picture Dyed polymers for photovoltaic cells. 6 Advances in the separation For many years, CEA has been applying chemistry of actinides, all aspects of chemistry, in all its forms. Chemistry is at the very heart of all its by Pascal Baron major programs, whether low-carbon 10 The chemical specificities energies (nuclear energy and new energy technologies), biomedical and of actinides, environmental technologies or the by Philippe Moisy information technologies. 11 Uranium chemistry: significant P. Avavian/CEA – C. Dupont/CEA advances, Inset by Marinella Mazzanti top: Placing corrosion samples in a high-temperature furnace. 12 Chemistry and chemical P. Stroppa/CEA engineering, the COEX process, by Stéphane Grandjean bottom: Gas sensors incorporating “packaged” NEMS. P. Avavian/CEA 13 Supercritical fluids in chemical Pictogram on inside pages processes, © Fotolia by Audrey Hertz and Frédéric Charton Review published by CEA Communication Division 14 The chemistry of corrosion, Bâtiment Siège by Damien Féron, Christophe Gallé 91191 Gif-sur-Yvette Cedex (France) and Stéphane Gin Phone: + 33 (0)1 64 50 10 00 Fax (editor’s office): + 33 (0)1 64 50 17 22 14 17 Focus A Advances in modeling Executive publisher Xavier Clément in chemistry, by Philippe Guilbaud, Editor in chief Jean-Pierre Dognon, Didier Mathieu, 21 Understanding the chemical Marie-José Loverini (until 30/06/2011) Christophe Morell, André Grand mechanisms of radiolysis and Pascale Maldivi by Gérard Baldacchino Deputy editor Martine Trocellier [email protected] Scientific committee Bernard Bonin, Gilles Damamme, Céline Gaiffier, Étienne Klein, II. -

Chemical File Format Conversion Tools : a N Overview
International Journal of Engineering Research & Technology (IJERT) ISSN: 2278-0181 Vol. 3 Issue 2, February - 2014 Chemical File Format Conversion Tools : A n Overview Kavitha C. R Dr. T Mahalekshmi Research Scholar, Bharathiyar University Principal Dept of Computer Applications Sree Narayana Institute of Technology SNGIST Kollam, India Cochin, India Abstract— There are a lot of chemical data stored in large different chemical file formats. Three types of file format databases, repositories and other resources. These data are used conversion tools are discussed in section III. And the by different researchers in different applications in various areas conclusion is given in section IV followed by the references. of chemistry. Since these data are stored in several standard chemical file formats, there is a need for the inter-conversion of II. CHEMICAL FILE FORMATS chemical structures between different formats because all the formats are not supported by various software and tools used by A chemical is a collection of atoms bonded together the researchers. Therefore it becomes essential to convert one file in space. The structure of a chemical makes it unique and format to another. This paper reviews some of the chemical file gives it its physical and biological characteristics. This formats and also presents a few inter-conversion tools such as structure is represented in a variety of chemical file formats. Open Babel [1], Mol converter [2] and CncTranslate [3]. These formats are used to represent chemical structure records and its associated data fields. Some of the file Keywords— File format Conversion, Open Babel, mol formats are CML (Chemical Markup Language), SDF converter, CncTranslate, inter- conversion tools.