Phosphoproteomics
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Tyrosine Kinase – Role and Significance in Cancer
Int. J. Med. Sci. 2004 1(2): 101-115 101 International Journal of Medical Sciences ISSN 1449-1907 www.medsci.org 2004 1(2):101-115 ©2004 Ivyspring International Publisher. All rights reserved Review Tyrosine kinase – Role and significance in Cancer Received: 2004.3.30 Accepted: 2004.5.15 Manash K. Paul and Anup K. Mukhopadhyay Published:2004.6.01 Department of Biotechnology, National Institute of Pharmaceutical Education and Research, Sector-67, S.A.S Nagar, Mohali, Punjab, India-160062 Abstract Tyrosine kinases are important mediators of the signaling cascade, determining key roles in diverse biological processes like growth, differentiation, metabolism and apoptosis in response to external and internal stimuli. Recent advances have implicated the role of tyrosine kinases in the pathophysiology of cancer. Though their activity is tightly regulated in normal cells, they may acquire transforming functions due to mutation(s), overexpression and autocrine paracrine stimulation, leading to malignancy. Constitutive oncogenic activation in cancer cells can be blocked by selective tyrosine kinase inhibitors and thus considered as a promising approach for innovative genome based therapeutics. The modes of oncogenic activation and the different approaches for tyrosine kinase inhibition, like small molecule inhibitors, monoclonal antibodies, heat shock proteins, immunoconjugates, antisense and peptide drugs are reviewed in light of the important molecules. As angiogenesis is a major event in cancer growth and proliferation, tyrosine kinase inhibitors as a target for anti-angiogenesis can be aptly applied as a new mode of cancer therapy. The review concludes with a discussion on the application of modern techniques and knowledge of the kinome as means to gear up the tyrosine kinase drug discovery process. -

Phosphoproteomics: Recent Advances in Analytical Techniques
International Journal of Pharmaceuticals Analysis, ISSN: 0975-3079, Volume 1, Issue 2, 2009, pp-31-36 Phosphoproteomics: Recent advances in analytical techniques Gomase V.S. and Ramu Akella Department of Biotechnology, Padmashree Dr. D.Y. Patil University, Navi Mumbai, 400614, India Abstract - Post-genomic biology seeks identification and quantification of multiple proteins from complex mixtures and the research is still on. Despite recent progress in high-throughput proteomics, proteomic analysis of post-translationally modified [PTM] proteins remains particularly challenging. Several strategies for isolating phosphoproteins are explored herein. Quantification of phosphoproteins seems to be a novel solution to identify the underlying disease mechanisms, mostly cancer. Keywords - phosphorylation, enrichment, chemical modification, SILAC, mass spectrometry, HILIC Introduction Techniques Phosphoproteomics is a branch of proteomics Of late, there is an increasing interest in that focuses on deriving a comprehensive view of phosphoproteomics as reflected by a the extent & dynamics of protein phosphorylation considerable number of works describing various by way of identifying & characterizing proteins strategies, including the use of different isotopic that contain a phosphate group as a post- technologies [e.g., isotope-coded affinity tag, translational modification. The addition of stable isotope labeling by amino acids in cell phosphate is a key reversible modification culture], protein separation techniques [e.g., gel- catalyzed by protein kinases, and is known as based versus liquid chromatography-based ‘phosphorylation’. Protein kinases account for methods], and chemical modifications [e.g., β- 1.7% of the human genome and 40% of all the elimination and esterification] to identify proteins may be assumed to be phosphorylated phosphoproteins and/or phosphorylation sites in at any given time. -

Principles of Protein Phosphorylation Biophysical Chemistry 1, Fall 2010
Principles of protein phosphorylation Biophysical Chemistry 1, Fall 2010 Signalling “cascades” Structural biology of phosphorylation Web assignment: http://pkr.genomics.purdue.edu Reversible protein phosphorylation Enzymatic reaction Posttranslational Control ΔG~12kcal/mol Kinases and phosphatases Phosphatase Kinase dephospho- phosphorylates rylates General Examples Signalling overview Cells are way too complex! MAPK/ERK Signaling Pathway RousExample:sarcoma virus Rous (RSV) sarcoma virus (RSV) gag - encodes capsid proteins pol - encodes reverse transcriptase env - encodes envelope proteins src - encodes a tyrosine kinase that attaches phosphate groups to the amino acid tyrosine in host cell proteins MutationsExample:, viruses Rous and cancer sarcoma virus (RSV) v-src lacks the C-terminal inhibitory phosphorylation site (tyrosine-527), and is therefore constitutively active as opposed to normal src (c-src) Continuous cell profileration tumor BiophysicsStructural of signalling Effect of Phosphorylation Phosphorylation is an important regulatory mechanism Can reversible attach/detach a phosphate and therefore switch “on”/”off” the function Effect of phosphorylation is manifold • Conformational change • Ordering/disordering • Electrostatic effects • Alternate binding behavior Signalling by reorientation Reorientation: A conformational switch Rmsd = 2.5Å DHP (red) and Z-Score = 4.6 DHPs74e (blue) (>3.6 same fold) Title: A phosphorylation-induced conformation change in dematin headpiece Author(s): Jiang ZHG, McKnight CJ Source: STRUCTURE Volume: 14 Issue: 2 Pages: 379-387 Published: FEB 2006 Order/disorderDisordering:transitions NtrC, a molecular switch upon phosphorylation Orange-yellow: unphosphorylated NtrC blue-cyan: phosphorylated NtrC Volkman et al., Science 2001, 291, 2429-33 Alternate Binding: SRC SH2 Src/SH2 interactions: binding vs release domain binding ExpectedCan conformationalwe understand effects (predict) the effect of phosphorylation Electrostatics Hydrogen bonding Size Mutational analysis Experimental data Lubman, O.Y. -

Enrichment Strategies for Phosphoproteomics: State-Of-The-Art
Rev Anal Chem 31 (2012): 29–41 © 2012 by Walter de Gruyter • Berlin • Boston. DOI 10.1515/revac-2011-0025 Enrichment strategies for phosphoproteomics: state-of-the-art Barbora Salovska 1,2, *, Ales Tichy 1,2 , Martina MIP molecularly imprinted polymer Rezacova 1 , Jirina Vavrova 2 and Eva Novotna 2 MOAC metal oxide affi nity chromatography MS mass spectrometry 1 Department of Medical Biochemistry , Faculty of NTA nitriloacetic acid Medicine in Hradec Kralove, Charles University in PTMs post-translational modifi cations Prague, Hradec Kr á lov é , Czech Republic , pS phosphoserine e-mail: [email protected] pT phosphothreonine 2 Department of Radiobiology , Faculty of Military Health pY phosphotyrosine Sciences, University of Defence, Hradec Kralove , RPLC reversed phase liquid chromatography Czech Republic SAX strong anion-exchange chromatography SCX strong cation-exchange chromatography * Corresponding author Introduction Abstract Protein phosphorylation is a key regulator in many biological The human genome involves approximately 30,000 protein- processes, such as homeostasis, cellular signaling and com- coding genes; the human proteome contains several million munication, transcriptional and translational regulation, and different protein effectors. This is due to alternative splic- apoptosis. The defects in this tightly controlled reversible post- ing of genes and post-translational modifi cations (PTMs). translational modifi cation have been described to contribute Several hundred PTMs are currently known, among them to genesis and -

Computational Biology, Protein Chemistry and Mass Spectrometry
CORE Metadata, citation and similar papers at core.ac.uk Provided by Elsevier - Publisher Connector FEBS Letters 580 (2006) 4764–4770 Minireview Phosphoproteomics toolbox: Computational biology, protein chemistry and mass spectrometry Majbrit Hjerrild*, Steen Gammeltoft Department of Clinical Biochemistry, Glostrup Hospital, Nordre Ringvej, DK-2600 Glostrup, Denmark Received 1 June 2006; revised 21 July 2006; accepted 25 July 2006 Available online 4 August 2006 Edited by Francesc Posas tion, or interaction with other proteins. Regulation of the Abstract Protein phosphorylation is important for regulation of most biological functions and up to 50% of all proteins are cell cycle, membrane transport and permeability, cell adhesion, thought to be modified by protein kinases. Increased knowledge neurotransmission, and metabolism are examples of biological about potential phosphorylation of a protein may increase our functions that are modulated through protein phosphorylation understanding of the molecular processes in which it takes part. [1]. The human genome contains 518 different protein kinases Despite the importance of protein phosphorylation, identification and identification of their biological targets is an active re- of phosphoproteins and localization of phosphorylation sites is search area [2]. Even though the number of identified phospho- still a major challenge in proteomics. However, high-throughput proteins is rapidly increasing especially due to development of methods for identification of phosphoproteins are being devel- high-throughput methods for the identification of phospho- oped, in particular within the fields of bioinformatics and mass proteins, in particular within the fields of bioinformatics and spectrometry. In this review, we present a toolbox of current mass spectrometry, it is believed that only a small fraction of technology applied in phosphoproteomics including compu- tational prediction, chemical approaches and mass spectro- physiological phosphorylation sites has been assigned. -
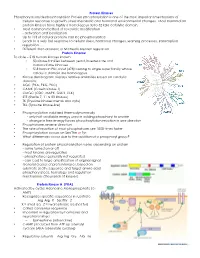
Protein Kinases Phosphorylation/Dephosphorylation Protein Phosphorylation Is One of the Most Important Mechanisms of Cellular Re
Protein Kinases Phosphorylation/dephosphorylation Protein phosphorylation is one of the most important mechanisms of cellular responses to growth, stress metabolic and hormonal environmental changes. Most mammalian protein kinases have highly a homologous 30 to 32 kDa catalytic domain. • Most common method of reversible modification - activation and localization • Up to 1/3 of cellular proteins can be phosphorylated • Leads to a very fast response to cellular stress, hormonal changes, learning processes, transcription regulation .... • Different than allosteric or Michealis Menten regulation Protein Kinome To date – 518 human kinases known • 50 kinase families between yeast, invertebrate and mammaliane kinomes • 518 human PKs, most (478) belong to single super family whose catalytic domain are homologous. • Kinase dendrogram displays relative similarities based on catalytic domains. • AGC (PKA, PKG, PKC) • CAMK (Casein kinase 1) • CMGC (CDC, MAPK, GSK3, CLK) • STE (Sterile 7, 11 & 20 kinases) • TK (Tryosine kinases memb and cyto) • TKL (Tyrosine kinase-like) • Phosphorylation stabilized thermodynamically - only half available energy used in adding phosphoryl to protein - change in free energy forces phosphorylation reaction in one direction • Phosphatases reverse direction • The rate of reaction of most phosphatases are 1000 times faster • Phosphorylation occurs on Ser/The or Tyr • What differences occur due to the addition of a phosphoryl group? • Regulation of protein phosphorylation varies depending on protein - some turned on or off -

Β-Catenin Signaling Dynamics Regulate Cell Fate in Differentiating Neural Stem Cells
β-Catenin signaling dynamics regulate cell fate in differentiating neural stem cells Alyssa B. Rosenblooma, Marcin Tarczynski a, Nora Lama, Ravi S. Kaneb,1, Lukasz J. Bugaja,c,1, and David V. Schaffera,d,e,f,1 aDepartment of Bioengineering, University of California, Berkeley, CA 94720; bSchool of Chemical & Biomolecular Engineering, Georgia Institute of Technology, Atlanta, GA 30332; cDepartment of Bioengineering, University of Pennsylvania, Philadelphia, PA 19104; dDepartment of Chemical and Biomolecular Engineering, University of California, Berkeley, CA 94720; eDepartment of Molecular and Cell Biology, University of California, Berkeley, CA 94720; and fHelen Wills Neuroscience Institute, University of California, Berkeley, CA 94720 Edited by Randall T. Moon, University of Washington, Seattle, WA, and approved September 21, 2020 (received for review May 4, 2020) Stem cells undergo differentiation in complex and dynamic environ- of how signaling dynamics impact cellular function. Optogenetics ments wherein instructive signals fluctuate on various timescales. has recently emerged as a field in which light—which can readily Thus, cells must be equipped to properly respond to the timing of be varied in intensity, space, and time—is harnessed to precisely signals, for example, to distinguish sustained signaling from transient modulate cell-signaling pathways. In this approach, light-sensitive noise. However, how stem cells respond to dynamic variations in proteins are engineered to interface with specific signaling path- differentiation cues is not well characterized. Here, we use optoge- ways, and the subsequent introduction of such an optogenetic netic activation of β-catenin signaling to probe the dynamic responses construct into cells renders the signaling pathway responsive to of differentiating adult neural stem cells (NSCs). -

Structural Basis of O-Glcnac Recognition by Mammalian 14-3-3 Proteins
Structural basis of O-GlcNAc recognition by mammalian 14-3-3 proteins Clifford A. Tolemana,1, Maria A. Schumachera,1, Seok-Ho Yub, Wenjie Zenga, Nathan J. Coxa, Timothy J. Smitha, Erik J. Soderblomc, Amberlyn M. Wandsb, Jennifer J. Kohlerb, and Michael Boycea,2 aDepartment of Biochemistry, Duke University School of Medicine, Durham, NC 27710; bDepartment of Biochemistry, University of Texas Southwestern Medical Center, Dallas, TX 75390; and cDuke Proteomics and Metabolomics Core Facility, Center for Genomic and Computational Biology, Duke University, Durham, NC 27710 Edited by Carolyn R. Bertozzi, Stanford University, Stanford, CA, and approved April 23, 2018 (received for review December 24, 2017) O-GlcNAc is an intracellular posttranslational modification that gov- Results erns myriad cell biological processes and is dysregulated in human We developed a biochemical approach to test the hypothesis that diseases. Despite this broad pathophysiological significance, the O-GlcNAc is specifically recognized by mammalian reader pro- biochemical effects of most O-GlcNAcylation events remain unchar- teins. First, we derived a consensus O-GlcNAcylated peptide acterized. One prevalent hypothesis is that O-GlcNAc moieties may sequence by aligning 802 mapped Ser-O-GlcNAc sites (34–36) be recognized by “reader” proteins to effect downstream signaling. (Fig. 1A)(www.phosphosite.org). We noted that a Pro-Val-Ser However, no general O-GlcNAc readers have been identified, leav- tripeptide observed previously in smaller datasets (37, 38) also ing a considerable gap in the field. To elucidate O-GlcNAc signaling emerged in our sequence, suggesting that this motif may be mechanisms, we devised a biochemical screen for candidate O-GlcNAc important for O-GlcNAc modification and/or recognition. -

233668.Full.Pdf
bioRxiv preprint doi: https://doi.org/10.1101/233668; this version posted December 15, 2017. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC-ND 4.0 International license. Comparative Qualitative Phosphoproteomics Analysis Identifies Shared Phosphorylation Motifs and Associated Biological Processes in Flowering Plants Shireen Al-Momani1, Da Qi1, Zhe Ren2, Andrew R Jones1,* 1Institute of Integrative Biology, University of Liverpool, Liverpool, L69 7ZB, UK 2BGI-Shenzhen, Shenzhen 518083, China *= corresponding author, [email protected] Running title: Shared Phosphorylation Motifs in Flowering Plants Abbreviations GO Gene Ontology pProtein phosphorylated protein pS phospho-serine pT phospho-threonine pY phospho-tyrosine Summary Phosphorylation is regarded as one of the most prevalent post-translational modifications and plays a key role in regulating cellular processes. In this work we carried out a comparative bioinformatics analysis of phosphoproteomics data, to profile two model species representing the largest subclasses in flowering plants the dicot Arabidopsis thaliana and the monocot Oryza sativa, to understand the extent to which phosphorylation signaling and function is conserved across evolutionary divergent plants. Using pre-existing mass spectrometry phosphoproteomics datasets and bioinformatic tools and resources, we identified 6,537 phosphopeptides from 3,189 phosphoproteins in Arabidopsis and 2,307 phosphopeptides from 1,613 phosphoproteins in rice. The relative abundance ratio of serine, threonine, and tyrosine phosphorylation sites in rice and Arabidopsis were highly similar: 88.3: 11.4: 0.4 and 86.7: 12.8: 0.5, respectively. -

The Origins of Protein Phosphorylation
historical perspective The origins of protein phosphorylation Philip Cohen The reversible phosphorylation of proteins is central to the regulation of most aspects of cell func- tion but, even after the first protein kinase was identified, the general significance of this discovery was slow to be appreciated. Here I review the discovery of protein phosphorylation and give a per- sonal view of the key findings that have helped to shape the field as we know it today. he days when protein phosphorylation was an abstruse backwater, best talked Tabout between consenting adults in private, are over. My colleagues no longer cringe on hearing that “phosphorylase kinase phosphorylates phosphorylase” and their eyes no longer glaze over when a “”kinase kinase kinase” is mentioned. This is because protein phosphorylation has gradu- ally become an integral part of all the sys- tems they are studying themselves. Indeed it would be difficult to find anyone today who would disagree with the statement that “the reversible phosphorylation of proteins regu- lates nearly every aspect of cell life”. Phosphorylation and dephosphorylation, catalysed by protein kinases and protein phosphatases, can modify the function of a protein in almost every conceivable way; for Carl and Gerty Cori, the 1947 Nobel Laureates. Picture: Science Photo Library. example by increasing or decreasing its bio- logical activity, by stabilizing it or marking it for destruction, by facilitating or inhibiting movement between subcellular compart- so long before its general significance liver enzyme that catalysed the phosphory- ments, or by initiating or disrupting pro- was appreciated? lation of casein3. Soon after, Fischer and tein–protein interactions. -

Protein Tyrosine Kinases: Their Roles and Their Targeting in Leukemia
cancers Review Protein Tyrosine Kinases: Their Roles and Their Targeting in Leukemia Kalpana K. Bhanumathy 1,*, Amrutha Balagopal 1, Frederick S. Vizeacoumar 2 , Franco J. Vizeacoumar 1,3, Andrew Freywald 2 and Vincenzo Giambra 4,* 1 Division of Oncology, College of Medicine, University of Saskatchewan, Saskatoon, SK S7N 5E5, Canada; [email protected] (A.B.); [email protected] (F.J.V.) 2 Department of Pathology and Laboratory Medicine, College of Medicine, University of Saskatchewan, Saskatoon, SK S7N 5E5, Canada; [email protected] (F.S.V.); [email protected] (A.F.) 3 Cancer Research Department, Saskatchewan Cancer Agency, 107 Wiggins Road, Saskatoon, SK S7N 5E5, Canada 4 Institute for Stem Cell Biology, Regenerative Medicine and Innovative Therapies (ISBReMIT), Fondazione IRCCS Casa Sollievo della Sofferenza, 71013 San Giovanni Rotondo, FG, Italy * Correspondence: [email protected] (K.K.B.); [email protected] (V.G.); Tel.: +1-(306)-716-7456 (K.K.B.); +39-0882-416574 (V.G.) Simple Summary: Protein phosphorylation is a key regulatory mechanism that controls a wide variety of cellular responses. This process is catalysed by the members of the protein kinase su- perfamily that are classified into two main families based on their ability to phosphorylate either tyrosine or serine and threonine residues in their substrates. Massive research efforts have been invested in dissecting the functions of tyrosine kinases, revealing their importance in the initiation and progression of human malignancies. Based on these investigations, numerous tyrosine kinase inhibitors have been included in clinical protocols and proved to be effective in targeted therapies for various haematological malignancies. -

The T-Cell Protein Tyrosine Phosphatase Is Phosphorylated On
CORE Metadata, citation and similar papers at core.ac.uk Provided by Cold Spring Harbor Laboratory Institutional Repository Biochem. J. (2004) 380, 939–949 (Printed in Great Britain) 939 The T-cell protein tyrosine phosphatase is phosphorylated on Ser-304 by cyclin-dependent protein kinases in mitosis Patricia BUKCZYNSKA*1, Manuela KLINGLER-HOFFMANN*1, Kenneth I. MITCHELHILL†2, Mark H. C. LAM*3, Melissa CICCOMANCINI*4, Nicholas K. TONKS‡, Boris SARCEVIC§, Bruce E. KEMP† and Tony TIGANIS*5 *Department of Biochemistry and Molecular Biology, Monash University, Melbourne, Victoria 3800, Australia, †St. Vincent’s Institute of Medical Research, Melbourne, Victoria 3065, Australia, ‡Cold Spring Harbor Laboratory, Cold Spring Harbor, NY 11724, U.S.A., and §Cancer Research Program, Garvan Institute of Medical Research, Darlinghurst, New South Wales 2010, Australia Two alternatively spliced forms of the human protein tyrosine no apparent effect on TC45 localization. Ser-304 phosphorylation phosphatase TCPTP (T-cell protein tyrosine phosphatase) exist: was ablated when cells were treated with the CDK (cyclin- a 48 kDa form that is targeted to the endoplasmic reticulum dependent protein kinase) inhibitors roscovitine or SU9516, (TC48) and a shorter 45 kDa form that is targeted to the but remained unaltered when ERK1/2 activation was inhibited nucleus (TC45). In this study we have identified Ser-304 (Phe301- with the MEK (mitogen-activated protein kinase/extracellular- Asp-His-Ser304-Pro-Asn-Lys307) as a major TCPTP phosphory- signal-regulated kinase kinase) inhibitor PD98059. In addition, lation site and demonstrate that TC45, but not TC48, is phos- recombinant CDKs, but not the Polo-like kinase Plk1, phos- phorylated on this site in vivo.