Definitions of Terms Relating to Mass Spectrometry (IUPAC Recommendations 2013)*
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

DFT-Based Reaction Pathway Analysis of Hexadiene Cyclization Via Carbenium Ion Intermediates: Mechanistic Study of Light Alkane Aromatization Catalysis
J. Phys. Chem. B 2004, 108, 971-980 971 DFT-Based Reaction Pathway Analysis of Hexadiene Cyclization via Carbenium Ion Intermediates: Mechanistic Study of Light Alkane Aromatization Catalysis Yogesh V. Joshi, Aditya Bhan, and Kendall T. Thomson* School of Chemical Engineering, Purdue UniVersity, West Lafayette, Indiana 47907 ReceiVed: July 28, 2003; In Final Form: NoVember 4, 2003 We conducted density functional theory calculations to identify the complete cyclization mechanism (ring formation and ring expansion) for protonated hexadiene in the gas-phase as a precursory means of studying aromatization of light alkanes in acidic zeolite catalysts. We identify the rate-determining step to consist of ring expansion from a methylcyclopenta carbenium precursor to a stable cyclohexa carbenium intermediate, exhibiting an activation barrier of 9.6 kcal/mol and proceeding through a bicyclic intermediate starting from a secondary cyclopentyl carbocation. This pathway for ring closure was preferred over tertiary precursor expansion and direct cyclization to a cyclohexyl carbocation. Expecting carbocation intermediates to be represented by alkoxide species near Brønsted acid sites, we calculated the relative stability of primary, secondary, and tertiary alkoxide analogues to cyclopentyl carbocation intermediates involved in ring expansion and notice a reversal of stability relative to gas-phase carbocation stabilities (i.e., primary > secondary > tertiary stability). However, on the basis of the notion that transition state stability depends heavily -
Appendix A: Symbols and Prefixes
Appendix A: Symbols and Prefixes (Appendix A last revised November 2020) This appendix of the Author's Kit provides recommendations on prefixes, unit symbols and abbreviations, and factors for conversion into units of the International System. Prefixes Recommended prefixes indicating decimal multiples or submultiples of units and their symbols are as follows: Multiple Prefix Abbreviation 1024 yotta Y 1021 zetta Z 1018 exa E 1015 peta P 1012 tera T 109 giga G 106 mega M 103 kilo k 102 hecto h 10 deka da 10-1 deci d 10-2 centi c 10-3 milli m 10-6 micro μ 10-9 nano n 10-12 pico p 10-15 femto f 10-18 atto a 10-21 zepto z 10-24 yocto y Avoid using compound prefixes, such as micromicro for pico and kilomega for giga. The abbreviation of a prefix is considered to be combined with the abbreviation/symbol to which it is directly attached, forming with it a new unit symbol, which can be raised to a positive or negative power and which can be combined with other unit abbreviations/symbols to form abbreviations/symbols for compound units. For example: 1 cm3 = (10-2 m)3 = 10-6 m3 1 μs-1 = (10-6 s)-1 = 106 s-1 1 mm2/s = (10-3 m)2/s = 10-6 m2/s Abbreviations and Symbols Whenever possible, avoid using abbreviations and symbols in paragraph text; however, when it is deemed necessary to use such, define all but the most common at first use. The following is a recommended list of abbreviations/symbols for some important units. -

Chapter 9 Titrimetric Methods 413
Chapter 9 Titrimetric Methods Chapter Overview Section 9A Overview of Titrimetry Section 9B Acid–Base Titrations Section 9C Complexation Titrations Section 9D Redox Titrations Section 9E Precipitation Titrations Section 9F Key Terms Section 9G Chapter Summary Section 9H Problems Section 9I Solutions to Practice Exercises Titrimetry, in which volume serves as the analytical signal, made its first appearance as an analytical method in the early eighteenth century. Titrimetric methods were not well received by the analytical chemists of that era because they could not duplicate the accuracy and precision of a gravimetric analysis. Not surprisingly, few standard texts from the 1700s and 1800s include titrimetric methods of analysis. Precipitation gravimetry developed as an analytical method without a general theory of precipitation. An empirical relationship between a precipitate’s mass and the mass of analyte— what analytical chemists call a gravimetric factor—was determined experimentally by taking a known mass of analyte through the procedure. Today, we recognize this as an early example of an external standardization. Gravimetric factors were not calculated using the stoichiometry of a precipitation reaction because chemical formulas and atomic weights were not yet available! Unlike gravimetry, the development and acceptance of titrimetry required a deeper understanding of stoichiometry, of thermodynamics, and of chemical equilibria. By the 1900s, the accuracy and precision of titrimetric methods were comparable to that of gravimetric methods, establishing titrimetry as an accepted analytical technique. 411 412 Analytical Chemistry 2.0 9A Overview of Titrimetry We are deliberately avoiding the term In titrimetry we add a reagent, called the titrant, to a solution contain- analyte at this point in our introduction ing another reagent, called the titrand, and allow them to react. -
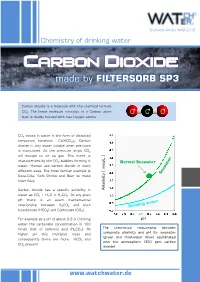
Carbon Dioxide - Made by FILTERSORB SP3
Technical Article MAR 2015 Chemistry of drinking water Carbon dioxide - made by FILTERSORB SP3 Carbon dioxide is a molecule with the chemical formula CO2. The linear molecule consists of a Carbon atom O C O that is doubly bonded with two Oxygen atoms. CO2 exists in water in the form of dissolved temporary hardness Ca(HCO3)2. Carbon dioxide is only water soluble when pressure is maintained. As the pressure drops CO2 will escape to air as gas. This event is characterized by the CO2 bubbles forming in /L ] Normal Seawater water. Human use carbon dioxide in many meq different ways. The most familiar example is Coca-Cola, Soft Drinks and Beer to make them fizzy. Carbon dioxide has a specific solubility in [ Alkalinity water as CO2 + H2O ⇌ H2CO3. At any given pH there is an exact mathematical relationship between H2CO3 and both bicarbonate (HCO3) and Carbonate (CO3). For example at a pH of about 9.3 in Drinking pH water the carbonate concentration is 100 The theoretical relationship between times that of carbonic acid (H2CO3). At carbonate alkalinity and pH for seawater higher pH this multiplier rises and (green and freshwater (blue) equilibrated consequently there are more HCO and 3 with the atmosphere (350 ppm carbon CO present 3 dioxide) www.watchwater.de Water ® Technology FILTERSORB SP3 WATCH WATER & Chemicals Making the healthiest water How SP3 Water functions in human body ? Answer: Carbon dioxide is a waste product of the around these hollow spaces will spasm and respiratory system, and of several other constrict. chemical reactions in the body such as the Transportation of oxygen to the tissues: Oxygen creation of ATP. -

Fundamental Understanding of Chemical Processes in Extreme Ultraviolet Resist Materials
Lawrence Berkeley National Laboratory Recent Work Title Fundamental understanding of chemical processes in extreme ultraviolet resist materials. Permalink https://escholarship.org/uc/item/50h4g627 Journal The Journal of chemical physics, 149(15) ISSN 0021-9606 Authors Kostko, Oleg Xu, Bo Ahmed, Musahid et al. Publication Date 2018-10-01 DOI 10.1063/1.5046521 Peer reviewed eScholarship.org Powered by the California Digital Library University of California Fundamental understanding of chemical processes in extreme ultraviolet resist materials Oleg Kostko,1 Bo Xu,1 Musahid Ahmed,1 Daniel S. Slaughter,1 D. Frank Ogletree,2 Kristina D. Closser,2 David G. Prendergast,2 Patrick Naulleau,3 Deirdre L. Olynick,2 Paul D. Ashby,2 Yi Liu,2 William D. Hinsberg,4 Gregory M. Wallraff5 1Chemical Sciences Division, Lawrence Berkeley National Laboratory, Berkeley, CA, USA 2Molecular Foundry, Lawrence Berkeley National Laboratory, Berkeley, CA, USA 3Center for X‐Ray Optics, Lawrence Berkeley National Laboratory, Berkeley, CA, USA 4Columbia Hill Technical Consulting, Fremont, CA, USA 5IBM, Almaden, CA, USA ABSTRACT New photoresists are needed to advance extreme ultraviolet (EUV) lithography. Tailored design of efficient photoresists is enabled by a fundamental understanding of EUV induced chemistry. Processes that occur in the resist film after absorption of an EUV photon are discussed and a novel approach to study these processes on a fundamental level is described. The processes of photoabsorption, electron emission, and molecular fragmentation were studied experimentally in the gas‐phase on analogues of the monomer units employed in chemically amplified EUV resists. To demonstrate the dependence of the EUV absorption cross‐section on selective light harvesting substituents, halogenated methylpenols were characterized employing the following techniques. -

A Complete Guide to Benzene
A Complete Guide to Benzene Benzene is an important organic chemical compound with the chemical formula C6H6. The benzene molecule is composed of six carbon atoms joined in a ring with one hydrogen atom attached to each. As it contains only carbon and hydrogen atoms, benzene is classed as a hydrocarbon. Benzene is a natural constituent of crude oil and is one of the elementary petrochemicals. Due to the cyclic continuous pi bond between the carbon atoms, benzene is classed as an aromatic hydrocarbon, the second [n]- annulene ([6]-annulene). It is sometimes abbreviated Ph–H. Benzene is a colourless and highly flammable liquid with a sweet smell. Source: Wikipedia Protecting people and the environment Protecting both people and the environment whilst meeting the operational needs of your business is very important and, if you have operations in the UK you will be well aware of the requirements of the CoSHH Regulations1 and likewise the Code of Federal Regulations (CFR) in the US2. Similar legislation exists worldwide, the common theme being an onus on hazard identification, risk assessment and the provision of appropriate control measures (bearing in mind the hierarchy of controls) as well as health surveillance in most cases. And whilst toxic gasses such as hydrogen sulphide and carbon monoxide are a major concern because they pose an immediate (acute) danger to life, long term exposure to relatively low level concentrations of other gasses or vapours such as volatile organic compounds (VOC) are of equal importance because of the chronic illnesses that can result from that ongoing exposure. Benzene, a common VOC Organic means the chemistry of carbon based compounds, which are substances that results from a combination of two or more different chemical elements. -

Analytical Chemistry Laboratory Manual 2
ANALYTICAL CHEMISTRY LABORATORY MANUAL 2 Ankara University Faculty of Pharmacy Department of Analytical Chemistry Analytical Chemistry Practices Contents INTRODUCTION TO QUANTITATIVE ANALYSIS ......................................................................... 2 VOLUMETRIC ANALYSIS .............................................................................................................. 2 Volumetric Analysis Calculations ................................................................................................... 3 Dilution Factor ................................................................................................................................ 4 Standard Solutions ........................................................................................................................... 5 Primary standard .............................................................................................................................. 5 Characteristics of Quantitative Reaction ......................................................................................... 5 Preparation of 1 L 0.1 M HCl Solution ........................................................................................... 6 Preparation of 1 L 0.1 M NaOH Solution ....................................................................................... 6 NEUTRALIZATION TITRATIONS ...................................................................................................... 7 STANDARDIZATION OF 0.1 N NaOH SOLUTION ...................................................................... -

Analytical Chemistry in the 21St Century: Challenges, Solutions, and Future Perspectives of Complex Matrices Quantitative Analyses in Biological/Clinical Field
Review Analytical Chemistry in the 21st Century: Challenges, Solutions, and Future Perspectives of Complex Matrices Quantitative Analyses in Biological/Clinical Field 1, 2, 2, 3 Giuseppe Maria Merone y, Angela Tartaglia y, Marcello Locatelli * , Cristian D’Ovidio , Enrica Rosato 3, Ugo de Grazia 4 , Francesco Santavenere 5, Sandra Rossi 5 and Fabio Savini 5 1 Department of Neuroscience, Imaging and Clinical Sciences, University of Chieti–Pescara “G. d’Annunzio”, Via dei Vestini 31, 66100 Chieti, Italy; [email protected] 2 Department of Pharmacy, University of Chieti-Pescara “G. d’Annunzio”, Via Dei Vestini 31, 66100 Chieti (CH), Italy; [email protected] 3 Section of Legal Medicine, Department of Medicine and Aging Sciences, University of Chieti–Pescara “G. d’Annunzio”, 66100 Chieti, Italy; [email protected] (C.D.); [email protected] (E.R.) 4 Laboratory of Neurological Biochemistry and Neuropharmacology, Fondazione IRCCS Istituto Neurologico Carlo Besta, Via Celoria 11, 20133 Milano, Italy; [email protected] 5 Pharmatoxicology Laboratory—Hospital “Santo Spirito”, Via Fonte Romana 8, 65124 Pescara, Italy; [email protected] (F.S.); [email protected] (S.R.); [email protected] (F.S.) * Correspondence: [email protected]; Tel.: +39-0871-3554590; Fax: +39-0871-3554911 These authors contributed equally to the work. y Received: 16 September 2020; Accepted: 16 October 2020; Published: 30 October 2020 Abstract: Nowadays, the challenges in analytical chemistry, and mostly in quantitative analysis, include the development and validation of new materials, strategies and procedures to meet the growing need for rapid, sensitive, selective and green methods. -

About SI Units
Units SI units: International system of units (French: “SI” means “Système Internationale”) The seven SI base units are: Mass: kg Defined by the prototype “standard kilogram” located in Paris. The standard kilogram is made of Pt metal. Length: m Originally defined by the prototype “standard meter” located in Paris. Then defined as 1,650,763.73 wavelengths of the orange-red radiation of Krypton86 under certain specified conditions. (Official definition: The distance traveled by light in vacuum during a time interval of 1 / 299 792 458 of a second) Time: s The second is defined as the duration of a certain number of oscillations of radiation coming from Cs atoms. (Official definition: The second is the duration of 9,192,631,770 periods of the radiation of the hyperfine- level transition of the ground state of the Cs133 atom) Current: A Defined as the current that causes a certain force between two parallel wires. (Official definition: The ampere is that constant current which, if maintained in two straight parallel conductors of infinite length, of negligible circular cross-section, and placed 1 meter apart in vacuum, would produce between these conductors a force equal to 2 × 10-7 Newton per meter of length. Temperature: K One percent of the temperature difference between boiling point and freezing point of water. (Official definition: The Kelvin, unit of thermodynamic temperature, is the fraction 1 / 273.16 of the thermodynamic temperature of the triple point of water. Amount of substance: mol The amount of a substance that contains Avogadro’s number NAvo = 6.0221 × 1023 of atoms or molecules. -

Physical and Analytical Electrochemistry: the Fundamental
Electrochemical Systems The simplest and traditional electrochemical process occurring at the boundary between an electronically conducting phase (the electrode) and an ionically conducting phase (the electrolyte solution), is the heterogeneous electro-transfer step between the electrode and the electroactive species of interest present in the solution. An example is the plating of nickel. Ni2+ + 2e- → Ni Physical and Analytical The interface is where the action occurs but connected to that central event are various processes that can Electrochemistry: occur in parallel or series. Figure 2 demonstrates a more complex interface; it represents a molecular scale snapshot The Fundamental Core of an interface that exists in a fuel cell with a solid polymer (capable of conducting H+) as an electrolyte. of Electrochemistry However, there is more to an electrochemical system than a single by Tom Zawodzinski, Shelley Minteer, interface. An entire circuit must be made and Gessie Brisard for measurable current to flow. This circuit consists of the electrochemical cell plus external wiring and circuitry The common event for all electrochemical processes is that (power sources, measuring devices, of electron transfer between chemical species; or between an etc). The cell consists of (at least) electrode and a chemical species situated in the vicinity of the two electrodes separated by (at least) one electrolyte solution. Figure 3 is electrode, usually a pure metal or an alloy. The location where a schematic of a simple circuit. Each the electron transfer reactions take place is of fundamental electrode has an interface with a importance in electrochemistry because it regulates the solution. Electrons flow in the external behavior of most electrochemical systems. -

Contents • Introduction to Proteomics and Mass Spectrometry
Contents • Introduction to Proteomics and Mass spectrometry - What we need to know in our quest to explain life - Fundamental things you need to know about mass spectrometry • Interfaces and ion sources - Electrospray ionization (ESI) - conventional and nanospray - Heated nebulizer atmospheric pressure chemical ionization - Matrix assisted laser desorption • Types of MS analyzers - Magnetic sector - Quadrupole - Time-of-flight - Hybrid - Ion trap In the next step in our quest to explain what is life The human genome project has largely been completed and many other genomes are surrendering to the gene sequencers. However, all this knowledge does not give us the information that is needed to explain how living cells work. To do that, we need to study proteins. In 2002, mass spectrometry has developed to the point where it has the capacity to obtain the "exact" molecular weight of many macromolecules. At the present time, this includes proteins up to 150,000 Da. Proteins of higher molecular weights (up to 500,000 Da) can also be studied by mass spectrometry, but with less accuracy. The paradigm for sequencing of peptides and identification of proteins has changed – because of the availability of the human genome database, peptides can be identified merely by their masses or by partial sequence information, often in minutes, not hours. This new capacity is shifting the emphasis of biomedical research back to the functional aspects of cell biochemistry, the expression of particular sets of genes and their gene products, the proteins of the cell. These are the new goals of the biological scientist: o to know which proteins are expressed in each cell, preferably one cell at a time o to know how these proteins are modified, information that cannot necessarily be deduced from the nucleotide sequence of individual genes. -

1.3.4 Atoms and Molecules Name Symbol Definition SI Unit
1.3.4 Atoms and molecules Name Symbol Definition SI unit Notes nucleon number, A 1 mass number proton number, Z 1 atomic number neutron number N N = A - Z 1 electron rest mass me kg (1) mass of atom, ma, m kg atomic mass 12 atomic mass constant mu mu = ma( C)/12 kg (1), (2) mass excess ∆ ∆ = ma - Amu kg elementary charge, e C proton charage Planck constant h J s Planck constant/2π h h = h/2π J s 2 2 Bohr radius a0 a0 = 4πε0 h /mee m -1 Rydberg constant R∞ R∞ = Eh/2hc m 2 fine structure constant α α = e /4πε0 h c 1 ionization energy Ei J electron affinity Eea J (1) Analogous symbols are used for other particles with subscripts: p for proton, n for neutron, a for atom, N for nucleus, etc. (2) mu is equal to the unified atomic mass unit, with symbol u, i.e. mu = 1 u. In biochemistry the name dalton, with symbol Da, is used for the unified atomic mass unit, although the name and symbols have not been accepted by CGPM. Chapter 1 - 1 Name Symbol Definition SI unit Notes electronegativity χ χ = ½(Ei +Eea) J (3) dissociation energy Ed, D J from the ground state D0 J (4) from the potential De J (4) minimum principal quantum n E = -hcR/n2 1 number (H atom) angular momentum see under Spectroscopy, section 3.5. quantum numbers -1 magnetic dipole m, µ Ep = -m⋅⋅⋅B J T (5) moment of a molecule magnetizability ξ m = ξB J T-2 of a molecule -1 Bohr magneton µB µB = eh/2me J T (3) The concept of electronegativity was intoduced by L.