Thermodynamics Part II
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
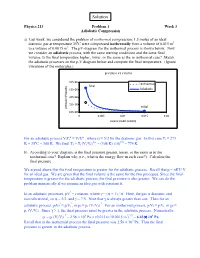
3-1 Adiabatic Compression
Solution Physics 213 Problem 1 Week 3 Adiabatic Compression a) Last week, we considered the problem of isothermal compression: 1.5 moles of an ideal diatomic gas at temperature 35oC were compressed isothermally from a volume of 0.015 m3 to a volume of 0.0015 m3. The pV-diagram for the isothermal process is shown below. Now we consider an adiabatic process, with the same starting conditions and the same final volume. Is the final temperature higher, lower, or the same as the in isothermal case? Sketch the adiabatic processes on the p-V diagram below and compute the final temperature. (Ignore vibrations of the molecules.) α α For an adiabatic process ViTi = VfTf , where α = 5/2 for the diatomic gas. In this case Ti = 273 o 1/α 2/5 K + 35 C = 308 K. We find Tf = Ti (Vi/Vf) = (308 K) (10) = 774 K. b) According to your diagram, is the final pressure greater, lesser, or the same as in the isothermal case? Explain why (i.e., what is the energy flow in each case?). Calculate the final pressure. We argued above that the final temperature is greater for the adiabatic process. Recall that p = nRT/ V for an ideal gas. We are given that the final volume is the same for the two processes. Since the final temperature is greater for the adiabatic process, the final pressure is also greater. We can do the problem numerically if we assume an idea gas with constant α. γ In an adiabatic processes, pV = constant, where γ = (α + 1) / α. -

Isobaric Expansion Engines: New Opportunities in Energy Conversion for Heat Engines, Pumps and Compressors
energies Concept Paper Isobaric Expansion Engines: New Opportunities in Energy Conversion for Heat Engines, Pumps and Compressors Maxim Glushenkov 1, Alexander Kronberg 1,*, Torben Knoke 2 and Eugeny Y. Kenig 2,3 1 Encontech B.V. ET/TE, P.O. Box 217, 7500 AE Enschede, The Netherlands; [email protected] 2 Chair of Fluid Process Engineering, Paderborn University, Pohlweg 55, 33098 Paderborn, Germany; [email protected] (T.K.); [email protected] (E.Y.K.) 3 Chair of Thermodynamics and Heat Engines, Gubkin Russian State University of Oil and Gas, Leninsky Prospekt 65, Moscow 119991, Russia * Correspondence: [email protected] or [email protected]; Tel.: +31-53-489-1088 Received: 12 December 2017; Accepted: 4 January 2018; Published: 8 January 2018 Abstract: Isobaric expansion (IE) engines are a very uncommon type of heat-to-mechanical-power converters, radically different from all well-known heat engines. Useful work is extracted during an isobaric expansion process, i.e., without a polytropic gas/vapour expansion accompanied by a pressure decrease typical of state-of-the-art piston engines, turbines, etc. This distinctive feature permits isobaric expansion machines to serve as very simple and inexpensive heat-driven pumps and compressors as well as heat-to-shaft-power converters with desired speed/torque. Commercial application of such machines, however, is scarce, mainly due to a low efficiency. This article aims to revive the long-known concept by proposing important modifications to make IE machines competitive and cost-effective alternatives to state-of-the-art heat conversion technologies. Experimental and theoretical results supporting the isobaric expansion technology are presented and promising potential applications, including emerging power generation methods, are discussed. -

High Efficiency and Large-Scale Subsurface Energy Storage with CO2
PROCEEDINGS, 43rd Workshop on Geothermal Reservoir Engineering Stanford University, Stanford, California, February 12-14, 2018 SGP-TR-213 High Efficiency and Large-scale Subsurface Energy Storage with CO2 Mark R. Fleming1, Benjamin M. Adams2, Jimmy B. Randolph1,3, Jonathan D. Ogland-Hand4, Thomas H. Kuehn1, Thomas A. Buscheck3, Jeffrey M. Bielicki4, Martin O. Saar*1,2 1University of Minnesota, Minneapolis, MN, USA; 2ETH-Zurich, Zurich, Switzerland; 3TerraCOH Inc., Minneapolis, MN, USA; 4The Ohio State University, Columbus, OH, USA; 5Lawrence Livermore National Laboratory, Livermore, CA, USA. *Corresponding Author: [email protected] Keywords: geothermal energy, multi-level geothermal systems, sedimentary basin, carbon dioxide, electricity generation, energy storage, power plot, CPG ABSTRACT Storing large amounts of intermittently produced solar or wind power for later, when there is a lack of sunlight or wind, is one of society’s biggest challenges when attempting to decarbonize energy systems. Traditional energy storage technologies tend to suffer from relatively low efficiencies, severe environmental concerns, and limited scale both in capacity and time. Subsurface energy storage can solve the drawbacks of many other energy storage approaches, as it can be large scale in capacity and time, environmentally benign, and highly efficient. When CO2 is used as the (pressure) energy storage medium in reservoirs underneath caprocks at depths of at least ~1 km (to ensure the CO2 is in its supercritical state), the energy generated after the energy storage operation can be greater than the energy stored. This is possible if reservoir temperatures and CO2 storage durations combine to result in more geothermal energy input into the CO2 at depth than what the CO2 pumps at the surface (and other machinery) consume. -

Muddiest Point – Entropy and Reversible I Am Confused About Entropy and How It Is Different in a Reversible Versus Irreversible Case
1 Muddiest Point { Entropy and Reversible I am confused about entropy and how it is different in a reversible versus irreversible case. Note: Some of the discussion below follows from the previous muddiest points comment on the general idea of a reversible and an irreversible process. You may wish to have a look at that comment before reading this one. Let's talk about entropy first, and then we will consider how \reversible" gets involved. Generally we divide the universe into two parts, a system (what we are studying) and the surrounding (everything else). In the end the total change in the entropy will be the sum of the change in both, dStotal = dSsystem + dSsurrounding: This total change of entropy has only two possibilities: Either there is no spontaneous change (equilibrium) and dStotal = 0, or there is a spontaneous change because we are not at equilibrium, and dStotal > 0. Of course the entropy change of each piece, system or surroundings, can be positive or negative. However, the second law says the sum must be zero or positive. Let's start by thinking about the entropy change in the system and then we will add the entropy change in the surroundings. Entropy change in the system: When you consider the change in entropy for a process you should first consider whether or not you are looking at an isolated system. Start with an isolated system. An isolated system is not able to exchange energy with anything else (the surroundings) via heat or work. Think of surrounding the system with a perfect, rigid insulating blanket. -

Section 15-6: Thermodynamic Cycles
Answer to Essential Question 15.5: The ideal gas law tells us that temperature is proportional to PV. for state 2 in both processes we are considering, so the temperature in state 2 is the same in both cases. , and all three factors on the right-hand side are the same for the two processes, so the change in internal energy is the same (+360 J, in fact). Because the gas does no work in the isochoric process, and a positive amount of work in the isobaric process, the First Law tells us that more heat is required for the isobaric process (+600 J versus +360 J). 15-6 Thermodynamic Cycles Many devices, such as car engines and refrigerators, involve taking a thermodynamic system through a series of processes before returning the system to its initial state. Such a cycle allows the system to do work (e.g., to move a car) or to have work done on it so the system can do something useful (e.g., removing heat from a fridge). Let’s investigate this idea. EXPLORATION 15.6 – Investigate a thermodynamic cycle One cycle of a monatomic ideal gas system is represented by the series of four processes in Figure 15.15. The process taking the system from state 4 to state 1 is an isothermal compression at a temperature of 400 K. Complete Table 15.1 to find Q, W, and for each process, and for the entire cycle. Process Special process? Q (J) W (J) (J) 1 ! 2 No +1360 2 ! 3 Isobaric 3 ! 4 Isochoric 0 4 ! 1 Isothermal 0 Entire Cycle No 0 Table 15.1: Table to be filled in to analyze the cycle. -

Thermodynamics I - Enthalpy
CHEM 2880 - Kinetics Thermodynamics I - Enthalpy Tinoco Chapter 2 Secondary Reference: J.B. Fenn, Engines, Energy and Entropy, Global View Publishing, Pittsburgh, 2003. 1 CHEM 2880 - Kinetics Thermodynamics • An essential foundation for understanding physical and biological sciences. • Relationships and interconversions between various forms of energy (mechanical, electrical, chemical, heat, etc) • An understanding of the maximum efficiency with which we can transform one form of energy into another • Preferred direction by which a system evolves, i.e. will a conversion (reaction) go or not • Understanding of equilibrium • It is not based on the ideas of molecules or atoms. A linkage between these and thermo can be achieved using statistical methods. • It does not tell us about the rate of a process (how fast). Domain of kinetics. 2 CHEM 2880 - Kinetics Surroundings, boundaries, system When considering energy relationships it is important to define your point of reference. 3 CHEM 2880 - Kinetics Types of Systems Open: both mass and Closed: energy can be energy may leave and enter exchanged no matter can enter or leave Isolated: neither mass nor energy can enter or leave. 4 CHEM 2880 - Kinetics Energy Transfer Energy can be transferred between the system and the surroundings as heat (q) or work (w). This leads to a change in the internal energy (E or U) of the system. Heat • the energy transfer that occurs when two bodies at different temperatures come in contact with each other - the hotter body tends to cool while the cooler one warms -

Thermodynamics and Phase Diagrams
Thermodynamics and Phase Diagrams Arthur D. Pelton Centre de Recherche en Calcul Thermodynamique (CRCT) École Polytechnique de Montréal C.P. 6079, succursale Centre-ville Montréal QC Canada H3C 3A7 Tél : 1-514-340-4711 (ext. 4531) Fax : 1-514-340-5840 E-mail : [email protected] (revised Aug. 10, 2011) 2 1 Introduction An understanding of phase diagrams is fundamental and essential to the study of materials science, and an understanding of thermodynamics is fundamental to an understanding of phase diagrams. A knowledge of the equilibrium state of a system under a given set of conditions is the starting point in the description of many phenomena and processes. A phase diagram is a graphical representation of the values of the thermodynamic variables when equilibrium is established among the phases of a system. Materials scientists are most familiar with phase diagrams which involve temperature, T, and composition as variables. Examples are T-composition phase diagrams for binary systems such as Fig. 1 for the Fe-Mo system, isothermal phase diagram sections of ternary systems such as Fig. 2 for the Zn-Mg-Al system, and isoplethal (constant composition) sections of ternary and higher-order systems such as Fig. 3a and Fig. 4. However, many useful phase diagrams can be drawn which involve variables other than T and composition. The diagram in Fig. 5 shows the phases present at equilibrium o in the Fe-Ni-O2 system at 1200 C as the equilibrium oxygen partial pressure (i.e. chemical potential) is varied. The x-axis of this diagram is the overall molar metal ratio in the system. -

Isothermal Process It Is the Process in Which Other Physical Quantities Might Change but the Temperature of the System Remains Or Is Forced to Remain Constant
Sajit Chandra Shakya Department of Physics Kathmandu Don Bosco College New Baneshwor, Kathmandu Isothermal process It is the process in which other physical quantities might change but the temperature of the system remains or is forced to remain constant. For example, the constant temperature of human body. Under constant temperature, the volume of a gas system is inversely proportional to the pressure applied, the phenomena being called Boyle's Law, written in symbols as 1 V ∝ P 1 Or, V = KB× where KB is a constant quantity. P Or, PV = KB …………….. (i) This means whatever be the values of volume and pressure, their product will be constant. So, P1V1 = KB, P2V2 = KB, P3V3 = KB, etc, Or, P1V1 = P2V2 = P3V3, etc. The requirements for an isothermal process are as follows: 1. The process should be carried very slowly so that there is an ample time for compensation of heat in case of any loss or addition. 2. The boundaries of the system should be highly conducting so that there is a path for heat to flow into or flow away from a closed space in case of any energy loss or oversupply. 3. The boundaries should be made very thin because the resistance of the substance for heat conduction will be less for thin boundaries. Since in an isothermal change, the temperature remains constant, the internal energy also does not change, i.e. dU = 0. So if dQ amount of heat is given to a system which undergoes isothermal change, the relation for the first law of thermodynamics would be dQ = dU + dW Or, dQ = 0 + PdV Or, dQ = PdV This means all the heat supplied will be utilized for performing external work and consequently its value will be very high compared to other processes. -

Thermodynamics for Cryogenics
Thermodynamics for Cryogenics Ram C. Dhuley USPAS – Cryogenic Engineering June 21 – July 2, 2021 Goals of this lecture • Revise important definitions in thermodynamics – system and surrounding, state properties, derived properties, processes • Revise laws of thermodynamics and learn how to apply them to cryogenic systems • Learn about ‘ideal’ thermodynamic processes and their application to cryogenic systems • Prepare background for analyzing real-world helium liquefaction and refrigeration cycles. 2 Ram C. Dhuley | USPAS Cryogenic Engineering (Jun 21 - Jul 2, 2021) Introduction • Thermodynamics deals with the relations between heat and other forms of energy such as mechanical, electrical, or chemical energy. • Thermodynamics has its basis in attempts to understand combustion and steam power but is still “state of the art” in terms of practical engineering issues for cryogenics, especially in process efficiency. James Dewar (invented vacuum flask in 1892) https://physicstoday.scitation.org/doi/10.1063/1.881490 3 Ram C. Dhuley | USPAS Cryogenic Engineering (Jun 21 - Jul 2, 2021) Definitions • Thermodynamic system is the specified region in which heat, work, and mass transfer are being studied. • Surrounding is everything else other than the system. • Thermodynamic boundary is a surface separating the system from the surrounding. Boundary System Surrounding 4 Ram C. Dhuley | USPAS Cryogenic Engineering (Jun 21 - Jul 2, 2021) Definitions (continued) Types of thermodynamic systems • Isolated system has no exchange of mass or energy with its surrounding • Closed system exchanges heat but not mass • Open system exchanges both heat and mass with its surrounding Thermodynamic State • Thermodynamic state is the condition of the system at a given time, defined by ‘state’ properties • More details on next slides • Two state properties define a state of a “homogenous” system • Usually, three state properties are needed to define a state of a non-homogenous system (example: two phase, mixture) 5 Ram C. -

The First Law of Thermodynamics Continued Pre-Reading: §19.5 Where We Are
Lecture 7 The first law of thermodynamics continued Pre-reading: §19.5 Where we are The pressure p, volume V, and temperature T are related by an equation of state. For an ideal gas, pV = nRT = NkT For an ideal gas, the temperature T is is a direct measure of the average kinetic energy of its 3 3 molecules: KE = nRT = NkT tr 2 2 2 3kT 3RT and vrms = (v )av = = r m r M p Where we are We define the internal energy of a system: UKEPE=+∑∑ interaction Random chaotic between atoms motion & molecules For an ideal gas, f UNkT= 2 i.e. the internal energy depends only on its temperature Where we are By considering adding heat to a fixed volume of an ideal gas, we showed f f Q = Nk∆T = nR∆T 2 2 and so, from the definition of heat capacity Q = nC∆T f we have that C = R for any ideal gas. V 2 Change in internal energy: ∆U = nCV ∆T Heat capacity of an ideal gas Now consider adding heat to an ideal gas at constant pressure. By definition, Q = nCp∆T and W = p∆V = nR∆T So from ∆U = Q W − we get nCV ∆T = nCp∆T nR∆T − or Cp = CV + R It takes greater heat input to raise the temperature of a gas a given amount at constant pressure than constant volume YF §19.4 Ratio of heat capacities Look at the ratio of these heat capacities: we have f C = R V 2 and f + 2 C = C + R = R p V 2 so C p γ = > 1 CV 3 For a monatomic gas, CV = R 3 5 2 so Cp = R + R = R 2 2 C 5 R 5 and γ = p = 2 = =1.67 C 3 R 3 YF §19.4 V 2 Problem An ideal gas is enclosed in a cylinder which has a movable piston. -

Chap. 2. the First Law of Thermodynamics� Law of Energy Conservation
Chap. 2. The First Law of Thermodynamics! Law of Energy Conservation! System - part of the world we are interest in ! Surroundings - region outside the system! World or Universe - system plus surroundings! Open system - transfer of matter between system and surroundings! Closed system - no transfer of matter! Isolated system - closed, no mechanical and thermal contact ! Internal energy of system work done on system! (initial)! Internal energy of system Heat transferred to system! (final)! First law holds however small the heat and work are.! Infinitesimal changes:! work on system due to expansion or contraction! electric or other work on system ! First law holds however small the heat and work are.! Infinitesimal changes:! work on system due to expansion or contraction! electric or other work on system! At constant volume,! Heat capacity at constant volume - the amount of heat transferred to the system per unit increase of temperature! First law holds however small the heat and work are.! Infinitesimal changes:! work due to expansion or contraction! electric or other work! At constant volume,! Heat capacity at constant volume - the amount of heat transferred to the system per unit increase of temperature! Most processes occur at constant pressure. What is the relation between heat absorbed at constant pressure and the energy?! Enthalpy:! heat content, a state function in the unit of energy! The heat absorbed at constant pressure is the same as enthalpy change of the system given that its pressure is the same as the external pressure.! Enthalpy:! -

Second Law Analysis of Rankine Cycle
Chapter 10 Lyes KADEM [Thermodynamics II] 2007 Refrigeration Cycles The objective of refrigeration cycles is to transfer the heat from a low temperature region to a high temperature region. - if the objective of the cycle is to decrease the lowest temperature, we call it: a Refrigeration cycle. - if the objective of the cycle is to increase the highest temperature, we call it: a heat pump. Carnot cycle As the Carnot cycle is the ideal cycle, let us first try to convert it to a refrigeration cycle, and then we will discuss practical limitations. Figure (10.1) shows a reversed Carnot cycle, the efficiency of this cycle will be: TL η 1−= TH However, if we try to apply the Carnot cycle, we will face some technical difficulties: 1-2: It is not advisable to compress a mixture of vapor and gas, since the liquid droplets would cause excessive wear. → shift point 1 to the right (saturated vapor). 3-4: It would be quite expensive to construct a device to be used in the expansion process that would be nearly isentropic (no losses allowed). It is much simpler to reduce the pressure irreversibly by using an expansion valve (isenthalpic process). Refrigeration cycles 44 Chapter 10 Lyes KADEM [Thermodynamics II] 2007 Figure.10.1. Reversed Carnot cycle. The Ideal vapor-compression refrigeration cycle 1-2: isentropic compression in a compressor. 2-3: isobaric heat rejection in a condenser. 3-4: throttling in an expansion device. 4-1: isobaric heat absorption in an evaporator. Figure.10.2. Ideal vapor- refrigeration cycle. Refrigeration cycles 45 Chapter 10 Lyes KADEM [Thermodynamics II] 2007 The coefficient of performance (COP) of a refrigeration cycle is: benefit Q& in COPR == Cost W&in The coefficient of performance (COP) of a heat pump is: benefit Q&out COPHP == Cost W&in COP can attain 4 for refrigerators and perhaps 5 for heat pumps.