Phenolic Compounds and Bioactive Properties of Ruscus Aculeatus L
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Redalyc.Pharmacognostical and Phytochemical Studies on Ziziphora
Journal of Pharmacy & Pharmacognosy Research E-ISSN: 0719-4250 [email protected] Asociación de Académicos de Ciencias Farmacéuticas de Antofagasta Chile Zhu, Yun; Xiong, Yuan; Wang, Hehua; Li, Peng Pharmacognostical and phytochemical studies on Ziziphora clinopodioides Lam. – A Kazakh and Uygur ethnomedicinal plant Journal of Pharmacy & Pharmacognosy Research, vol. 5, núm. 6, noviembre-diciembre, 2017, pp. 354-364 Asociación de Académicos de Ciencias Farmacéuticas de Antofagasta Antofagasta, Chile Available in: http://www.redalyc.org/articulo.oa?id=496053946004 How to cite Complete issue Scientific Information System More information about this article Network of Scientific Journals from Latin America, the Caribbean, Spain and Portugal Journal's homepage in redalyc.org Non-profit academic project, developed under the open access initiative © 2017 Journal of Pharmacy & Pharmacognosy Research, 5 (6), 354-364, 2017 ISSN 0719-4250 http://jppres.com/jppres Original Article | Artículo Original Pharmacognostical and phytochemical studies on Ziziphora clinopodioides Lam. – A Kazakh and Uygur ethnomedicinal plant [Estudios farmacognósticos y fitoquímicos sobre Ziziphora clinopodioides Lam. - Una planta etnomedicinal kazaja y uygur] Yun Zhu1, Yuan Xiong1, Hehua Wang2, Peng Li1* 1School of Pharmacy, Shihezi University/Key Laboratory of Xinjiang Endemic Phytomedicine Resources Ministry of Education, Shihezi Xinjiang 832002, PR China. 2Hebi City People's Hospital Infection Management Section, Hebi Henan 458030, PR China. *E-mail: [email protected] Abstract Resumen Context: Ziziphora clinopodioides Lam. (Lamiaceae) is an annual or Contexto: Ziziphora clinopodioides Lam. (Lamiaceae) es una hierba anual perennial herb or subshrub widely distributed from the Mediterranean to o perenne o arbusto ampliamente distribuida desde el Mediterráneo a central Asia and Afghanistan. In Xinjiang, China, the whole herb has been Asia central y Afganistán. -

Alyssum) and the Correct Name of the Goldentuft Alyssum
ARNOLDIA VE 1 A continuation of the BULLETIN OF POPULAR INFORMATION of the Arnold Arboretum, Harvard University VOLUME 26 JUNE 17, 1966 NUMBERS 6-7 ORNAMENTAL MADWORTS (ALYSSUM) AND THE CORRECT NAME OF THE GOLDENTUFT ALYSSUM of the standard horticultural reference works list the "Madworts" as MANYa group of annuals, biennials, perennials or subshrubs in the family Cru- ciferae, which with the exception of a few species, including the goldentuft mad- wort, are not widely cultivated. The purposes of this article are twofold. First, to inform interested gardeners, horticulturists and plantsmen that this exception, with a number of cultivars, does not belong to the genus Alyssum, but because of certain critical and technical characters, should be placed in the genus Aurinia of the same family. The second goal is to emphasize that many species of the "true" .~lyssum are notable ornamentals and merit greater popularity and cul- tivation. The genus Alyssum (now containing approximately one hundred and ninety species) was described by Linnaeus in 1753 and based on A. montanum, a wide- spread European species which is cultivated to a limited extent only. However, as medicinal and ornamental garden plants the genus was known in cultivation as early as 1650. The name Alyssum is of Greek derivation : a meaning not, and lyssa alluding to madness, rage or hydrophobia. Accordingly, the names Mad- wort and Alyssum both refer to the plant’s reputation as an officinal herb. An infu- sion concocted from the leaves and flowers was reputed to have been administered as a specific antidote against madness or the bite of a rabid dog. -

Outline of Angiosperm Phylogeny
Outline of angiosperm phylogeny: orders, families, and representative genera with emphasis on Oregon native plants Priscilla Spears December 2013 The following listing gives an introduction to the phylogenetic classification of the flowering plants that has emerged in recent decades, and which is based on nucleic acid sequences as well as morphological and developmental data. This listing emphasizes temperate families of the Northern Hemisphere and is meant as an overview with examples of Oregon native plants. It includes many exotic genera that are grown in Oregon as ornamentals plus other plants of interest worldwide. The genera that are Oregon natives are printed in a blue font. Genera that are exotics are shown in black, however genera in blue may also contain non-native species. Names separated by a slash are alternatives or else the nomenclature is in flux. When several genera have the same common name, the names are separated by commas. The order of the family names is from the linear listing of families in the APG III report. For further information, see the references on the last page. Basal Angiosperms (ANITA grade) Amborellales Amborellaceae, sole family, the earliest branch of flowering plants, a shrub native to New Caledonia – Amborella Nymphaeales Hydatellaceae – aquatics from Australasia, previously classified as a grass Cabombaceae (water shield – Brasenia, fanwort – Cabomba) Nymphaeaceae (water lilies – Nymphaea; pond lilies – Nuphar) Austrobaileyales Schisandraceae (wild sarsaparilla, star vine – Schisandra; Japanese -

ORNAMENTAL GARDEN PLANTS of the GUIANAS: an Historical Perspective of Selected Garden Plants from Guyana, Surinam and French Guiana
f ORNAMENTAL GARDEN PLANTS OF THE GUIANAS: An Historical Perspective of Selected Garden Plants from Guyana, Surinam and French Guiana Vf•-L - - •• -> 3H. .. h’ - — - ' - - V ' " " - 1« 7-. .. -JZ = IS^ X : TST~ .isf *“**2-rt * * , ' . / * 1 f f r m f l r l. Robert A. DeFilipps D e p a r t m e n t o f B o t a n y Smithsonian Institution, Washington, D.C. \ 1 9 9 2 ORNAMENTAL GARDEN PLANTS OF THE GUIANAS Table of Contents I. Map of the Guianas II. Introduction 1 III. Basic Bibliography 14 IV. Acknowledgements 17 V. Maps of Guyana, Surinam and French Guiana VI. Ornamental Garden Plants of the Guianas Gymnosperms 19 Dicotyledons 24 Monocotyledons 205 VII. Title Page, Maps and Plates Credits 319 VIII. Illustration Credits 321 IX. Common Names Index 345 X. Scientific Names Index 353 XI. Endpiece ORNAMENTAL GARDEN PLANTS OF THE GUIANAS Introduction I. Historical Setting of the Guianan Plant Heritage The Guianas are embedded high in the green shoulder of northern South America, an area once known as the "Wild Coast". They are the only non-Latin American countries in South America, and are situated just north of the Equator in a configuration with the Amazon River of Brazil to the south and the Orinoco River of Venezuela to the west. The three Guianas comprise, from west to east, the countries of Guyana (area: 83,000 square miles; capital: Georgetown), Surinam (area: 63, 037 square miles; capital: Paramaribo) and French Guiana (area: 34, 740 square miles; capital: Cayenne). Perhaps the earliest physical contact between Europeans and the present-day Guianas occurred in 1500 when the Spanish navigator Vincente Yanez Pinzon, after discovering the Amazon River, sailed northwest and entered the Oyapock River, which is now the eastern boundary of French Guiana. -

Chapter 5: the Shoot System I: the Stem
Chapter 5 The Shoot System I: The Stem THE FUNCTIONS AND ORGANIZATION OF THE SHOOT SYSTEM PRIMARY GROWTH AND STEM ANATOMY Primary Tissues of Dicot Stems Develop from the Primary Meristems The Distribution of the Primary Vascular Bundles Depends on the Position of Leaves Primary Growth Differs in Monocot and Dicot Stems SECONDARY GROWTH AND THE ANATOMY OF WOOD Secondary Xylem and Phloem Develop from Vascular Cambium Wood Is Composed of Secondary Xylem Gymnosperm Wood Differs from Angiosperm Wood Bark Is Composed of Secondary Phloem and Periderm Buds Are Compressed Branches Waiting to Elongate Some Monocot Stems Have Secondary Growth STEM MODIFICATIONS FOR SPECIAL FUNCTIONS THE ECONOMIC VALUE OF WOODY STEMS SUMMARY ECONOMIC BOTANY: How Do You Make A Barrel? 1 KEY CONCEPTS 1. The shoot system is composed of the stem and its lateral appendages: leaves, buds, and flowers. Leaves are arranged in different patterns (phyllotaxis): alternate, opposite, whorled, and spiral. 2. Stems provide support to the leaves, buds, and flowers. They conduct water and nutrients and produce new cells in meristems (shoot apical meristem, primary and secondary meristems). 3. Dicot stems and monocot stems are usually different. Dicot stems tend to have vascular bundles distributed in a ring, whereas in monocot stems they tend to be scattered. 4. Stems are composed of the following: epidermis, cortex and pith, xylem and phloem, and periderm. 5. Secondary xylem is formed by the division of cells in the vascular cambium and is called wood. The bark is composed of all of the tissues outside the vascular cambium, including the periderm (formed from cork cambium) and the secondary phloem. -

GENOME EVOLUTION in MONOCOTS a Dissertation
GENOME EVOLUTION IN MONOCOTS A Dissertation Presented to The Faculty of the Graduate School At the University of Missouri In Partial Fulfillment Of the Requirements for the Degree Doctor of Philosophy By Kate L. Hertweck Dr. J. Chris Pires, Dissertation Advisor JULY 2011 The undersigned, appointed by the dean of the Graduate School, have examined the dissertation entitled GENOME EVOLUTION IN MONOCOTS Presented by Kate L. Hertweck A candidate for the degree of Doctor of Philosophy And hereby certify that, in their opinion, it is worthy of acceptance. Dr. J. Chris Pires Dr. Lori Eggert Dr. Candace Galen Dr. Rose‐Marie Muzika ACKNOWLEDGEMENTS I am indebted to many people for their assistance during the course of my graduate education. I would not have derived such a keen understanding of the learning process without the tutelage of Dr. Sandi Abell. Members of the Pires lab provided prolific support in improving lab techniques, computational analysis, greenhouse maintenance, and writing support. Team Monocot, including Dr. Mike Kinney, Dr. Roxi Steele, and Erica Wheeler were particularly helpful, but other lab members working on Brassicaceae (Dr. Zhiyong Xiong, Dr. Maqsood Rehman, Pat Edger, Tatiana Arias, Dustin Mayfield) all provided vital support as well. I am also grateful for the support of a high school student, Cady Anderson, and an undergraduate, Tori Docktor, for their assistance in laboratory procedures. Many people, scientist and otherwise, helped with field collections: Dr. Travis Columbus, Hester Bell, Doug and Judy McGoon, Julie Ketner, Katy Klymus, and William Alexander. Many thanks to Barb Sonderman for taking care of my greenhouse collection of many odd plants brought back from the field. -

Listado De Todas Las Plantas Que Tengo Fotografiadas Ordenado Por Familias Según El Sistema APG III (Última Actualización: 2 De Septiembre De 2021)
Listado de todas las plantas que tengo fotografiadas ordenado por familias según el sistema APG III (última actualización: 2 de Septiembre de 2021) GÉNERO Y ESPECIE FAMILIA SUBFAMILIA GÉNERO Y ESPECIE FAMILIA SUBFAMILIA Acanthus hungaricus Acanthaceae Acanthoideae Metarungia longistrobus Acanthaceae Acanthoideae Acanthus mollis Acanthaceae Acanthoideae Odontonema callistachyum Acanthaceae Acanthoideae Acanthus spinosus Acanthaceae Acanthoideae Odontonema cuspidatum Acanthaceae Acanthoideae Aphelandra flava Acanthaceae Acanthoideae Odontonema tubaeforme Acanthaceae Acanthoideae Aphelandra sinclairiana Acanthaceae Acanthoideae Pachystachys lutea Acanthaceae Acanthoideae Aphelandra squarrosa Acanthaceae Acanthoideae Pachystachys spicata Acanthaceae Acanthoideae Asystasia gangetica Acanthaceae Acanthoideae Peristrophe speciosa Acanthaceae Acanthoideae Barleria cristata Acanthaceae Acanthoideae Phaulopsis pulchella Acanthaceae Acanthoideae Barleria obtusa Acanthaceae Acanthoideae Pseuderanthemum carruthersii ‘Rubrum’ Acanthaceae Acanthoideae Barleria repens Acanthaceae Acanthoideae Pseuderanthemum carruthersii var. atropurpureum Acanthaceae Acanthoideae Brillantaisia lamium Acanthaceae Acanthoideae Pseuderanthemum carruthersii var. reticulatum Acanthaceae Acanthoideae Brillantaisia owariensis Acanthaceae Acanthoideae Pseuderanthemum laxiflorum Acanthaceae Acanthoideae Brillantaisia ulugurica Acanthaceae Acanthoideae Pseuderanthemum laxiflorum ‘Purple Dazzler’ Acanthaceae Acanthoideae Crossandra infundibuliformis Acanthaceae Acanthoideae Ruellia -
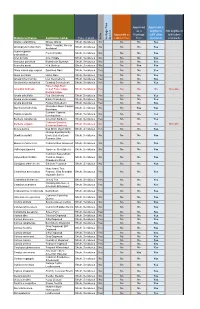
Botanical Name Common Name
Approved Approved & as a eligible to Not eligible to Approved as Frontage fulfill other fulfill other Type of plant a Street Tree Tree standards standards Heritage Tree Tree Heritage Species Botanical Name Common name Native Abelia x grandiflora Glossy Abelia Shrub, Deciduous No No No Yes White Forsytha; Korean Abeliophyllum distichum Shrub, Deciduous No No No Yes Abelialeaf Acanthropanax Fiveleaf Aralia Shrub, Deciduous No No No Yes sieboldianus Acer ginnala Amur Maple Shrub, Deciduous No No No Yes Aesculus parviflora Bottlebrush Buckeye Shrub, Deciduous No No No Yes Aesculus pavia Red Buckeye Shrub, Deciduous No No Yes Yes Alnus incana ssp. rugosa Speckled Alder Shrub, Deciduous Yes No No Yes Alnus serrulata Hazel Alder Shrub, Deciduous Yes No No Yes Amelanchier humilis Low Serviceberry Shrub, Deciduous Yes No No Yes Amelanchier stolonifera Running Serviceberry Shrub, Deciduous Yes No No Yes False Indigo Bush; Amorpha fruticosa Desert False Indigo; Shrub, Deciduous Yes No No No Not eligible Bastard Indigo Aronia arbutifolia Red Chokeberry Shrub, Deciduous Yes No No Yes Aronia melanocarpa Black Chokeberry Shrub, Deciduous Yes No No Yes Aronia prunifolia Purple Chokeberry Shrub, Deciduous Yes No No Yes Groundsel-Bush; Eastern Baccharis halimifolia Shrub, Deciduous No No Yes Yes Baccharis Summer Cypress; Bassia scoparia Shrub, Deciduous No No No Yes Burning-Bush Berberis canadensis American Barberry Shrub, Deciduous Yes No No Yes Common Barberry; Berberis vulgaris Shrub, Deciduous No No No No Not eligible European Barberry Betula pumila -

Unearthing Belowground Bud Banks in Fire-Prone Ecosystems
Unearthing belowground bud banks in fire-prone ecosystems 1 2 3 Author for correspondence: Juli G. Pausas , Byron B. Lamont , Susana Paula , Beatriz Appezzato-da- Juli G. Pausas 4 5 Glo'ria and Alessandra Fidelis Tel: +34 963 424124 1CIDE-CSIC, C. Naquera Km 4.5, Montcada, Valencia 46113, Spain; 2Department of Environment and Agriculture, Curtin Email [email protected] University, PO Box U1987, Perth, WA 6845, Australia; 3ICAEV, Universidad Austral de Chile, Campus Isla Teja, Casilla 567, Valdivia, Chile; 4Depto Ci^encias Biologicas,' Universidade de Sao Paulo, Av P'adua Dias 11., CEP 13418-900, Piracicaba, SP, Brazil; 5Instituto de Bioci^encias, Vegetation Ecology Lab, Universidade Estadual Paulista (UNESP), Av. 24-A 1515, 13506-900 Rio Claro, Brazil Summary To be published in New Phytologist (2018) Despite long-time awareness of the importance of the location of buds in plant biology, research doi: 10.1111/nph.14982 on belowground bud banks has been scant. Terms such as lignotuber, xylopodium and sobole, all referring to belowground bud-bearing structures, are used inconsistently in the literature. Key words: bud bank, fire-prone ecosystems, Because soil efficiently insulates meristems from the heat of fire, concealing buds below ground lignotuber, resprouting, rhizome, xylopodium. provides fitness benefits in fire-prone ecosystems. Thus, in these ecosystems, there is a remarkable diversity of bud-bearing structures. There are at least six locations where belowground buds are stored: roots, root crown, rhizomes, woody burls, fleshy -

Networks in a Large-Scale Phylogenetic Analysis: Reconstructing Evolutionary History of Asparagales (Lilianae) Based on Four Plastid Genes
Networks in a Large-Scale Phylogenetic Analysis: Reconstructing Evolutionary History of Asparagales (Lilianae) Based on Four Plastid Genes Shichao Chen1., Dong-Kap Kim2., Mark W. Chase3, Joo-Hwan Kim4* 1 College of Life Science and Technology, Tongji University, Shanghai, China, 2 Division of Forest Resource Conservation, Korea National Arboretum, Pocheon, Gyeonggi- do, Korea, 3 Jodrell Laboratory, Royal Botanic Gardens, Kew, Richmond, United Kingdom, 4 Department of Life Science, Gachon University, Seongnam, Gyeonggi-do, Korea Abstract Phylogenetic analysis aims to produce a bifurcating tree, which disregards conflicting signals and displays only those that are present in a large proportion of the data. However, any character (or tree) conflict in a dataset allows the exploration of support for various evolutionary hypotheses. Although data-display network approaches exist, biologists cannot easily and routinely use them to compute rooted phylogenetic networks on real datasets containing hundreds of taxa. Here, we constructed an original neighbour-net for a large dataset of Asparagales to highlight the aspects of the resulting network that will be important for interpreting phylogeny. The analyses were largely conducted with new data collected for the same loci as in previous studies, but from different species accessions and greater sampling in many cases than in published analyses. The network tree summarised the majority data pattern in the characters of plastid sequences before tree building, which largely confirmed the currently recognised phylogenetic relationships. Most conflicting signals are at the base of each group along the Asparagales backbone, which helps us to establish the expectancy and advance our understanding of some difficult taxa relationships and their phylogeny. -

In Vitro Cultivation of Ruscus Aculeatus L. and Ruscus Hypoglossum L
In vitro cultivation of Ruscus aculeatus L. and Ruscus hypoglossum L. (Liliaceae) Teodora A. Ivanova PhD thesis, Sofia, 2012 Institute of Biodiversity and Ecosystem Research, Bulgarian Academy of Sciences; 23 Acad. G. Bonchev Str., Sofia 1000, Bulgaria; e-mail: [email protected]; PhD supervisor: Assoc. Prof. Tatyana Stoeva Summary Ruscus aculeatus L. and R. hypoglossum (Liliaceae) are small evergreen shrubs used both in gardening and as cut foliage. The rhizomes of R. aculeatus are widely collected as a drug source of steroid saponins with antiinflammatory, venotonic and antihaemoroidal activity. Both species are considered conservationally important in several countries due to the fact that cut foliage and rhizomes have been collected predominantly from the wild. In Bulgaria a regulated regime of gathering was prescribed for R. aculeatus and R. hypoglossum. The natural slow reproduction cycle and specific culture requirements hinder wide field cultivation. Recently, in vitro cultivation of both species has been performed for the production of planting material and conservation purposes mainly with material of Spanish, Portuguese and Romanian origin. However many technological aspects remained unsolved and the micropropagation protocols contained unequivocal and unclear parameters. The aim of this thesis was to initiate in vitro cultures of R. aculeatus and R. hypoglossum from donor material of Bulgarian wild origin and to assess the influence of different culture factors on in vitro propagation and conservation: explant type, plant growth regulators and their concentration, culture type and sugar content. The characterization of the clonal variability and effect of culture conditions on plant genetic integrity was conducted by means of flow cytometry (DNA content and genome stability) and PAGE isozyme analyses of peroxidase (POD), asterase (EST), and acid phosphatase (ACP). -

Tamilnadu Board Class 11 Bio-Botany Chapter 3
Unit II: Plant Morphology and Taxonomy of Angiosperm Chapter 3 Vegetative Morphology Learning Objectives of shape, size and structure of plants and their parts (roots, stems, leaves, The learner will be able to, flowers, fruits and seeds). Study of • Explore the parts of the flowering morphology is important in taxonomy. plants Morphological features are important • Differentiate vegetative morphology in determining productivity of crops. and reproductive morphology Morphological characters indicate the specific habitats of living as well as the • Compare various root systems and fossil plants and help to correlate the their modifications distribution in space and time of fossil • Understand the stem modifications plants. Morphological features are also and functions significant for phylogeny. • Interpret the structure of leaf and functions of leaf Plant Morphology can be studied under two broad categories: A. Vegetative morphology – It Chapter Outline includes shoot system and root 3.1 Habit system 3.2 Plant habitat B. Reproductive morphology – It 3.3 Life Span includes Flower/inflorescence, Fruit and Seed 3.4 Parts of a flowering plant 3.5 Root System A. Vegetative morphology 3.6 Shoot system Vegetative morphology deals with the 3.7 Leaf study of shape, size and structure of plants and their parts roots, stems and The study of various external features of leaves. To understand the vegetative the organism is known as morphology. morphology the following important Plant morphology also known as components are to be studied. They are, external morphology deals with the study 1) Habit, 2) Habitat and 3) Lifespan. 63 TN_GOVT_BOTANY_XI_Page 063-086 CH03.indd 63 02-06-2018 13:48:22 3.1 Habit IV.