Structure of Prokaryotes: Bacteria and Archaea
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Review Pili in Gram-Negative and Gram-Positive Bacteria – Structure
Cell. Mol. Life Sci. 66 (2009) 613 – 635 1420-682X/09/040613-23 Cellular and Molecular Life Sciences DOI 10.1007/s00018-008-8477-4 Birkhuser Verlag, Basel, 2008 Review Pili in Gram-negative and Gram-positive bacteria – structure, assembly and their role in disease T. Profta,c,* and E. N. Bakerb,c a School of Medical Sciences, Department of Molecular Medicine & Pathology, University of Auckland, Private Bag 92019, Auckland 1142 (New Zealand), Fax: +64-9-373-7492, e-mail: [email protected] b School of Biological Sciences, University of Auckland, Auckland (New Zealand) c Maurice Wilkins Centre for Molecular Biodiscovery, University of Auckland (New Zealand) Received 08 August 2008; received after revision 24 September 2008; accepted 01 October 2008 Online First 27 October 2008 Abstract. Many bacterial species possess long fila- special form of bacterial cell movement, known as mentous structures known as pili or fimbriae extend- twitching motility. In contrast, the more recently ing from their surfaces. Despite the diversity in pilus discovered pili in Gram-positive bacteria are formed structure and biogenesis, pili in Gram-negative bac- by covalent polymerization of pilin subunits in a teria are typically formed by non-covalent homopo- process that requires a dedicated sortase enzyme. lymerization of major pilus subunit proteins (pilins), Minor pilins are added to the fiber and play a major which generates the pilus shaft. Additional pilins may role in host cell colonization. be added to the fiber and often function as host cell This review gives an overview of the structure, adhesins. Some pili are also involved in biofilm assembly and function of the best-characterized pili formation, phage transduction, DNA uptake and a of both Gram-negative and Gram-positive bacteria. -
Prokaryotes (Domains Bacteria & Archaea)
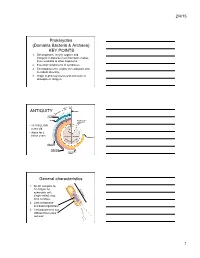
2/4/15 Prokaryotes (Domains Bacteria & Archaea) KEY POINTS 1. Decomposers: recycle organic and inorganic molecules in environment; makes them available to other organisms. 2. Essential components of symbioses. 3. Encompasses the origins of metabolism and metabolic diversity. 4. Origin of photosynthesis and formation of atmospheric Oxygen Ceno- Meso- zoic zoic ANTIQUITY Humans Paleozoic Colonization of land Animals Origin of solar system and Earth • >3.5 BILLION years old. • Alone for 2 1 4 billion years Proterozoic Archaean Prokaryotes Billions of 2 years ago3 Multicellular eukaryotes Single-celled eukaryotes Atmospheric oxygen General characteristics 1. Small: compare to 10-100µm for 0.5-5µm eukaryotic cell; single-celled; may form colonies. 2. Lack membrane- enclosed organelles. 3. Cell wall present, but different from plant cell wall. 1 2/4/15 General characteristics 4. Occur everywhere, most numerous organisms. – More individuals in a handful of soil then there are people that have ever lived. – By far more individuals in our gut than eukaryotic cells that are actually us. General characteristics 5. Metabolic diversity established nutritional modes of eukaryotes. General characteristics 6. Important decomposers and recyclers 2 2/4/15 General characteristics 6. Important decomposers and recyclers • Form the basis of global nutrient cycles. General characteristics 7. Symbionts!!!!!!! • Parasites • Pathogenic organisms. • About 1/2 of all human diseases are caused by Bacteria General characteristics 7. Symbionts!!!!!!! • Parasites • Pathogenic organisms. • Extremely important in agriculture as well. Pierce’s disease is caused by Xylella fastidiosa, a Gamma Proteobacteria. It causes over $56 million in damage annually in California. That’s with $34 million spent to control it! = $90 million in California alone. -

Battistuzzi2009chap07.Pdf
Eubacteria Fabia U. Battistuzzia,b,* and S. Blair Hedgesa shown increasing support for lower-level phylogenetic Department of Biology, 208 Mueller Laboratory, The Pennsylvania clusters (e.g., classes and below), they have also shown the State University, University Park, PA 16802-5301, USA; bCurrent susceptibility of eubacterial phylogeny to biases such as address: Center for Evolutionary Functional Genomics, The Biodesign horizontal gene transfer (HGT) (20, 21). Institute, Arizona State University, Tempe, AZ 85287-5301, USA In recent years, three major approaches have been used *To whom correspondence should be addressed (Fabia.Battistuzzi@ asu.edu) for studying prokaryote phylogeny with data from com- plete genomes: (i) combining gene sequences in a single analysis of multiple genes (e.g., 7, 9, 10), (ii) combining Abstract trees from individual gene analyses into a single “super- tree” (e.g., 22, 23), and (iii) using the presence or absence The ~9400 recognized species of prokaryotes in the of genes (“gene content”) as the raw data to investigate Superkingdom Eubacteria are placed in 25 phyla. Their relationships (e.g., 17, 18). While the results of these dif- relationships have been diffi cult to establish, although ferent approaches have not agreed on many details of some major groups are emerging from genome analyses. relationships, there have been some points of agreement, A molecular timetree, estimated here, indicates that most such as support for the monophyly of all major classes (85%) of the phyla and classes arose in the Archean Eon and some phyla (e.g., Proteobacteria and Firmicutes). (4000−2500 million years ago, Ma) whereas most (95%) of 7 ese A ndings, although criticized by some (e.g., 24, 25), the families arose in the Proterozoic Eon (2500−542 Ma). -

Review Article: Inhibition of Methanogenic Archaea by Statins As a Targeted Management Strategy for Constipation and Related Disorders
Alimentary Pharmacology and Therapeutics Review article: inhibition of methanogenic archaea by statins as a targeted management strategy for constipation and related disorders K. Gottlieb*, V. Wacher*, J. Sliman* & M. Pimentel† *Synthetic Biologics, Inc., Rockville, SUMMARY MD, USA. † Gastroenterology, Cedars-Sinai Background Medical Center, Los Angeles, CA, USA. Observational studies show a strong association between delayed intestinal transit and the production of methane. Experimental data suggest a direct inhibitory activity of methane on the colonic and ileal smooth muscle and Correspondence to: a possible role for methane as a gasotransmitter. Archaea are the only con- Dr K. Gottlieb, Synthetic Biologics, fi Inc., 9605 Medical Center Drive, rmed biological sources of methane in nature and Methanobrevibacter Rockville, MD 20850, USA. smithii is the predominant methanogen in the human intestine. E-mail: [email protected] Aim To review the biosynthesis and composition of archaeal cell membranes, Publication data archaeal methanogenesis and the mechanism of action of statins in this context. Submitted 8 September 2015 First decision 29 September 2015 Methods Resubmitted 7 October 2015 Narrative review of the literature. Resubmitted 20 October 2015 Accepted 20 October 2015 Results EV Pub Online 11 November 2015 Statins can inhibit archaeal cell membrane biosynthesis without affecting This uncommissioned review article was bacterial numbers as demonstrated in livestock and humans. This opens subject to full peer-review. the possibility of a therapeutic intervention that targets a specific aetiologi- cal factor of constipation while protecting the intestinal microbiome. While it is generally believed that statins inhibit methane production via their effect on cell membrane biosynthesis, mediated by inhibition of the HMG- CoA reductase, there is accumulating evidence for an alternative or addi- tional mechanism of action where statins inhibit methanogenesis directly. -

The First Cells Were Most Likely Very Simple Prokaryotic Forms. Ra- Spirochetes
T HE O RIGIN OF E UKARYOTIC C ELLS The first cells were most likely very simple prokaryotic forms. Ra- spirochetes. Ingestion of prokaryotes that resembled present-day diometric dating indicates that the earth is 4 to 5 billion years old cyanobacteria could have led to the endosymbiotic development of and that prokaryotes may have arisen more than 3.5 billion years chloroplasts in plants. ago. Eukaryotes are thought to have first appeared about 1.5 billion Another hypothesis for the evolution of eukaryotic cells proposes years ago. that the prokaryotic cell membrane invaginated (folded inward) to en- The eukaryotic cell might have evolved when a large anaerobic close copies of its genetic material (figure 1b). This invagination re- (living without oxygen) amoeboid prokaryote ingested small aerobic (liv- sulted in the formation of several double-membrane-bound entities ing with oxygen) bacteria and stabilized them instead of digesting them. (organelles) in a single cell. These entities could then have evolved This idea is known as the endosymbiont hypothesis (figure 1a) and into the eukaryotic mitochondrion, nucleus, and chloroplasts. was first proposed by Lynn Margulis, a biologist at Boston Univer- Although the exact mechanism for the evolution of the eu- sity. (Symbiosis is an intimate association between two organisms karyotic cell will never be known with certainty, the emergence of of different species.) According to this hypothesis, the aerobic bac- the eukaryotic cell led to a dramatic increase in the complexity and teria developed into mitochondria, which are the sites of aerobic diversity of life-forms on the earth. At first, these newly formed eu- respiration and most energy conversion in eukaryotic cells. -

Microbiology of Endodontic Infections
Scient Open Journal of Dental and Oral Health Access Exploring the World of Science ISSN: 2369-4475 Short Communication Microbiology of Endodontic Infections This article was published in the following Scient Open Access Journal: Journal of Dental and Oral Health Received August 30, 2016; Accepted September 05, 2016; Published September 12, 2016 Harpreet Singh* Abstract Department of Conservative Dentistry & Endodontics, Gian Sagar Dental College, Patiala, Punjab, India Root canal system acts as a ‘privileged sanctuary’ for the growth and survival of endodontic microbiota. This is attributed to the special environment which the microbes get inside the root canals and several other associated factors. Although a variety of microbes have been isolated from the root canal system, bacteria are the most common ones found to be associated with Endodontic infections. This article gives an in-depth view of the microbiology involved in endodontic infections during its different stages. Keywords: Bacteria, Endodontic, Infection, Microbiology Introduction Microorganisms play an unequivocal role in infecting root canal system. Endodontic infections are different from the other oral infections in the fact that they occur in an environment which is closed to begin with since the root canal system is an enclosed one, surrounded by hard tissues all around [1,2]. Most of the diseases of dental pulp and periradicular tissues are associated with microorganisms [3]. Endodontic infections occur and progress when the root canal system gets exposed to the oral environment by one reason or the other and simultaneously when there is fall in the body’s immune when the ingress is from a carious lesion or a traumatic injury to the coronal tooth structure.response [4].However, To begin the with, issue the if notmicrobes taken arecare confined of, ultimately to the leadsintra-radicular to the egress region of pathogensIn total, and bacteria their by-productsdetected from from the the oral apical cavity foramen fall into to 13 the separate periradicular phyla, tissues. -

Fatty Acid Diets: Regulation of Gut Microbiota Composition and Obesity and Its Related Metabolic Dysbiosis
International Journal of Molecular Sciences Review Fatty Acid Diets: Regulation of Gut Microbiota Composition and Obesity and Its Related Metabolic Dysbiosis David Johane Machate 1, Priscila Silva Figueiredo 2 , Gabriela Marcelino 2 , Rita de Cássia Avellaneda Guimarães 2,*, Priscila Aiko Hiane 2 , Danielle Bogo 2, Verônica Assalin Zorgetto Pinheiro 2, Lincoln Carlos Silva de Oliveira 3 and Arnildo Pott 1 1 Graduate Program in Biotechnology and Biodiversity in the Central-West Region of Brazil, Federal University of Mato Grosso do Sul, Campo Grande 79079-900, Brazil; [email protected] (D.J.M.); [email protected] (A.P.) 2 Graduate Program in Health and Development in the Central-West Region of Brazil, Federal University of Mato Grosso do Sul, Campo Grande 79079-900, Brazil; pri.fi[email protected] (P.S.F.); [email protected] (G.M.); [email protected] (P.A.H.); [email protected] (D.B.); [email protected] (V.A.Z.P.) 3 Chemistry Institute, Federal University of Mato Grosso do Sul, Campo Grande 79079-900, Brazil; [email protected] * Correspondence: [email protected]; Tel.: +55-67-3345-7416 Received: 9 March 2020; Accepted: 27 March 2020; Published: 8 June 2020 Abstract: Long-term high-fat dietary intake plays a crucial role in the composition of gut microbiota in animal models and human subjects, which affect directly short-chain fatty acid (SCFA) production and host health. This review aims to highlight the interplay of fatty acid (FA) intake and gut microbiota composition and its interaction with hosts in health promotion and obesity prevention and its related metabolic dysbiosis. -

World Journal of Clinical Cases
World Journal of W J C C Clinical Cases Submit a Manuscript: http://www.f6publishing.com World J Clin Cases 2018 April 16; 6(4): 54-63 DOI: 10.12998/wjcc.v6.i4.54 ISSN 2307-8960 (online) ORIGINAL ARTICLE Observational Study Correlations between microbial communities in stool and clinical indicators in patients with metabolic syndrome Lang Lin, Zai-Bo Wen, Dong-Jiao Lin, Jiang-Ting Dong, Jie Jin, Fei Meng Lang Lin, Zai-Bo Wen, Dong-Jiao Lin, Jiang-Ting Dong, the use is non-commercial. See: http://creativecommons.org/ Department of Gastroenterology, Cangnan People’s Hospital, licenses/by-nc/4.0/ Cangnan 325800, Zhejiang Province, China Manuscript source: Unsolicited manuscript Jie Jin, Fei Meng, Department of Research Service, Zhiyuan Medical Inspection Institute CO., LTD, Hangzhou 310030, Correspondence to: Lang Lin, MSc, Chief Doctor, Department Zhejiang Province, China of Gastroenterology, Cangnan People’s Hospital, Lingxi Town, Yucang Road No.195, Cangnan 325800, Zhejiang Province, ORCID number: Lang Lin (0000-0001-5879-7487); Zai-Bo Wen China. [email protected] (0000-0003-1290-0404); Dong-Jiao Lin (0000-0001-5186-8182); Jiang- Telephone: +86-577-64767351 Ting Dong (0000-0002-6433-0143); Jie Jin (0000-0003-1481-5107); Fei Fax: +86-577-64767351 Meng (0000-0003-1233-4270). Received: January 2, 2018 Author contributions: Lin L formulated the problem; Wen ZB, Peer-review started: January 2, 2018 Lin DJ and Dong JT collected samples; Meng F performed 16S First decision: January 18, 2018 rDNA sequencing; Jin J analyzed the data; Lin L and Jin J wrote Revised: February 2, 2018 the paper. -

Table S4. Phylogenetic Distribution of Bacterial and Archaea Genomes in Groups A, B, C, D, and X
Table S4. Phylogenetic distribution of bacterial and archaea genomes in groups A, B, C, D, and X. Group A a: Total number of genomes in the taxon b: Number of group A genomes in the taxon c: Percentage of group A genomes in the taxon a b c cellular organisms 5007 2974 59.4 |__ Bacteria 4769 2935 61.5 | |__ Proteobacteria 1854 1570 84.7 | | |__ Gammaproteobacteria 711 631 88.7 | | | |__ Enterobacterales 112 97 86.6 | | | | |__ Enterobacteriaceae 41 32 78.0 | | | | | |__ unclassified Enterobacteriaceae 13 7 53.8 | | | | |__ Erwiniaceae 30 28 93.3 | | | | | |__ Erwinia 10 10 100.0 | | | | | |__ Buchnera 8 8 100.0 | | | | | | |__ Buchnera aphidicola 8 8 100.0 | | | | | |__ Pantoea 8 8 100.0 | | | | |__ Yersiniaceae 14 14 100.0 | | | | | |__ Serratia 8 8 100.0 | | | | |__ Morganellaceae 13 10 76.9 | | | | |__ Pectobacteriaceae 8 8 100.0 | | | |__ Alteromonadales 94 94 100.0 | | | | |__ Alteromonadaceae 34 34 100.0 | | | | | |__ Marinobacter 12 12 100.0 | | | | |__ Shewanellaceae 17 17 100.0 | | | | | |__ Shewanella 17 17 100.0 | | | | |__ Pseudoalteromonadaceae 16 16 100.0 | | | | | |__ Pseudoalteromonas 15 15 100.0 | | | | |__ Idiomarinaceae 9 9 100.0 | | | | | |__ Idiomarina 9 9 100.0 | | | | |__ Colwelliaceae 6 6 100.0 | | | |__ Pseudomonadales 81 81 100.0 | | | | |__ Moraxellaceae 41 41 100.0 | | | | | |__ Acinetobacter 25 25 100.0 | | | | | |__ Psychrobacter 8 8 100.0 | | | | | |__ Moraxella 6 6 100.0 | | | | |__ Pseudomonadaceae 40 40 100.0 | | | | | |__ Pseudomonas 38 38 100.0 | | | |__ Oceanospirillales 73 72 98.6 | | | | |__ Oceanospirillaceae -

Engineering the Human Microbiome, a Natural Double-Barreled Approach Towards Solving Both Our Current Antibiotic Resistance and Sepsis Problems
American Journal of www.biomedgrid.com Biomedical Science & Research ISSN: 2642-1747 --------------------------------------------------------------------------------------------------------------------------------- Opinion Copy Right@ Toleman MA Engineering the Human Microbiome, A Natural Double-Barreled Approach Towards Solving both our Current Antibiotic Resistance and Sepsis Problems Martins W1, Mathias J1, Babenko D2 and Toleman MA1* 1Cardiff University Medical school, Department of Immunology and Infection, The Heath hospital, UK 2Karaganda Medical University, Kazakhstan *Corresponding author: Toleman MA, Cardiff University Medical school, Department of Immunology and Infection, The Heath hospital, Cardiff, UK. To Cite This Article: Martins W, Mathias J, Babenko D, Toleman MA, Engineering the Human Microbiome, A Natural Double-Barreled Approach Towards Solving both our Current Antibiotic Resistance and Sepsis Problems. Am J Biomed Sci & Res. 2020 - 11(3). AJBSR.MS.ID.001632. DOI: 10.34297/AJBSR.2020.11.001632. Received: December 12, 2020; Published: December 18, 2020 Opinion resistance mechanism responsible for this rise was an enzyme Many of the bacteria that cause life-threatening infection live called CTX-M-15 that gave resistance to the penicillin, monobactam, in very close association with us. Escherichia coli, for example and cephalosporin classes of the ß-lactam family of antibiotics (c. causes 80% of common community associated urinary tract infections and is also the main cause of serious (blood-stream) in several enteric pathogens including E. coli in New Delhi, India infection throughout Europe [1,2]. However, E. coli routinely lives 50% of all antibiotics) [7]. Its gene blaCTX-M-15 was first isolated in 2000 [8]. Later publications indicated that it was widespread as a commensal organism in our gut. -

Prokaryotes, Protists, Reconstructing the Photosynthesis, Evolution of Living Things
Prokaryotes, Protists, Reconstructing the Photosynthesis, evolution of living things Endosymbiosis • Systematists study evolutionary relationships Ch 28 & 29 •Look for shared derived (=different from ancestor) traits to group organisms • Evidence used: morphology, 26 February 2009 development, and molecular data (especially DNA sequences) ECOL 182R UofA K. E. Bonine 1 2 Why can’t we figure it out perfectly? • More distant history is obscured by more changes trypanosomes • Among oldest lineages of Bacteria and Archaea in particular, lots of “lateral gene transfer.” Makes it difficult to infer relationships from phylogeny of red blood cells single genes. 3 4 Early prokaryote fossil Diversity of Prokaryotes Bacteria & Archaea 5 6 1 What are microbes? Life can be divided into 3 domains • Only a minority make us sick •Robert Koch, Germ Theory of Disease • In ordinary English, might be anything small 3.8bya –bacteria 1.5bya –yeast –protists – viruses •Prokaryotes = bacteria + archaea • In science, classify by evolutionary • Prokaryote was ancestral and only form for relationships… 7 billions of years 8 Eukarya Scheme has been revised before: Haeckel (1894) Whittaker (1959) Woese (1977) Woese (1990) Three kingdoms Five kingdoms Six kingdoms Three domains Eubacteria Bacteria Monera (prokaryotes) Protista Archaebacteria Archaea Protista Protista Fungi Fungi Plantae Eukarya are Prokaryotes monophyletic, Plantae Plantae Animalia Animalia Animalia paraphyleticparaphyletic, polyphyletic? 9 modified from Wikipedia 10 Shared by all 3 domains Unique to -

Current Issues of Nano-Bio-Science
Current Issues of Nano-Bio-Science CeNS Winterschool 2003 Mauterndorf, Austria 24-28 February 2003 Current Issues of Nano-Bio-Science CeNS Winterschool 2003 Mauterndorf, Austria 24-28 February 2003 in cooperation with SFB 486 and SFB 513 Program Committee Organisation Christoph Bräuchle Monika Kaempfe Jan von Delft Evelyn Morgenroth Hermann Gaub Joachim Rädler Jörg P. Kotthaus Paul Leiderer Joachim Rädler Internal Students’ Seminar: Schedule Saturday, 22nd February 7:30 pm Meeting in front of the castle’s lower gate to go the “Skialm” together 8:00 pm Get-together at the „Skialm“ in Mauterndorf (in the Skiing center) (kitchen closes at 9:30 pm) Sunday, 23rd February 9:45 am Introduction (W. Parak, F. Simmel) 10:00 am Simon Keller (AG Rädler) 10:30 am Stefan Griessl (AG Heckl) 11:00 am Coffee break 11:30 am Christine Meyer (AG Kotthaus) 12:00 pm Michael Sindel (AG von Delft) 12:30 pm Lunch (not provided), informal discussions 5:00 pm Ferdinand Kühner (AG Gaub) 5:30 pm Stefan Beyer (NG Simmel) Teresa Pellegrino (NG Parak) 6:00 pm Stefan Kowarik (AG Feldmann) 6:30 pm Break 7:00 pm Niklolay Petkov (AG Bein) 7:30 pm Sebastian Gritschneder (AG Reichling) 8:00 pm Ralf Bausinger / Johanna Kirstein (AG Bräuchle) 8:30 pm Conclusion Program Monday, 24th February 2003 8.30 – 8.45 Opening 8.45 – 9.45 Cees Dekker, Delft University of Technology Carbon nanotubes as model systems for nanoelectronics and nanosensors 9.45 – 10.45 Ulf Diederichsen, Universität Göttingen Molecular architecture with biooligomers 10.45 – 11.15 Coffee Break 11.15 – 12.15 Wolfgang