Histomorphometric Analysis of the Temporomandibular Joint Condyle in Young and Mature Sheep
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Musculoskeletal Morphing from Human to Mouse
Procedia IUTAM Procedia IUTAM 00 (2011) 1–9 2011 Symposium on Human Body Dynamics Musculoskeletal Morphing from Human to Mouse Yoshihiko Nakamuraa,∗, Yosuke Ikegamia, Akihiro Yoshimatsua, Ko Ayusawaa, Hirotaka Imagawaa, and Satoshi Ootab aDepartment of Mechano-Informatics, Graduate School of Information and Science and Technology, University of Tokyo, 7-3-1, Hongo, Bunkyo-ku, Tokyo, Japan bBioresource Center, Riken, 3-1-1 Takanodai, Tsukuba-shi, Ibaragi, Japan Abstract The analysis of movement provides various insights of human body such as biomechanical property of muscles, function of neural systems, physiology of sensory-motor system, skills of athletic movements, and more. Biomechan- ical modeling and robotics computation have been integrated to extend the applications of musculoskeletal analysis of human movements. The analysis would also provide valuable means for the other mammalian animals. One of current approaches of post-genomic research focuses to find connections between the phenotype and the genotype. The former means the visible morphological or behavioral expression of an animal, while the latter implies its genetic expression. Knockout mice allows to study the developmental pathway from the genetic disorders to the behavioral disorders. Would musculoskeletal analysis of mice also offer scientific means for such study? This paper reports our recent technological development to build the musculoskeletal model of a laboratory mouse. We propose mapping the musculoskeletal model of human to a laboratory mouse based on the morphological similarity between the two mammals. Although the model will need fine adjustment based on the CT data or else, we can still use the mapped musculoskeletal model as an approximate model of the mouse’s musculoskeletal system. -

Sesamoid Bone of the Medial Collateral Ligament of the Knee Joint
CASE REPORT Eur. J. Anat. 21 (4): 309-313 (2017) Sesamoid bone of the medial collateral ligament of the knee joint Omar M. Albtoush, Konstantin Nikolaou, Mike Notohamiprodjo Department of Diagnostic and Interventional Radiology, Karls Eberhard Universität Tübingen, Hoppe-Seyler-Str. 3, 72076 Tübingen, Germany SUMMARY tomical relations and the exclusion of other possi- bilities. The variable occurrence of the sesamoid bones This article supports the theory stating that the supports the theory stating that the development development and evolution of the sesamoid bones and evolution of these bones are controlled are controlled through the interaction between in- through the interaction between intrinsic genetic trinsic genetic factors and extrinsic epigenetic stim- factors and extrinsic stimuli. In the present article uli, which can explain their variable occurrence. we report a sesamoid bone at the medial collateral ligament of the knee joint, a newly discovered find- CASE REPORT ing in human and veterinary medicine. We present a case of a 51-year-old female pa- Key words: Sesamoid – MCL – Knee – Fabella – tient, who presented with mild pain at the medial Cyamella aspect of the left knee. No trauma has been re- ported. An unenhanced spiral CT-Scan was per- INTRODUCTION formed with 2 mm thickness, 120 kvp and 100 mAs, which showed preserved articulation of the New structural anatomical discoveries are not so knee joint with neither joint effusion, nor narrowing often encountered. However, their potential occur- of the joint space nor articulating cortical irregulari- rence should be kept in mind, which can eventually ties (Fig. 1). Mild subchondral sclerosis was de- help in a better understanding of patients’ symp- picted at the medial tibial plateau as a sign of early toms and subsequently improve the management osteoarthritis. -

Palpation Techniques
Palpation Techniques Bearbeitet von Wolfgang Stelzenmüller, Michelle Hertrich, Gertrud Graubart Champe, Bernhard Reichert 1. Auflage 2010. Taschenbuch. 500 S. Paperback ISBN 978 3 13 146341 8 Format (B x L): 19,5 x 27 cm Weitere Fachgebiete > Medizin > Komplementäre Medizin, Asiatische Medizin (TCM), Heilpraktiker Zu Inhaltsverzeichnis schnell und portofrei erhältlich bei Die Online-Fachbuchhandlung beck-shop.de ist spezialisiert auf Fachbücher, insbesondere Recht, Steuern und Wirtschaft. Im Sortiment finden Sie alle Medien (Bücher, Zeitschriften, CDs, eBooks, etc.) aller Verlage. Ergänzt wird das Programm durch Services wie Neuerscheinungsdienst oder Zusammenstellungen von Büchern zu Sonderpreisen. Der Shop führt mehr als 8 Millionen Produkte. 140 6 Knee Joint Iliotibial tract Gerdy tubercle Fig. 6.49 Palpation of the iliotibial tract—anterior edge. Fig. 6.51 Palpation of the Gerdy tubercle. With the knee in slight flexion, the patient is instructed to isometrically contract the quadriceps. The hip is also flexed, abducted, and medially rotated. Using a perpendicular palpation technique, the edges of the tract can be identified slightly proximal to the level of the base of the patella (Fig. 6.50). Note • The tract is found directly over the lateral epicondyle when the knee is in 30−40° flexion. Less flexion shifts the tract so that it is then anterior to the epicondyle, while more flexion moves it posteriorly. It now be- comes apparent that the iliotibial tract must slide over the epicondyle during the gait cycle. This can oc- casionally cause symptoms. • A significant number of tract fibers extend down to the lateral edge of the patella and insert slightly distal to the vastus lateralis tendon. -

Axial Skeleton- Skull, Spinal Column Appendicular Skeleton – Limbs and Girdle
How many bones do humans have? • When you were born you had over 300 bones. As you grew, some of these bones began to fuse together. The result? An adult has only 206 bones! Factoids: • The human hand has 27 bones; your face has 14! • The longest bone in your body? Your thigh bone, the femur -- it's about 1/4 of your height. The smallest is the stirrup bone in the ear which can measure 1/10 of an inch. • Did you know that humans and giraffes have the same number of bones in their necks? Giraffe neck vertebrae are just much, much longer! • You have over 230 moveable and semi-moveable joints in your body. The Skeletal System Parts of the skeletal system Bones (skeleton) Joints Cartilages Ligaments (bone to bone)(tendon=bone to muscle) Divided into two divisions Axial skeleton- skull, spinal column Appendicular skeleton – limbs and girdle Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Functions of Bones Support of the body Protection of soft organs Movement due to attached skeletal muscles Storage of minerals and fats Blood cell formation Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Bones of the Human Body The skeleton has 206 bones Two basic types of bone tissue Compact bone Homogeneous Spongy bone Small needle-like pieces of bone Figure 5.2b Many open spaces Copyright © 2003 Pearson Education, Inc. publishing as Benjamin Cummings Bones are classified by their shape: 1. Long- bones are longer than they are wide (arms, legs) 2. Short- usually square in shape, cube like (wrist, ankle) 3. -

Injuries and Normal Variants of the Pediatric Knee
Revista Chilena de Radiología, año 2016. ARTÍCULO DE REVISIÓN Injuries and normal variants of the pediatric knee Cristián Padilla C.a,* , Cristián Quezada J.a,b, Nelson Flores N.a, Yorky Melipillán A.b and Tamara Ramírez P.b a. Imaging Center, Hospital Clínico Universidad de Chile, Santiago, Chile. b. Radiology Service, Hospital de Niños Roberto del Río, Santiago, Chile. Abstract: Knee pathology is a reason for consultation and a prevalent condition in children, which is why it is important to know both the normal variants as well as the most frequent pathologies. In this review a brief description is given of the main pathologies and normal variants that affect the knee in children, not only the main clinical characteristics but also the findings described in the different, most used imaging techniques (X-ray, ultrasound, computed tomography and magnetic resonance imaging [MRI]). Keywords: Knee; Paediatrics; Bone lesions. Introduction posteromedial distal femoral metaphysis, near the Pediatric knee imaging studies are used to evaluate insertion site of the medial twin muscle or adductor different conditions, whether traumatic, inflammatory, magnus1. It is a common finding on radiography and developmental or neoplastic. magnetic resonance imaging (MRI), incidental, with At a younger age the normal evolution of the more frequency between ages 10-15 years, although images during the skeletal development of the distal it can be present at any age until the physeal closure, femur, proximal tibia and proximal fibula should be after which it resolves1. In frontal radiography, it ap- known to avoid diagnostic errors. Older children and pears as a radiolucent, well circumscribed, cortical- adolescents present a higher frequency of traumatic based lesion with no associated soft tissue mass, with and athletic injuries. -

(AMIC) and Microfractures for Focal Chondral Defects of the Knee: a Medium-Term Comparative Study
life Communication Autologous Matrix-Induced Chondrogenesis (AMIC) and Microfractures for Focal Chondral Defects of the Knee: A Medium-Term Comparative Study Filippo Migliorini 1 , Jörg Eschweiler 1, Nicola Maffulli 2,3,4,* , Hanno Schenker 1, Alice Baroncini 1 , Markus Tingart 1 and Björn Rath 1,5 1 Department of Orthopedics and Trauma Surgery, University Clinic Aachen, RWTH Aachen University Clinic, 52064 Aachen, Germany; [email protected] (F.M.); [email protected] (J.E.); [email protected] (H.S.); [email protected] (A.B.); [email protected] (M.T.); [email protected] (B.R.) 2 Department of Medicine, Surgery and Dentistry, University of Salerno, Via S. Allende, 84081 Baronissi, Italy 3 School of Pharmacy and Bioengineering, Keele University School of Medicine, Thornburrow Drive, Stoke-on-Trent ST5 5BG, UK 4 Centre for Sports and Exercise Medicine, Barts and the London School of Medicine and Dentistry, Queen Mary University of London, Mile End Hospital, 275 Bancroft Road, London E1 4DG, UK 5 Department of Orthopedics, Klinikum Wels-Grieskirchen, A-4600 Wels, Austria * Correspondence: [email protected] Abstract: Introduction: The potential of autologous matrix-induced chondrogenesis (AMIC) to restore unipolar focal chondral defects of the knee is promising. However, the outcome compared to Citation: Migliorini, F.; Eschweiler, J.; microfracturing (MFx) for certain defect sizes (2–3 cm2) is still uncertain. Therefore, the present study Maffulli, N.; Schenker, H.; Baroncini, compared primary isolated AMIC versus MFx in a cohort of patients with borderline sized focal A.; Tingart, M.; Rath, B. Autologous unipolar chondral defects of the knee at midterm follow-up. -

Occipital Condyle Screws: Indications and Technique
163 Review of Techniques on Advanced Techniques in Complex Cervical Spine Surgery Occipital condyle screws: indications and technique Aju Bosco1, Ilyas Aleem2, Frank La Marca3 1Assistant Professor in Orthopedics and Spine Surgery, Orthopedic Spine Surgery Unit, Institute of Orthopedics and Traumatology, Madras Medical College, Chennai, India; 2Department of Orthopedic Surgery, University of Michigan, 2912 Taubman Center, Ann Arbor, MI, USA; 3Professor of Neurological Surgery, Henry Ford Health System, Jackson, MI, USA Contributions: (I) Conception and design: All authors; (II) Administrative support: All authors; (III) Provision of study materials or patients: A Bosco, I Aleem; (IV) Collection and assembly of data: A Bosco; (V) Data analysis and interpretation: A Bosco, I Aleem; (VI) Manuscript writing: All authors; (VII) Final approval of manuscript: All authors. Correspondence to: Aju Bosco, MS, DNB, FNB (Spine Surgery). Assistant Professor in Orthopedics and Spine Surgery, Orthopedic Spine Surgery Unit, Institute of Orthopedics and Traumatology, Madras Medical College, EVR Road, Park Town, Chennai, India. Email: [email protected]. Abstract: Occipitocervical instability is a life threatening and disabling disorder caused by a myriad of pathologies. Restoring the anatomical integrity and stability of the occipitocervical junction (OCJ) is essential to achieve optimal clinical outcomes. Surgical stabilization of the OCJ is challenging and technically demanding. There is a paucity of options available for anchorage in the cephalad part of the construct in occipitocervical fixation systems due to the intricate topography of the craniocervical junction combined with the risk of injury to the surrounding anatomical structures. Surgical techniques and instrumentation for stabilizing the unstable OCJ have undergone several modifications over the years and have primarily depended on occipital squama-based fixations. -

Humeral Condylar Fractures and Incomplete Ossification of the Humeral Condyle in Dogs ANDY MOORES
A five-year old springer PRACTICE ANIMAL COMPANION spaniel that was treated for an intercondylar Y fracture Humeral condylar fractures and incomplete ossification of the humeral condyle in dogs ANDY MOORES HUMERAL condylar fractures are among the most common fractures seen in dogs and account for approximately 20 per cent of the author’s canine fracture caseload, although this is a referral population and probably does not represent the true incidence of these injuries. It has long been recognised that spaniels are predisposed to humeral condylar fractures. It is now recognised that many of these dogs have a condition known as incomplete ossification of the humeral condyle (IOHC) that predisposes them to condylar fractures, often occurring during normal activity or associated with only minor trauma. This article discusses the management of humeral condylar fractures and IOHC. HUMERAL CONDYLAR FRACTURES the force from sudden impacts is primarily directed lat- Andy Moores erally. Secondly, the lateral epicondylar ridge is smaller graduated from Bristol in 1996. He CLASSIFICATION and weaker than its medial counterpart. Lateral condylar spent five years in Humeral condylar fractures can be divided into lateral fractures are most prevalent in skeletally immature dogs. small animal practice condylar, medial condylar and intercondylar fractures. In one retrospective review, 67 per cent of cases were before returning to Bristol to complete Lateral and medial humeral condylar fractures involve less than one year of age, the most common age being a residency in small only one epicondylar ridge of the condyle. Intercondylar four months (Denny 1983). Lateral and medial condylar animal surgery. In 2004, he joined the fractures involve both the medial and lateral epicondylar fractures are often associated with a minor fall, although Royal Veterinary ridges and are commonly described as ʻYʼ or ʻTʼ frac- in some dogs with IOHC they can occur during relative- College as a lecturer in small animal tures depending on the orientation of the fracture lines ly normal activity. -

AMIC—Autologous Matrix-Induced Chondrogenesis Technique in Patellar Cartilage Defects Treatment: a Retrospective Study with a Mid-Term Follow-Up
Journal of Clinical Medicine Article AMIC—Autologous Matrix-Induced Chondrogenesis Technique in Patellar Cartilage Defects Treatment: A Retrospective Study with a Mid-Term Follow-Up Daniele Tradati 1 , Paola De Luca 2 , Alessio Maione 1, Francesco Mattia Uboldi 1 , Piero Volpi 3, Laura de Girolamo 2,* and Massimo Berruto 1 1 U.O.S Chirurgia Articolare del Ginocchio, I Clinica Ortopedica, ASST Gaetano Pini-CTO Gaetano Pini, Piazza Cardinal Ferrari 1, 20122 Milan, Italy; [email protected] (D.T.); [email protected] (A.M.); [email protected] (F.M.U.); [email protected] (M.B.) 2 IRCCS Istituto Ortopedico Galeazzi, Orthopaedic Biotechnology Lab, 20161 Milan, Italy; [email protected] 3 U.O. Ortopedia del ginocchio e Traumatologia dello sport, Istituto Clinico Humanitas IRCCS, Via Alessandro Manzoni 56, 20089 Rozzano, Italy; [email protected] * Correspondence: [email protected]; Tel.: +39-026-621-4059 Received: 24 March 2020; Accepted: 16 April 2020; Published: 20 April 2020 Abstract: Background: Knee cartilage defects can be retrieved in 60% of patients undergoing knee arthroscopy, especially in the patellofemoral joint. Different techniques have been proposed to treat patellar defects, although most of them are associated with short-term results. In this study Autologous Matrix Induced Chondrogenesis (AMIC), combining subchondral microfractures with a collagen membrane (type I and III collagen), was used in the treatment of isolated patellar cartilage defects. Methods: Twenty-four patients were enrolled in this retrospective study. Subjective-International Knee Documentation Committee (IKDC), Visual Analog Scale for Pain (VAS), and Kujala score were collected at 1, 3, 6, and 12 months after surgery, whereas the Tegner Activity Level Scale was determined preoperatively and at final follow-up (final-FU). -

Musculoskeletal System
4 Musculoskeletal System Learning Objectives Upon completion of this chapter, you will be able to • Identify and define the combining forms, prefixes, and suffixes introduced in this chapter. • Correctly spell and pronounce medical terms and major anatomical structures relating to the musculoskeletal system. • Locate and describe the major organs of the musculoskeletal system and their functions. • Correctly place bones in either the axial or the appendicular skeleton. • List and describe the components of a long bone. • Identify bony projections and depressions. • Identify the parts of a synovial joint. • Describe the characteristics of the three types of muscle tissue. • Use movement terminology correctly. • Identify and define musculoskeletal system anatomical terms. • Identify and define selected musculoskeletal system pathology terms. • Identify and define selected musculoskeletal system diagnostic procedures. • Identify and define selected musculoskeletal system therapeutic procedures. • Identify and define selected medications relating to the musculoskeletal system. • Define selected abbreviations associated with the musculoskeletal system. 83 M04_FREM0254_06_SE_C04.indd 83 18/12/14 10:12 pm Section I: Skeletal System at a Glance Function The skeletal system consists of 206 bones that make up the internal framework of the body, called the skeleton. The skeleton supports the body, protects internal organs, serves as a point of attachment for skeletal muscles for body movement, produces blood cells, and stores minerals. Organs Here -

A Randomized Controlled Trial Demonstrating Sustained Benefit of Autologous Matrix-Induced Chondrogenesis Over Microfracture at Five Years
International Orthopaedics (SICOT) DOI 10.1007/s00264-016-3391-0 ORIGINAL PAPER A randomized controlled trial demonstrating sustained benefit of Autologous Matrix-Induced Chondrogenesis over microfracture at five years Martin Volz1 & Jens Schaumburger 2 & Hubert Frick1 & Joachim Grifka2 & Sven Anders 2 Received: 9 August 2016 /Accepted: 26 December 2016 # The Author(s) 2017. This article is published with open access at Springerlink.com Abstract Introduction Purpose Autologous Matrix-Induced Chondrogenesis (AMIC®) utilizing a type I/III collagen membrane was com- Cartilage has a low intrinsic regenerative and reparative capac- pared with microfracture (MFx) alone in focal cartilage le- ity, and cartilage defects can potentially lead to severe osteoar- sions of the knee at one, two and five years. thritis in the long term. A variety of surgical techniques that aim Methods Forty-seven patients (aged 37 ± 10 years, mean de- to resurface and repair the damaged articular cartilage include fect size 3.6 ± 1.6 cm2) were randomized and treated either perforation of the subchondral bone, mosaicplasty, and autolo- with MFx, with sutured or glued AMIC® in a prospective gous chondrocyte transplantation. Marrow stimulation multicentre clinical trial. methods, such as microfracturing (MFx), involve penetration Results After improvement for the first two years in all sub- of the subchondral bone plate to access the bone marrow com- groups, a progressive and significant score degradation was partment. The resulting blood clot enriched with bone marrow observed in the MFx group, while all functional parameters elements is thought to provide a favourable microenvironment remained stable for least five years in the AMIC® groups. -
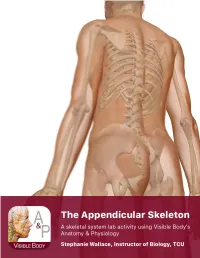
Lab Manual Appendicular Skele
1 PRE-LAB EXERCISES When studying the skeletal system, the bones are often sorted into two broad categories: the axial skeleton and the appendicular skeleton. This lab focuses on the appendicular skeleton, which is formed from the pectoral and pelvic girdles and the upper and lower limbs. View Module 7.2 Axial and Appendicular Skeleton to highlight the bones of the appendicular skeleton and compare them to those of the axial skeleton. Examine Module 11.1 Appendicular Skeleton to view only the bones of the appendicular skeleton. In addition to learning about all the bones of the appendicular skeleton, it is also important to identify some significant bone markings. Bone markings can have many shapes, including holes, round or sharp projections, and shallow or deep valleys, among others. These markings on the bones serve many purposes, including forming attachments to other bones or muscles and allowing passage of a blood vessel or nerve. It is helpful to understand the meanings of some of the more common bone marking terms. Before we get started, look up the definitions of these common bone marking terms: Canal: Condyle: Facet: Fissure: Foramen: (see Module 10.18 Foramina of Skull) Fossa: Margin: Process: Proximal: Trochanter: Tubercle: Tuberosity: Throughout this exercise, you will notice bold terms. This is meant to focus your attention on these important words. Make sure you pay attention to any bold words and know how to explain their definitions and/or where they are located. Use the following modules to guide your exploration of the appendicular skeleton. As you explore these bones in Visible Body’s app, also locate the bones and bone markings on any available charts, models, or specimens.