2019 Medicines in Development for Diabetes and Related Conditions
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Exenatide QW: a New Treatment Option for Type 2 Diabetes Offering Ease of Use, Improved Efficacy, and Reduced Side Effects
FEATURE ARTICLE Commentary: Exenatide QW: A New Treatment Option for Type 2 Diabetes Offering Ease of Use, Improved Efficacy, and Reduced Side Effects Charles F. Shaefer, Jr., MD Editor’s note: Once-weekly exenatide, (also called incretin mimetics) cur- with GLP-1 receptor agonists is the which has recently been approved for rently in use.3,4 only form of anti-diabetes therapy patients with type 2 diabetes, has great Validation of the acceptance offering proven weight loss. Finally, potential as a new diabetes therapy in clinical practice of GLP-1 GLP-1 receptor agonist therapy is in the primary care setting. This receptor agonists can be found in one of the recommended treatment commentary and the feature article that the recently released American strategies offering a low incidence of follows it (p. 95) offer an overview of Diabetes Association (ADA)/ hypoglycemia, an important aspect this new therapeutic tool and important European Association for the Study of diabetes therapy learned from the insights about its clinical utility. In the of Diabetes (EASD) position state- Action to Control Cardiovascular interest of transparency, however, we ment on the management of type 2 Risk in Diabetes trial.6 want to point out that the authors of diabetes, which placed these drugs As clinicians consider what to do both articles are affiliated with Amylin alongside older, well-accepted agents when metformin and lifestyle change Pharmaceuticals, which manufactures such as sulfonylureas (SUs) and do not adequately control a patient’s exenatide QW and markets it under the thiozolidinediones (TZDs) as a sec- A1C, they frequently ask, “incretin 5 trade name Bydureon. -

GLP-1 Receptor Agonists
Cognitive Vitality Reports® are reports written by neuroscientists at the Alzheimer’s Drug Discovery Foundation (ADDF). These scientific reports include analysis of drugs, drugs-in- development, drug targets, supplements, nutraceuticals, food/drink, non-pharmacologic interventions, and risk factors. Neuroscientists evaluate the potential benefit (or harm) for brain health, as well as for age-related health concerns that can affect brain health (e.g., cardiovascular diseases, cancers, diabetes/metabolic syndrome). In addition, these reports include evaluation of safety data, from clinical trials if available, and from preclinical models. GLP-1 Receptor Agonists Evidence Summary GLP-1 agonists are beneficial for patients with type 2 diabetes and obesity. Some evidence suggests benefits for Alzheimer’s disease. It is unclear whether it is beneficial for individuals without underlying metabolic disease. Semaglutide seems to be most effective for metabolic dysfunction, though liraglutide has more preclinical data for Alzheimer’s disease. Neuroprotective Benefit: Evidence from many preclinical studies and a pilot biomarker study suggest some neuroprotective benefits with GLP-1 agonists. However, whether they may be beneficial for everyone or only a subset of individuals (e.g. diabetics) is unclear. Aging and related health concerns: GLP-1 agonists are beneficial for treating diabetes and cardiovascular complications relating to diabetes. It is not clear whether they have beneficial effects in otherwise healthy individuals. Safety: GLP-1 agonists are generally safe for most people with minor side effects. However, long-term side effects are not known. 1 Availability: Available Dose: Varies - see Chemical formula: C172H265N43O51 (Liraglutide) as a prescription chart at the end of MW: 3751.262 g/mol medicine. -

Computationally Guided Design of Dipeptidyl Peptidase-4 Inhibitors Lauren C
bioRxiv preprint doi: https://doi.org/10.1101/772137; this version posted September 18, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Computationally Guided Design of Dipeptidyl Peptidase-4 Inhibitors Lauren C. Reynolds1, Morgan P. Connolly2,3, Justin B. Siegel1,2,4* 1. Department of Chemistry, University of California Davis, Davis, California, United States of America 2. Genome Center, University of California Davis, Davis, California, United States of America 3. Microbiology Graduate Group, University of California Davis, Davis, California, United States of America 4. Department of Biochemistry and Molecular Medicine, University of California Davis, Davis, California, United States of America ABSTRACT: The Type 2 Diabetes Mellitus (T2DM) epidemic undoubtedly creates a need for the development of new phar- maceuticals. With the goal of generating new therapeutics for this disease, computational studies were conducted to design novel dipeptidyl peptidase-4 (DPP-4) inhibitors. Two candidates, generated by chemical intuition-driven design and bioiso- steric replacement, were found to have better docking scores than anagliptin, a currently available diabetes medication. INTRODUCTION debate in recent years for other gliptins as well. A recent Type 2 Diabetes Mellitus (T2DM) is dubbed the meta-analysis seems to suggest a possible increased pan- “epidemic of the 21st century”— affecting millions of pa- creatitis risk for sitagliptin -

Footprints an Informational Newsletter for Patients of APMA Member Podiatrists October 2017
footprints an informational newsletter for patients of APMA member podiatrists october 2017 special EDITION include a DPM for diabetes prevention & Manage Ment According to the CDC, more than 100 million US adults are living with either diabetes or prediabetes. If you have diabetes or prediabetes, it is essential to include a podiatrist for proper diabetes prevention and management. In fact, podiatrists can reduce amputation rates up to 80 percent. In order for you not to become a part of that statistic, here are a few simple things you can do to keep your risk for diabetic ulcers and amputation low: Inspect feet daily. Check your Don’t go barefoot. Don’t go 1 feet and toes every day for 5 without shoes, even in your cuts, bruises, sores, or changes to own home. The risk of cuts and the toenails, such as thickening or infection is too great for those discoloration. with diabetes. Wear thick, soft socks. Avoid Never try to remove calluses, 2 socks with seams, which could 6 corns, or warts by yourself. rub and cause blisters or other Over-the-counter products can burn skin injuries. the skin and cause irreparable damage to the foot for people Exercise. Walking can keep with diabetes. 3 weight down and improve circulation. Be sure to wear See a podiatrist. Regular appropriate athletic shoes when 7 checkups by a podiatrist— exercising. at least annually—are the best WALKING CAN KEEP way to ensure that your feet WEIGHT DOWN AND Have new shoes properly remain healthy. 4 measured and fitted. Foot size IMPROVE CIRCULATION. -

Objectives & Accreditation
Objectives & Accreditation ACTIVITY FORMAT Live TARGET AUDIENCE This activity is designed to meet the needs of physicians, physician assistants, pharmacists, registered nurses, nurse practitioners, advance practice registered nurses, and registered dietitians with an interest in lipid management. TYPE OF ACTIVITY Live Activity; Application/ Knowledge EDUCATIONAL OBJECTIVES At the conclusion of this activity, Registered Nurses and Nurse Practitioners should be able to provide appropriate care and counsel for patients and their families. At the conclusion of this activity, all participants should be able to: Session I: The Impact of Genetics in Lipidology? Apply insight from human population genetics studies to identify novel therapeutic targets for a better understanding for atherosclerotic cardiovascular disease. Describe the current and future technologies available for genetic screen includinghow to apply them in clinical practice both for diagnosis and risk assessment Discuss experimental techniques in development for genetic manipulation to target inherited lipid disorders Keynote I Discuss ApoC3 as both a risk factor for ASCVD and as potential for therapeutic manipulation Session II- Focus on Lipoprotein (a) Discuss the epidemiology of Lipoprotein(a) (LP(a)) for both thrombosis and ASCVD Discuss the pathophysiology of LP(a) and how this impacts increased risk of ASCVD and thrombosis Discuss the status of current and emerging treatment options targeting LP(a) Clinical Lipidology Track- Critically appraise emerging research -

Diabetic Foot Ulcers
REVIEW Review Diabetic foot ulcers William J Jeffcoate, Keith G Harding Ulceration of the foot in diabetes is common and disabling and frequently leads to amputation of the leg. Mortality is high and healed ulcers often recur. The pathogenesis of foot ulceration is complex, clinical presentation variable, and management requires early expert assessment. Interventions should be directed at infection, peripheral ischaemia, and abnormal pressure loading caused by peripheral neuropathy and limited joint mobility. Despite treatment, ulcers readily become chronic wounds. Diabetic foot ulcers have been neglected in health-care research and planning, and clinical practice is based more on opinion than scientific fact. Furthermore, the pathological processes are poorly understood and poorly taught and communication between the many specialties involved is disjointed and insensitive to the needs of patients. In this review, we describe the epidemiology, pathogenesis, operations above the ankle or those proximal to the and management of diabetic foot ulceration, and its effect tarsometatarsal joint, and a multinational WHO survey on on patients and society. The condition deserves more lower extremity amputation also included cases of attention, both from those who provide care and those who unoperated gangrene.13 fund research. Moreover, amputation is a marker not just of disease but also of disease management. The decision to operate Epidemiology is determined by many factors, which vary between Incidence and prevalence centres and patients. A high amputation rate might result Although accurate figures are difficult to obtain for the from high disease prevalence, late presentation, and prevalence or incidence of foot ulcers, the results of cross- inadequate resources, but could also reflect a particular sectional community surveys in the UK showed that 5·3% approach by local surgeons. -

Performance of the Insulin-Only Ilet Bionic Pancreas and The
e118 Diabetes Care Volume 44, June 2021 Performance of the Insulin-Only Luz E. Castellanos,1 Courtney A. Balliro,1 Jordan S. Sherwood,1 Rabab Jafri,1 iLet Bionic Pancreas and the Mallory A. Hillard,1 Evelyn Greaux,1 Rajendranath Selagamsetty,2 Hui Zheng,3 Bihormonal iLet Using Firas H. El-Khatib,2 Edward R. Damiano,2,4 and Dasiglucagon in Adults With Type Steven J. Russell1 1 Diabetes in a Home-Use Setting Diabetes Care 2021;44:e118–e120 | https://doi.org/10.2337/dc20-1086 Reductions in blood glucose levels in with insulin lispro (Eli Lilly) or aspart (Table 1). The mean CGM glucose and people with diabetes are often achieved (Novo Nordisk), the bihormonal iLet for time in range (70–180 mg/dL) were 149 at the expense of increased hypoglyce- 7dayswithdasiglucagon(4mg/mL) ±13mg/dLand72±8%,respectively,in mia. A novel approach is to automati- and insulin lispro or aspart, or both, us- the insulin-only period, and 139 ± 11 cally deliver microdose glucagon when ing the same glucose target (110 mg/ mg/dL and 79 ± 9%, respectively, in the automation of insulin delivery alone is dL), in random order. There were no re- bihormonal period. The mean daily car- not sufficient to prevent hypoglycemia. strictions on diet or exercise. The prima- bohydrates consumed to prevent or The approach requires a bihormonal de- ry outcomes were prespecified iLet treat hypoglycemia were 16 ± 13 g and vice and a stable form of glucagon or operational thresholds. The key second- 18 ± 21 g in the insulin-only and bihor- glucagon analog. -

Pioneering New Markets Changing the Standard of Care
ANNUAL 2020 REPORT Pioneering New Markets Changing the Standard of Care UNITED STATES SECURITIES AND EXCHANGE COMMISSION Washington, DC 20549 FORM 10-K ☒ ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 For the fiscal year ended December 31, 2020 □ TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 For the transition period from to Commission file number 000-19125 Ionis Pharmaceuticals, Inc. (Exact name of Registrant as specified in its charter) Delaware 33-0336973 (State or other jurisdiction of (IRS Employer incorporation or organization) Identification No.) 2855 Gazelle Court, Carlsbad, CA 92010 (Address of Principal Executive Offices) (Zip Code) 760-931-9200 (Registrant’s telephone number, including area code) Securities registered pursuant to Section 12(b) of the Act: Title of each class Trading symbol Name of each exchange on which registered Common Stock, $.001 Par Value ‘‘IONS’’ The Nasdaq Stock Market LLC Securities registered pursuant to Section 12(g) of the Act: None Indicate by check mark if the Registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☒ No □ Indicate by check if the Registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes □ No ☒ Indicate by check mark whether the Registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the Registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. -

TRULICITY, INN-Dulaglutide
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Trulicity 0.75 mg solution for injection in pre-filled pen Trulicity 1.5 mg solution for injection in pre-filled pen Trulicity 3 mg solution for injection in pre-filled pen Trulicity 4.5 mg solution for injection in pre-filled pen 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Trulicity 0.75 mg solution for injection in pre-filled pen Each pre-filled pen contains 0.75 mg of dulaglutide* in 0.5 ml solution. Trulicity 1.5 mg solution for injection in pre-filled pen Each pre-filled pen contains 1.5 mg of dulaglutide* in 0.5 ml solution. Trulicity 3 mg solution for injection in pre-filled pen Each pre-filled pen contains 3 mg of dulaglutide* in 0.5 ml solution. Trulicity 4.5 mg solution for injection in pre-filled pen Each pre-filled pen contains 4.5 mg of dulaglutide* in 0.5 ml solution. *produced in CHO cells by recombinant DNA technology. For the full list of excipients, see section 6.1. 3. PHARMACEUTICAL FORM Solution for injection. Clear, colourless solution. 4. CLINICAL PARTICULARS 4.1 Therapeutic indications Type 2 Diabetes Mellitus Trulicity is indicated for the treatment of adults with insufficiently controlled type 2 diabetes mellitus as an adjunct to diet and exercise • as monotherapy when metformin is considered inappropriate due to intolerance or contraindications • in addition to other medicinal products for the treatment of diabetes. For study results with respect to combinations, effects on glycaemic control and cardiovascular events, and the populations studied, see sections 4.4, 4.5 and 5.1. -

200 Adverse Drug Reactions in a Southern Hospital in Taiwan 201
200 Adverse Drug Reactions in a southern hospital in Taiwan Yuhong Lin1 1Kaohsiung Veterans General Hospital Tainan Branch, Tainan City, Taiwan Objective: The aim of this study was to evaluate the adverse drug reactions (ADRs) reporting in a southern hospital in Taiwan. Methods: It was a retrospective study of ADRs reporting in 2016. We analysed ADRs which contain gender and age, occurring sources, Anatomical Therapeutic Chemical (ATC) classification of suspected drugs, management of ADRs and so on. Results: The study collected eighty-nine ADRs reported. Most of ADRs reported were occurring in outpatient department (87.6%). The average age of ADRs reported was 67.6 years. Majority of all ADRs reported were females (55.1%). According to ATC classification system, the major classification of suspected drugs were Sensory organs (32.6%). Among the adverse reactions, Dermatologic Effects (37.1%) were the major type of ADRs. Also, the major management of ADRs was to stop using the current suspected drug (46.1%). Conclusion: Certainly, ADRs reporting are still a very important information to healthcare professionals. As a result, we put all information of ADRs reported into medical system in our hospital and it will improve the safety of medication use. In case of prescribe the suspected medicines, it can remind doctor to think of information about patient's ADRs reported. No drugs is administered without risk. Therefore, all healthcare professionals should have a responsibility to their patients, who themselves are becoming more aware of problems -
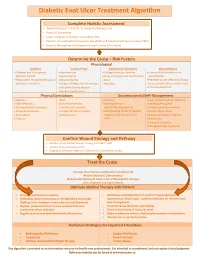
Diabetic Foot Ulcer Treatment Algorithm
Diabetic Foot Ulcer Treatment Algorithm Complete Holistic Assessment Medical/Surgical History & Co-morbidity Management Physical Examination Lower Leg (LLA) including monofilament test Perform Arterial Brachial Pressure Index (ABPI) and Toe Brachial Pressure Index (TBPI) Diabetic Management (Glycemic and Lipid Control & Nutrition) Determine the Cause – Risk Factors Physiological Diabetes Vascular Flow Autoimmune Disorders Wound History Diabetes and sub-optimal Hypertension Collagen vascular diseases History of foot infections or glycemic control Heart disease Immunosuppressant medications osteomyelitis Neuropathic changes with lack of Hyperlipidemia Gout Presence of toe infections (fungal protective sensation History of deep vein thrombosis Vasculitis or bacterial), callous and/or corns Peripheral artery disease Previous ulceration Venous Insufficiency Physical Limitations Socioeconomic/Self-Management Obesity Gait Smoking Lack of awareness for self-care Foot deformity Nutritional deficits Inadequate foot Inadequate hygiene Decreased level of activity Limited joint mobility wear/offloading devices Unsafe home environment Visual disturbances Congenital abnormalities Lack/Inability to afford diabetic Alcohol/drug abuse Amputation Osteoporosis supplies, foot care and foot Decreased Cognitive Ability Trauma wear Depression Financial insecurity Decreased level of activity Confirm Wound Etiology and Pathway Results of LLA, Monofilament Testing and ABPI/ TBPI Results of wound assessment Diagnostic -

Emerging Pharmacological Strategies for the Treatment
Emerging pharmacological strategies for the treatment of fibromyalgia LAWSON, Kim Available from Sheffield Hallam University Research Archive (SHURA) at: http://shura.shu.ac.uk/15300/ This document is the author deposited version. You are advised to consult the publisher's version if you wish to cite from it. Published version LAWSON, Kim (2017). Emerging pharmacological strategies for the treatment of fibromyalgia. World Journal of Pharmacology, 6 (1), 1-10. Repository use policy Copyright © and Moral Rights for the papers on this site are retained by the individual authors and/or other copyright owners. Users may download and/or print one copy of any article(s) in SHURA to facilitate their private study or for non- commercial research. You may not engage in further distribution of the material or use it for any profit-making activities or any commercial gain. Sheffield Hallam University Research Archive http://shura.shu.ac.uk World Journal of WJ P Pharmacology Submit a Manuscript: http://www.wjgnet.com/esps/ World J Pharmacol 207 March 9; 6(): -0 DOI: 0.5497/wjp.v6.i. ISSN 2220-392 (online) MINIREVIEWS Emerging pharmacological strategies for the treatment of ibromyalgia Kim Lawson Kim Lawson, Department of Biosciences and Chemistry, Bio presenting with a complex of symptoms dominated by molecular Sciences Research Centre, Shefield Hallam University, chronic widespread pain associated with the existence of a Faculty of Health and Wellbeing, Sheffield S1 1WB, United range of co-morbidities, such as fatigue, sleep disturbance, Kingdom cognitive impairment, anxiety and depression. Current treatments include drugs that target serotonin and nor- Author contributions: Lawson K researched the materials for the article and wrote the manuscript.