Volume 2, Chapter 11-6: Aquatic Insects: Hemimetabolous Insects
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Monte L. Bean Life Science Museum Brigham Young University Provo, Utah 84602 PBRIA a Newsletter for Plecopterologists
No. 10 1990/1991 Monte L. Bean Life Science Museum Brigham Young University Provo, Utah 84602 PBRIA A Newsletter for Plecopterologists EDITORS: Richard W, Baumann Monte L. Bean Life Science Museum Brigham Young University Provo, Utah 84602 Peter Zwick Limnologische Flußstation Max-Planck-Institut für Limnologie, Postfach 260, D-6407, Schlitz, West Germany EDITORIAL ASSISTANT: Bonnie Snow REPORT 3rd N orth A merican Stonefly S ymposium Boris Kondratieff hosted an enthusiastic group of plecopterologists in Fort Collins, Colorado during May 17-19, 1991. More than 30 papers and posters were presented and much fruitful discussion occurred. An enjoyable field trip to the Colorado Rockies took place on Sunday, May 19th, and the weather was excellent. Boris was such a good host that it was difficult to leave, but many participants traveled to Santa Fe, New Mexico to attend the annual meetings of the North American Benthological Society. Bill Stark gave us a way to remember this meeting by producing a T-shirt with a unique “Spirit Fly” design. ANNOUNCEMENT 11th International Stonefly Symposium Stan Szczytko has planned and organized an excellent symposium that will be held at the Tree Haven Biological Station, University of Wisconsin in Tomahawk, Wisconsin, USA. The registration cost of $300 includes lodging, meals, field trip and a T- Shirt. This is a real bargain so hopefully many colleagues and friends will come and participate in the symposium August 17-20, 1992. Stan has promised good weather and good friends even though he will not guarantee that stonefly adults will be collected during the field trip. Printed August 1992 1 OBITUARIES RODNEY L. -

Torix Rickettsia Are Widespread in Arthropods and Reflect a Neglected Symbiosis
GigaScience, 10, 2021, 1–19 doi: 10.1093/gigascience/giab021 RESEARCH RESEARCH Torix Rickettsia are widespread in arthropods and Downloaded from https://academic.oup.com/gigascience/article/10/3/giab021/6187866 by guest on 05 August 2021 reflect a neglected symbiosis Jack Pilgrim 1,*, Panupong Thongprem 1, Helen R. Davison 1, Stefanos Siozios 1, Matthew Baylis1,2, Evgeny V. Zakharov3, Sujeevan Ratnasingham 3, Jeremy R. deWaard3, Craig R. Macadam4,M. Alex Smith5 and Gregory D. D. Hurst 1 1Institute of Infection, Veterinary and Ecological Sciences, Faculty of Health and Life Sciences, University of Liverpool, Leahurst Campus, Chester High Road, Neston, Wirral CH64 7TE, UK; 2Health Protection Research Unit in Emerging and Zoonotic Infections, University of Liverpool, 8 West Derby Street, Liverpool L69 7BE, UK; 3Centre for Biodiversity Genomics, University of Guelph, 50 Stone Road East, Guelph, Ontario N1G2W1, Canada; 4Buglife – The Invertebrate Conservation Trust, Balallan House, 24 Allan Park, Stirling FK8 2QG, UK and 5Department of Integrative Biology, University of Guelph, Summerlee Science Complex, Guelph, Ontario N1G 2W1, Canada ∗Correspondence address. Jack Pilgrim, Institute of Infection, Veterinary and Ecological Sciences, Faculty of Health and Life Sciences, University of Liverpool, Liverpool, UK. E-mail: [email protected] http://orcid.org/0000-0002-2941-1482 Abstract Background: Rickettsia are intracellular bacteria best known as the causative agents of human and animal diseases. Although these medically important Rickettsia are often transmitted via haematophagous arthropods, other Rickettsia, such as those in the Torix group, appear to reside exclusively in invertebrates and protists with no secondary vertebrate host. Importantly, little is known about the diversity or host range of Torix group Rickettsia. -

New Records of Stoneflies (Plecoptera) with an Annotated Checklist of the Species for Pennsylvania
The Great Lakes Entomologist Volume 29 Number 3 - Fall 1996 Number 3 - Fall 1996 Article 2 October 1996 New Records of Stoneflies (Plecoptera) With an Annotated Checklist of the Species for Pennsylvania E. C. Masteller Behrend College Follow this and additional works at: https://scholar.valpo.edu/tgle Part of the Entomology Commons Recommended Citation Masteller, E. C. 1996. "New Records of Stoneflies (Plecoptera) With an Annotated Checklist of the Species for Pennsylvania," The Great Lakes Entomologist, vol 29 (3) Available at: https://scholar.valpo.edu/tgle/vol29/iss3/2 This Peer-Review Article is brought to you for free and open access by the Department of Biology at ValpoScholar. It has been accepted for inclusion in The Great Lakes Entomologist by an authorized administrator of ValpoScholar. For more information, please contact a ValpoScholar staff member at [email protected]. Masteller: New Records of Stoneflies (Plecoptera) With an Annotated Checklis 1996 THE GREAT LAKES ENTOMOlOGIST 107 NEW RECORDS OF STONEFLIES IPLECOPTERA} WITH AN ANNOTATED CHECKLIST OF THE SPECIES FOR PENNSYLVANIA E.C. Masteller1 ABSTRACT Original collections now record 134 species in nine families and 42 gen era. Seventeen new state records include, Allocapnia wrayi, Alloperla cau data, Leuctra maria, Soyedina carolinensis, Tallaperla elisa, Perlesta decipi· ens, P. placida, Neoperla catharae, N. occipitalis, N. stewarti, Cult us decisus decisus, Isoperla francesca, 1. frisoni, 1. lata,1. nana, 1. slossonae, Malirekus hastatus. Five species are removed from the list ofspecies for Pennsylvania. Surdick and Kim (1976) originally recorded 90 species of stoneflies in nine families and 32 genera from Pennsylvania. Since that time, Stark et al. -

RECENT PLECOPTERA LITERATURE (CALENDAR Zootaxa 795: 1-6
Oliver can be contacted at: Arscott, D. B., K. Tockner, and J. V. Ward. 2005. Lateral organization of O. Zompro, c/o Max-Planck-Institute of Limnology, aquatic invertebrates along the corridor of a braided floodplain P.O.Box 165, D-24302 Plön, Germany river. Journal of the North American Benthological Society 24(4): e-mail: [email protected] 934-954. Baillie, B. R., K. J. Collier, and J. Nagels. 2005. Effects of forest harvesting Peter Zwick and woody-debris removal on two Northland streams, New Pseudoretirement of Richard Baumann Zealand. New Zealand Journal of Marine and Freshwater Research 39(1): 1-15. I will officially retire from my position at Brigham Young Barquin, J., and R. G. Death. 2004. Patterns of invertebrate diversity in University on September 1, 2006. However, I will be able to maintain my streams and freshwater springs in Northern Spain. Archiv für workspace and research equipment at the Monte L. Bean Life Science Hydrobiologie 161: 329-349. Museum for a minimum of three years. At this time, I will work to complete Bednarek, A. T., and D. D. Hart. 2005. Modifying dam operations to restore many projects on stonefly systematics in concert with colleagues and rivers: Ecological responses to Tennessee River dam mitigation. friends. The stonefly collection will continue to grow and to by curated by Ecological Applications 15(3): 997-1008. Dr. C. Riley Nelson, Dr. Shawn Clark, and myself. I plan to be a major Beketov, M. A. 2005. Species composition of stream insects of northeastern “player” in stonefly research in North America for many years. -
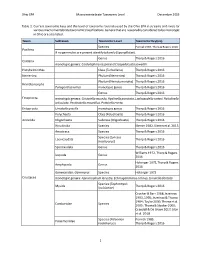
Ohio EPA Macroinvertebrate Taxonomic Level December 2019 1 Table 1. Current Taxonomic Keys and the Level of Taxonomy Routinely U
Ohio EPA Macroinvertebrate Taxonomic Level December 2019 Table 1. Current taxonomic keys and the level of taxonomy routinely used by the Ohio EPA in streams and rivers for various macroinvertebrate taxonomic classifications. Genera that are reasonably considered to be monotypic in Ohio are also listed. Taxon Subtaxon Taxonomic Level Taxonomic Key(ies) Species Pennak 1989, Thorp & Rogers 2016 Porifera If no gemmules are present identify to family (Spongillidae). Genus Thorp & Rogers 2016 Cnidaria monotypic genera: Cordylophora caspia and Craspedacusta sowerbii Platyhelminthes Class (Turbellaria) Thorp & Rogers 2016 Nemertea Phylum (Nemertea) Thorp & Rogers 2016 Phylum (Nematomorpha) Thorp & Rogers 2016 Nematomorpha Paragordius varius monotypic genus Thorp & Rogers 2016 Genus Thorp & Rogers 2016 Ectoprocta monotypic genera: Cristatella mucedo, Hyalinella punctata, Lophopodella carteri, Paludicella articulata, Pectinatella magnifica, Pottsiella erecta Entoprocta Urnatella gracilis monotypic genus Thorp & Rogers 2016 Polychaeta Class (Polychaeta) Thorp & Rogers 2016 Annelida Oligochaeta Subclass (Oligochaeta) Thorp & Rogers 2016 Hirudinida Species Klemm 1982, Klemm et al. 2015 Anostraca Species Thorp & Rogers 2016 Species (Lynceus Laevicaudata Thorp & Rogers 2016 brachyurus) Spinicaudata Genus Thorp & Rogers 2016 Williams 1972, Thorp & Rogers Isopoda Genus 2016 Holsinger 1972, Thorp & Rogers Amphipoda Genus 2016 Gammaridae: Gammarus Species Holsinger 1972 Crustacea monotypic genera: Apocorophium lacustre, Echinogammarus ischnus, Synurella dentata Species (Taphromysis Mysida Thorp & Rogers 2016 louisianae) Crocker & Barr 1968; Jezerinac 1993, 1995; Jezerinac & Thoma 1984; Taylor 2000; Thoma et al. Cambaridae Species 2005; Thoma & Stocker 2009; Crandall & De Grave 2017; Glon et al. 2018 Species (Palaemon Pennak 1989, Palaemonidae kadiakensis) Thorp & Rogers 2016 1 Ohio EPA Macroinvertebrate Taxonomic Level December 2019 Taxon Subtaxon Taxonomic Level Taxonomic Key(ies) Informal grouping of the Arachnida Hydrachnidia Smith 2001 water mites Genus Morse et al. -

Life History and Production of Mayflies, Stoneflies, and Caddisflies (Ephemeroptera, Plecoptera, and Trichoptera) in a Spring-Fe
Color profile: Generic CMYK printer profile Composite Default screen 1083 Life history and production of mayflies, stoneflies, and caddisflies (Ephemeroptera, Plecoptera, and Trichoptera) in a spring-fed stream in Prince Edward Island, Canada: evidence for population asynchrony in spring habitats? Michelle Dobrin and Donna J. Giberson Abstract: We examined the life history and production of the Ephemeroptera, Plecoptera, and Trichoptera (EPT) commu- nity along a 500-m stretch of a hydrologically stable cold springbrook in Prince Edward Island during 1997 and 1998. Six mayfly species (Ephemeroptera), 6 stonefly species (Plecoptera), and 11 caddisfly species (Trichoptera) were collected from benthic and emergence samples from five sites in Balsam Hollow Brook. Eleven species were abundant enough for life-history and production analysis: Baetis tricaudatus, Cinygmula subaequalis, Epeorus (Iron) fragilis,andEpeorus (Iron) pleuralis (Ephemeroptera), Paracapnia angulata, Sweltsa naica, Leuctra ferruginea, Amphinemura nigritta,and Nemoura trispinosa (Plecoptera), and Parapsyche apicalis and Rhyacophila brunnea (Trichoptera). Life-cycle timing of EPT taxa in Balsam Hollow Brook was generally similar to other literature reports, but several species showed extended emergence periods when compared with other studies, suggesting a reduction in synchronization of life-cycle timing, pos- sibly as a result of the thermal patterns in the stream. Total EPT secondary production (June 1997 to May 1998) was 2.74–2.80 g·m–2·year–1 dry mass (size-frequency method). Mayflies were dominant, with a production rate of 2.2 g·m–2·year–1 dry mass, followed by caddisflies at 0.41 g·m–2·year–1 dry mass, and stoneflies at 0.19 g·m–2·year–1 dry mass. -

About the Book the Format Acknowledgments
About the Book For more than ten years I have been working on a book on bryophyte ecology and was joined by Heinjo During, who has been very helpful in critiquing multiple versions of the chapters. But as the book progressed, the field of bryophyte ecology progressed faster. No chapter ever seemed to stay finished, hence the decision to publish online. Furthermore, rather than being a textbook, it is evolving into an encyclopedia that would be at least three volumes. Having reached the age when I could retire whenever I wanted to, I no longer needed be so concerned with the publish or perish paradigm. In keeping with the sharing nature of bryologists, and the need to educate the non-bryologists about the nature and role of bryophytes in the ecosystem, it seemed my personal goals could best be accomplished by publishing online. This has several advantages for me. I can choose the format I want, I can include lots of color images, and I can post chapters or parts of chapters as I complete them and update later if I find it important. Throughout the book I have posed questions. I have even attempt to offer hypotheses for many of these. It is my hope that these questions and hypotheses will inspire students of all ages to attempt to answer these. Some are simple and could even be done by elementary school children. Others are suitable for undergraduate projects. And some will take lifelong work or a large team of researchers around the world. Have fun with them! The Format The decision to publish Bryophyte Ecology as an ebook occurred after I had a publisher, and I am sure I have not thought of all the complexities of publishing as I complete things, rather than in the order of the planned organization. -

A Review of the Stoneflies of the Rock River, Illinois
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Illinois Digital Environment for Access to Learning and Scholarship Repository ILLINOI S UNIVERSITY OF ILLINOIS AT URBANA-CHAMPAIGN PRODUCTION NOTE University of Illinois at Urbana-Champaign Library Large-scale Digitization Project, 2007. A REVIEW OF THE STONEFLIES OF THE ROCK RIVER, ILLINOIS Dr. Donald W. Webb Center For Biodiversity Illinois Natural History Survey 607 East Peabody Drive Champaign, Illinois 61820 TECHNICAL REPORT 2002 (11) ILLINOIS NATURAL HISTORY SURVEY CENTER FOR BIODIVERSITY PREPARED FOR Division of Natural Heritage Office of Resource Conservation Illinois Department of Natural Resources One Natural Resources Way Springfield, IL 62702 Abstract During the 1990's, collecting was done along the Rock River in a effort to collect winter stoneflies (those species emerging from December through March). In 1997, collecting was done in and around Rock Island in an effort to collect Alloperla roberti. During April, May, and June of 2002, collecting for spring emerging stoneflies was conducted at nine sites along the Rock River from Rock Island to Rockton. Historically, 25 species of stoneflies (Insecta: Plecoptera) have been reported from the Rock River. Based on collecting from 1990-2002 eleven species (Acroneuria abnormis, Allocapnia granulata,Allocapnia vivipara Isoperla bilineata, Isoperla richardsoni,Perlesta golconda, Perlesta decipiens, Perlinella ephyre, Pteronarcys pictetii, Taeniopteryx burksi, Taeniopteryx nivalis) remain established within the Rock River. Acroneuria abnormis was previously very abundant along the length of the Rock River, but now is considered very rare. Allocapnia vivipara, the most common species of stonefly in Illinois and primarily a small stream species, appears to have been replaced by Allocapnia granulata in the Rock River. -

Thesis the Assimti...Ation and Elwination of Cesium By
THESIS THE ASSIMTI...ATION AND ELWINATION OF CESIUM BY FRESHWATER INVERTEBRATES Submitted by Tracy M. Tostowaryk Graduate Degree Program in Ecology In partial fulfillment of the requirements For the Degree of Master of Science Colorado State University Fort Collins, Colorado Fall 2000 QL 3bS.3b'5" .lb11 :2.0DO COLORADO STATE UNIVERSITY November 6, 2000 WE HEREBY RECOMMEND THAT THE THESIS PREPARED UNDER OUR SUPERVISION BY TRACY M. TOSTOWARYK ENTITLED "THE ASSIMILATION AND ELIMINATION OF CESIUM BY FRESHWATER INVERTEBRATES" BE ACCEPTED AS FULFILLING IN PART REQUIREMENTS FOR THE DEGREE OF MASTER OF SCIENCE. Adviser Co-Adviser ii COLORADO STATE UNIV. LIBRARIES ABSTRACT OF THESIS THE ASSIMILATION AND ELIMINATION OF CESIUM BY FRESHWATER INVERTEBRATES Freshwater invertebrates are important vectors of radioactive cesium e34Cs and 137CS) in aquatic food webs, yet little is known about their cesium :uptake and loss kinetics. This study provides a detailed investigation of cesium assimilation and elimination by freshwater invertebrates. Using five common freshwater invertebrates (Gammarus lacustris, Anisoptera sp. nymphs, Claassenia sabulosa and Megarcys signata nymphs, and Orconetes sp.), a variety of food types (oligochaete worms, mayfly nymphs and algae) and six temperature treatments (3.5 to 30°C), the following hypotheses were tested: 1) cesium elimination rates are a positive function of water temperature; 2) cesium elimination rates increase with decreasing body size; 3) assimilation efficiencies range between 0.6 and 0.8 for diet items low in clay. Cesium loss exhibited first order, non-linear kinetics, best described by a two component exponential model. Cesium assimilation efficiencies were higher for invertebrates fed oligochaetes (0.77) and algae (0.80) than those fed mayfly nymphs (0.20). -

Nymphs of North American Perlodinae Genera (Plecoptera: Perlodidae)
Great Basin Naturalist Volume 44 Number 3 Article 1 7-31-1984 Nymphs of North American Perlodinae genera (Plecoptera: Perlodidae) Kenneth W. Stewart North Texas State University, Denton, Texas Bill P. Stark Mississippi College, Clinton, Mississippi Follow this and additional works at: https://scholarsarchive.byu.edu/gbn Recommended Citation Stewart, Kenneth W. and Stark, Bill P. (1984) "Nymphs of North American Perlodinae genera (Plecoptera: Perlodidae)," Great Basin Naturalist: Vol. 44 : No. 3 , Article 1. Available at: https://scholarsarchive.byu.edu/gbn/vol44/iss3/1 This Article is brought to you for free and open access by the Western North American Naturalist Publications at BYU ScholarsArchive. It has been accepted for inclusion in Great Basin Naturalist by an authorized editor of BYU ScholarsArchive. For more information, please contact [email protected], [email protected]. The Great Basin Naturalist Published at Provo, Utah, by Brigham Young University ISSN 0017-3614 Volume 44 July 31, 1984 No. 3 NYMPHS OF NORTH AMERICAN PERLODINAE GENERA (PLECOPTERA: PERLODIDAE)' Kenneth VV. Stewart- and Bill P. Stark' Abstract.— Nymphs of the type or other representative species of the 22 North American Perlodinae genera are comparatively described and illustrated for the first time. The first complete generic key for the subfamily incorporates recent nymph discoveries and revisions in classification. References to all previous nymph descriptions and illustrations and major life cycle and food habits studies are given for the 53 North American species in the subfamilv, and a listing of species and their current distributions by states and provinces is provided for each genus. The previously unknown nymph of Chcrnokrihts misnomus is described and illustrated. -

A Comparison of Aquatic Invertebrate Assemblages Collected from the Green River in Dinosaur National Monument in 1962 and 2001
A Comparison of Aquatic Invertebrate Assemblages Collected from the Green River in Dinosaur National Monument in 1962 and 2001 Final Report for United States Department of the Interior National Park Service Dinosaur National Monument 4545 East Highway 40 Dinosaur, Colorado 81610-9724 Report Prepared by: Dr. Mark Vinson, Ph.D. & Ms. Erin Thompson National Aquatic Monitoring Center Department o f Fisheries and Wildlife Utah State University Logan, Utah 84322-5210 www.usu.edu/buglab 22 January 2002 i Foreword The work described in this report was conducted by personnel of the National Aquatic Monitoring Center, Utah State University, Logan, Utah. Mr. J. Matt Tagg aided in the identification of the aquatic invertebrates. Ms. Leslie Ogden provided computer assistance. Several people at Dinosaur National Monument helped us immensely with various project details. Steve Petersburg, Dana Dilsaver, and Dennis Ditmanson provided us with our research permit. Ann Elder helped with sample archiving. Christy Wright scheduled our trip and provided us with our river permit. We thank them all for all their help and good spirit. We also thank Mr. Walter Kittams (National Park Service, Regional Office, Omaha Nebraska) and Mr. Earl M. Semingsen (Superintendent, Dinosaur National Monument) for funding the study and the members of the 1962 University of Utah expedition: Dr. Angus M. Woodbury, Dr. Stephen Durrant, Mr. Delbert Argyle, Mr. Douglas Anderson, and Dr. Seville Flowers for their foresight to conduct the original study nearly 40 years ago. The concept and value of long-term ecological data is often bantered about, but its value is never more apparent then when we conduct studies like that presented here. -

100 Characters
40 Review and Update of Non-mollusk Invertebrate Species in Greatest Need of Conservation: Final Report Leon C. Hinz Jr. and James N. Zahniser Illinois Natural History Survey Prairie Research Institute University of Illinois 30 April 2015 INHS Technical Report 2015 (31) Prepared for: Illinois Department of Natural Resources State Wildlife Grant Program (Project Number T-88-R-001) Unrestricted: for immediate online release. Prairie Research Institute, University of Illinois at Urbana Champaign Brian D. Anderson, Interim Executive Director Illinois Natural History Survey Geoffrey A. Levin, Acting Director 1816 South Oak Street Champaign, IL 61820 217-333-6830 Final Report Project Title: Review and Update of Non-mollusk Invertebrate Species in Greatest Need of Conservation. Project Number: T-88-R-001 Contractor information: University of Illinois at Urbana/Champaign Institute of Natural Resource Sustainability Illinois Natural History Survey 1816 South Oak Street Champaign, IL 61820 Project Period: 1 October 2013—31 September 2014 Principle Investigator: Leon C. Hinz Jr., Ph.D. Stream Ecologist Illinois Natural History Survey One Natural Resources Way, Springfield, IL 62702-1271 217-785-8297 [email protected] Prepared by: Leon C. Hinz Jr. & James N. Zahniser Goals/ Objectives: (1) Review all SGNC listing criteria for currently listed non-mollusk invertebrate species using criteria in Illinois Wildlife Action Plan, (2) Assess current status of species populations, (3) Review criteria for additional species for potential listing as SGNC, (4) Assess stressors to species previously reviewed, (5) Complete draft updates and revisions of IWAP Appendix I and Appendix II for non-mollusk invertebrates. T-88 Final Report Project Title: Review and Update of Non-mollusk Invertebrate Species in Greatest Need of Conservation.