Nuclear Processes and Neutron Physics Prepared by Dr
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Spent Nuclear Fuel Pools in the US
Spent Nuclear Fuel Pools in the U.S.: Reducing the Deadly Risks of Storage front cover WITH SUPPORT FROM: WITH SUPPORT FROM: By Robert Alvarez 1112 16th St. NW, Suite 600, Washington DC 20036 - www.ips-dc.org May 2011 About the Author Robert Alvarez, an Institute for Policy Studies senior scholar, served as a Senior Policy Advisor to the Secre- tary of Energy during the Clinton administration. Institute for Policy Studies (IPS-DC.org) is a community of public scholars and organizers linking peace, justice, and the environment in the U.S. and globally. We work with social movements to promote true democracy and challenge concentrated wealth, corporate influence, and military power. Project On Government Oversight (POGO.org) was founded in 1981 as an independent nonprofit that investigates and exposes corruption and other misconduct in order to achieve a more effective, accountable, open, and ethical federal government. Institute for Policy Studies 1112 16th St. NW, Suite 600 Washington, DC 20036 http://www.ips-dc.org © 2011 Institute for Policy Studies [email protected] For additional copies of this report, see www.ips-dc.org Table of Contents Summary ...............................................................................................................................1 Introduction ..........................................................................................................................4 Figure 1: Explosion Sequence at Reactor No. 3 ........................................................4 Figure 2: Reactor No. 3 -

Neutron Stars
Chandra X-Ray Observatory X-Ray Astronomy Field Guide Neutron Stars Ordinary matter, or the stuff we and everything around us is made of, consists largely of empty space. Even a rock is mostly empty space. This is because matter is made of atoms. An atom is a cloud of electrons orbiting around a nucleus composed of protons and neutrons. The nucleus contains more than 99.9 percent of the mass of an atom, yet it has a diameter of only 1/100,000 that of the electron cloud. The electrons themselves take up little space, but the pattern of their orbit defines the size of the atom, which is therefore 99.9999999999999% Chandra Image of Vela Pulsar open space! (NASA/PSU/G.Pavlov et al. What we perceive as painfully solid when we bump against a rock is really a hurly-burly of electrons moving through empty space so fast that we can't see—or feel—the emptiness. What would matter look like if it weren't empty, if we could crush the electron cloud down to the size of the nucleus? Suppose we could generate a force strong enough to crush all the emptiness out of a rock roughly the size of a football stadium. The rock would be squeezed down to the size of a grain of sand and would still weigh 4 million tons! Such extreme forces occur in nature when the central part of a massive star collapses to form a neutron star. The atoms are crushed completely, and the electrons are jammed inside the protons to form a star composed almost entirely of neutrons. -
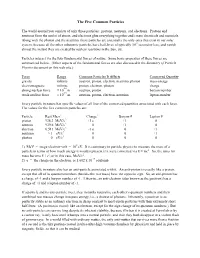
The Five Common Particles
The Five Common Particles The world around you consists of only three particles: protons, neutrons, and electrons. Protons and neutrons form the nuclei of atoms, and electrons glue everything together and create chemicals and materials. Along with the photon and the neutrino, these particles are essentially the only ones that exist in our solar system, because all the other subatomic particles have half-lives of typically 10-9 second or less, and vanish almost the instant they are created by nuclear reactions in the Sun, etc. Particles interact via the four fundamental forces of nature. Some basic properties of these forces are summarized below. (Other aspects of the fundamental forces are also discussed in the Summary of Particle Physics document on this web site.) Force Range Common Particles It Affects Conserved Quantity gravity infinite neutron, proton, electron, neutrino, photon mass-energy electromagnetic infinite proton, electron, photon charge -14 strong nuclear force ≈ 10 m neutron, proton baryon number -15 weak nuclear force ≈ 10 m neutron, proton, electron, neutrino lepton number Every particle in nature has specific values of all four of the conserved quantities associated with each force. The values for the five common particles are: Particle Rest Mass1 Charge2 Baryon # Lepton # proton 938.3 MeV/c2 +1 e +1 0 neutron 939.6 MeV/c2 0 +1 0 electron 0.511 MeV/c2 -1 e 0 +1 neutrino ≈ 1 eV/c2 0 0 +1 photon 0 eV/c2 0 0 0 1) MeV = mega-electron-volt = 106 eV. It is customary in particle physics to measure the mass of a particle in terms of how much energy it would represent if it were converted via E = mc2. -

Photons from 252Cf and 241Am-Be Neutron Sources H
Neutronenbestrahlungsraum Calibration Laboratory LB6411 Am-Be Quelle [email protected] Photons from 252Cf and 241Am-Be neutron sources H. Hoedlmoser, M. Boschung, K. Meier and S. Mayer Paul Scherrer Institut, CH-5232 Villigen PSI, Switzerland At the accredited PSI calibration laboratory neutron reference fields traceable to the standards of the Physikalisch-Technische Bundesanstalt (PTB) in Germany are available for the calibration of ambient and personal dose equivalent (rate) meters and passive dosimeters. The photon contribution to H*(10) in the neutron fields of the 252Cf and 241Am-Be sources was measured using various photon dose rate meters and active and passive dosimeters. Measuring photons from a neutron source involves considerable uncertainties due to the presence of neutrons, due to a non-zero neutron sensitivity of the photon detector and due to the energy response of the photon detectors. Therefore eight independent detectors and methods were used to obtain a reliable estimate for the photon contribution as an average of the individual methods. For the 241Am-Be source a photon contribution of approximately 4.9% was determined and for the 252Cf source a contribution of 3.6%. 1) Photon detectors 2) Photons from 252Cf and 241Am-Be neutron sources Figure 1 252Cf decays through -emission (~97%) and through spontaneous fission (~3%) with a half-life of 2.65 y. Both processes are accompanied by photon radiation. Furthermore the spectrum of fission products contains radioactive elements that produce additional gamma photons. In the 241Am-Be source 241Am decays through -emission and various low energy emissions: 241Am237Np + + with a half life of 432.6 y. -

Progress in Fusion-Driven Transmutation Research in China
Progress in Fusion-Driven Transmutation Research in China Yican WU Institute of Plasma Physics, Chinese Academy of Sciences P.O. Box 1126, Hefei, Anhui, 230031, China E-mail: [email protected] Fax: +86 551 5591310 1. Introduction Although the recent experiments and associated theoretical studies of fusion energy development have proven the feasibility of fusion power, it's commonly realized that it needs hard work before pure fusion energy could commercially and economically utilized. On the other hand, the fission nuclear industry has been falling on hard times recently since so far there has been no conclusion about how to deal with the long-lived wastes produced from the nuclear spent fuel and about how to solve the shortage of natural uranium ore in addition to nuclear safety and proliferation. It's a natural way to develop fusion-fission hybrid reactors including fuel producing reactors and waste transmuting reactors as an alternate strategy to speed up the time for producing energy since a hybrid reactor as a subcritical system can operate with lower fusion energy gain ratios Q, therefore the design of the fusion core for a hybrid system is easier than for a pure fusion reactor. The fusion-fission hybrid concept dates back to the earliest days of the fusion project when it was recognized that using fusion neutrons to breed nuclear fuel would vastly increase the energy from fusion plant. It appears to receive almost no attention since the mid 80's in the world, except in China who has given very serious consideration and has strong hybrid reactor activities since 1986 in the framework of the National Hi’Tech Program supported by the State Science and Technology Commission (SSTC) of China. -

Opportunities for Neutrino Physics at the Spallation Neutron Source (SNS)
Opportunities for Neutrino Physics at the Spallation Neutron Source (SNS) Paper submitted for the 2008 Carolina International Symposium on Neutrino Physics Yu Efremenko1,2 and W R Hix2,1 1University of Tennessee, Knoxville TN 37919, USA 2Oak Ridge National Laboratory, Oak Ridge TN 37981, USA E-mail: [email protected] Abstract. In this paper we discuss opportunities for a neutrino program at the Spallation Neutrons Source (SNS) being commissioning at ORNL. Possible investigations can include study of neutrino-nuclear cross sections in the energy rage important for supernova dynamics and neutrino nucleosynthesis, search for neutrino-nucleus coherent scattering, and various tests of the standard model of electro-weak interactions. 1. Introduction It seems that only yesterday we gathered together here at Columbia for the first Carolina Neutrino Symposium on Neutrino Physics. To my great astonishment I realized it was already eight years ago. However by looking back we can see that enormous progress has been achieved in the field of neutrino science since that first meeting. Eight years ago we did not know which region of mixing parameters (SMA. LMA, LOW, Vac) [1] would explain the solar neutrino deficit. We did not know whether this deficit is due to neutrino oscillations or some other even more exotic phenomena, like neutrinos decay [2], or due to the some other effects [3]. Hints of neutrino oscillation of atmospheric neutrinos had not been confirmed in accelerator experiments. Double beta decay collaborations were just starting to think about experiments with sensitive masses of hundreds of kilograms. Eight years ago, very few considered that neutrinos can be used as a tool to study the Earth interior [4] or for non- proliferation [5]. -

PGNAA Neutron Source Moderation Setup Optimization
Submitted to ‘Chinese Physics C PGNAA neutron source moderation setup optimization Zhang Jinzhao1(张金钊)Tuo Xianguo1(庹先国) (1.Chengdu University of Technology Applied Nuclear Techniques in Geoscience Key Laboratory of Sichuan Province,Chengdu 610059,China) Abstract: Monte Carlo simulations were carried out to design a prompt γ-ray neutron activation analysis (PGNAA) thermal neutron output setup using MCNP5 computer code. In these simulations the moderator materials, reflective materials and structure of the PGNAA 252Cf neutrons of thermal neutron output setup were optimized. Results of the calcuations revealed that the thin layer paraffin and the thick layer of heavy water moderated effect is best for 252Cf neutrons spectrum. The new design compared with the conventional neutron source design, the thermal neutron flux and rate were increased by 3.02 times and 3.27 times. Results indicate that the use of this design should increase the neutron flux of prompt gamma-ray neutron activation analysis significantly. Key word: PGNAA; neutron source; thermal neutron; moderation; reflection 1. Introduction study, Monte Carlo calculation was carried out for the Prompt gamma ray neutron activation analysis design of a 252Cf neutron source moderation setup for the (PGNAA) is a rapid, nondestructive, powerful analysis cement samples[7]. The model of Monte Carlo multielemental analysis technique, large samples of some simulation was verified by experiment[8, 9].We improve minor, trace light elements and is used in industrial the thermal neutron source yield rate of 252Cf neutron by control[1-5]. In a PGNAA analysis, the sample nuclear the PGNAA neutron source structure to the design. The composition is determined from prompt gamma rays calculation results for the new design were compared which produced through neutron inelastic scattering and with the previous, example: themal neutron flux rate, fast thermal neutron capture. -

1 Phys:1200 Lecture 36 — Atomic and Nuclear Physics
1 PHYS:1200 LECTURE 36 — ATOMIC AND NUCLEAR PHYSICS (4) This last lecture of the course will focus on nuclear energy. There is an enormous reservoir of energy in the nucleus and it can be released either in a controlled manner in a nuclear reactor, or in an uncontrolled manner in a nuclear bomb. The energy released in a nuclear reactor can be used to produce electricity. The two processes in which nuclear energy is released – nuclear fission and nuclear fusion, will be discussed in this lecture. The biological effects of nuclear radiation will also be discussed. 36‐1. Biological Effects of Nuclear Radiation.—Radioactive nuclei emit alpha, beta, and gamma radiation. These radiations are harmful to humans because they are ionizing radiation that have the ability to remove electrons from atoms and molecules in human cells. This can lead to the death or alterations of cells. Alteration of the cell can transform a healthy cell into a cancer cell. The hazards of radiation can be minimized by limiting ones overall exposure to radiation. However, there is still some uncertainty in the medical community about the possibility the effect of a single radioactive particle on the bottom. In other words, are the effects cumulative, or can a single exposure lead to cancer in the body. Exposure to radiation can produce either short term effects appearing within minutes of exposure, or long term effects that may appear in years or decades or even in future generations due to changes in DNA. The effects of absorbing ionizing radiation is measured in a unit called the rem. -

Enrico Fermi
Fermi, Enrico Inventors and Inventions Enrico Fermi Italian American physicist Fermi helped develop Fermi-Dirac statistics, which liceo (secondary school) and, on the advice of Amidei, elucidate the group behavior of elementary particles. joined the Scuola Normale Superiore at Pisa. This elite He also developed the theory of beta decay and college, attached to the University of Pisa, admitted only discovered neutron-induced artificial radioactivity. forty of Italy’s top students, who were given free board Finally, he succeeded in producing the first sustained and lodging. Fermi performed exceedingly well in the nuclear chain reaction, which led to the discovery highly competitive entrance exam. He completed his of nuclear energy and the development of the university education after only four years of research and atomic bomb. studies, receiving his Ph.D. in physics from the Univer- sity of Pisa and his undergraduate diploma from the Born: September 29, 1901; Rome, Italy Scuola Normale Superiore in July, 1922. He became Died: November 28, 1954; Chicago, Illinois an expert theoretical physicist and a talented exper- Primary field: Physics imentalist. This rare combination provided a solid foun- Primary inventions: Controlled nuclear chain dation for all his subsequent inventions. reaction; Fermi-Dirac statistics; theory of beta decay Life’s Work After postdoctoral work at the University of Göttingen, Early Life in Germany (1922-1923), and the University of Leiden, Enrico Fermi (ehn-REE-koh FUR-mee) was the third in the Netherlands (fall, 1924), Fermi took an interim po- child of Alberto Fermi and Ida de Gattis. Enrico was very sition at the University of Florence in December, 1924. -

Nuclear Weapons Technology 101 for Policy Wonks Bruce T
NUCLEAR WEAPONS TECHNOLOGY FOR POLICY WONKS NUCLEAR WEAPONS TECHNOLOGY 101 FOR POLICY WONKS BRUCE T. GOODWIN BRUCE T. GOODWIN BRUCE T. Center for Global Security Research Lawrence Livermore National Laboratory August 2021 NUCLEAR WEAPONS TECHNOLOGY 101 FOR POLICY WONKS BRUCE T. GOODWIN Center for Global Security Research Lawrence Livermore National Laboratory August 2021 NUCLEAR WEAPONS TECHNOLOGY 101 FOR POLICY WONKS | 1 This work was performed under the auspices of the U.S. Department of Energy by Lawrence Livermore National Laboratory in part under Contract W-7405-Eng-48 and in part under Contract DE-AC52-07NA27344. The views and opinions of the author expressed herein do not necessarily state or reflect those of the United States government or Lawrence Livermore National Security, LLC. ISBN-978-1-952565-11-3 LCCN-2021907474 LLNL-MI-823628 TID-61681 2 | BRUCE T. GOODWIN Table of Contents About the Author. 2 Introduction . .3 The Revolution in Physics That Led to the Bomb . 4 The Nuclear Arms Race Begins. 6 Fission and Fusion are "Natural" Processes . 7 The Basics of the Operation of Nuclear Explosives. 8 The Atom . .9 Isotopes . .9 Half-life . 10 Fission . 10 Chain Reaction . 11 Critical Mass . 11 Fusion . 14 Types of Nuclear Weapons . 16 Finally, How Nuclear Weapons Work . 19 Fission Explosives . 19 Fusion Explosives . 22 Staged Thermonuclear Explosives: the H-bomb . 23 The Modern, Miniature Hydrogen Bomb . 25 Intrinsically Safe Nuclear Weapons . 32 Underground Testing . 35 The End of Nuclear Testing and the Advent of Science-Based Stockpile Stewardship . 39 Stockpile Stewardship Today . 41 Appendix 1: The Nuclear Weapons Complex . -

Improved Algorithms and Coupled Neutron-Photon Transport For
Submitted to ‘Chinese Physics C’ Improved Algorithms and Coupled Neutron-Photon Transport for Auto-Importance Sampling Method * Xin Wang(王鑫)1,2 Zhen Wu(武祯)3 Rui Qiu(邱睿)1,2;1) Chun-Yan Li(李春艳)3 Man-Chun Liang(梁漫春)1 Hui Zhang(张辉) 1,2 Jun-Li Li(李君利)1,2 Zhi Gang(刚直)4 Hong Xu(徐红)4 1 Department of Engineering Physics, Tsinghua University, Beijing 100084, China 2Key Laboratory of Particle & Radiation Imaging, Ministry of Education, Beijing 100084, China 3 Nuctech Company Limited, Beijing 100084, China 4 State Nuclear Hua Qing (Beijing) Nuclear Power Technology R&D Center Co. Ltd., Beijing 102209, China Abstract: The Auto-Importance Sampling (AIS) method is a Monte Carlo variance reduction technique proposed for deep penetration problems, which can significantly improve computational efficiency without pre-calculations for importance distribution. However, the AIS method is only validated with several simple examples, and cannot be used for coupled neutron-photon transport. This paper presents improved algorithms for the AIS method, including particle transport, fictitious particle creation and adjustment, fictitious surface geometry, random number allocation and calculation of the estimated relative error. These improvements allow the AIS method to be applied to complicated deep penetration problems with complex geometry and multiple materials. A Completely coupled Neutron-Photon Auto-Importance Sampling (CNP-AIS) method is proposed to solve the deep penetration problems of coupled neutron-photon transport using the improved algorithms. The NUREG/CR-6115 PWR benchmark was calculated by using the methods of CNP-AIS, geometry splitting with Russian roulette and analog Monte Carlo, respectively. -

8.1 the Neutron-To-Proton Ratio
M. Pettini: Introduction to Cosmology | Lecture 8 PRIMORDIAL NUCLEOSYNTHESIS Our discussion at the end of the previous lecture concentrated on the rela- tivistic components of the Universe, photons and leptons. Baryons (which are non-relativistic) did not figure because they make a trifling contribu- tion to the energy density. However, from t ∼ 1 s a number of nuclear reactions involving baryons took place. The end result of these reactions was to lock up most of the free neutrons into 4He nuclei and to create trace amounts of D, 3He, 7Li and 7Be. This is Primordial Nucleosynthesis, also referred to as Big Bang Nucleosynthesis (BBN). 8.1 The Neutron-to-Proton Ratio Before neutrino decoupling at around 1 MeV, neutrons and protons are kept in mutual thermal equilibrium through charged-current weak interactions: + n + e ! p +ν ¯e − p + e ! n + νe (8.1) − n ! p + e +ν ¯e While equilibrium persists, the relative number densities of neutrons and protons are given by a Boltzmann factor based on their mass difference: n ∆m c2 1:5 = exp − = exp − (8.2) p eq kT T10 where 10 ∆m = (mn − mp) = 1:29 MeV = 1:5 × 10 K (8.3) 10 and T10 is the temperature in units of 10 K. As discussed in Lecture 7.1.2, this equilibrium will be maintained so long as the timescale for the weak interactions is short compared with the timescale of the cosmic expansion (which increases the mean distance between parti- cles). In 7.2.1 we saw that the ratio of the two rates varies approximately as: Γ T 3 ' : (8.4) H 1:6 × 1010 K 1 This steep dependence on temperature can be appreciated as follows.