Exclusion of Dlx5/6 Expression from the Distal-Most Mandibular Arches Enables BMP-Mediated Specification of the Distal Cap
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Multifactorial Erβ and NOTCH1 Control of Squamous Differentiation and Cancer
Multifactorial ERβ and NOTCH1 control of squamous differentiation and cancer Yang Sui Brooks, … , Karine Lefort, G. Paolo Dotto J Clin Invest. 2014;124(5):2260-2276. https://doi.org/10.1172/JCI72718. Research Article Oncology Downmodulation or loss-of-function mutations of the gene encoding NOTCH1 are associated with dysfunctional squamous cell differentiation and development of squamous cell carcinoma (SCC) in skin and internal organs. While NOTCH1 receptor activation has been well characterized, little is known about how NOTCH1 gene transcription is regulated. Using bioinformatics and functional screening approaches, we identified several regulators of the NOTCH1 gene in keratinocytes, with the transcription factors DLX5 and EGR3 and estrogen receptor β (ERβ) directly controlling its expression in differentiation. DLX5 and ERG3 are required for RNA polymerase II (PolII) recruitment to the NOTCH1 locus, while ERβ controls NOTCH1 transcription through RNA PolII pause release. Expression of several identified NOTCH1 regulators, including ERβ, is frequently compromised in skin, head and neck, and lung SCCs and SCC-derived cell lines. Furthermore, a keratinocyte ERβ–dependent program of gene expression is subverted in SCCs from various body sites, and there are consistent differences in mutation and gene-expression signatures of head and neck and lung SCCs in female versus male patients. Experimentally increased ERβ expression or treatment with ERβ agonists inhibited proliferation of SCC cells and promoted NOTCH1 expression and squamous differentiation both in vitro and in mouse xenotransplants. Our data identify a link between transcriptional control of NOTCH1 expression and the estrogen response in keratinocytes, with implications for differentiation therapy of squamous cancer. Find the latest version: https://jci.me/72718/pdf Research article Multifactorial ERβ and NOTCH1 control of squamous differentiation and cancer Yang Sui Brooks,1,2 Paola Ostano,3 Seung-Hee Jo,1,2 Jun Dai,1,2 Spiro Getsios,4 Piotr Dziunycz,5 Günther F.L. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

¬LACZ-REPORTER MAPPING of Dlx5/6 EXPRESSION AND
Received: 26 December 2019 Revised: 10 May 2020 Accepted: 11 May 2020 DOI: 10.1002/cne.24952 RESEARCH ARTICLE LacZ-reporter mapping of Dlx5/6 expression and genoarchitectural analysis of the postnatal mouse prethalamus Luis Puelles1 | Carmen Diaz2 | Thorsten Stühmer3 | José L. Ferran1 | Margaret Martínez-de la Torre1 | John L. R. Rubenstein3 1Department of Human Anatomy and Psychobiology and IMIB-Arrixaca Institute, Abstract University of Murcia, Murcia, Spain We present here a thorough and complete analysis of mouse P0-P140 prethalamic 2 Department of Medical Sciences, School of histogenetic subdivisions and corresponding nuclear derivatives, in the context of Medicine and Institute for Research in Neurological Disabilities, University of Castilla- local tract landmarks. The study used as fundamental material brains from a transgenic La Mancha, Albacete, Spain mouse line that expresses LacZ under the control of an intragenic enhancer of Dlx5 3Nina Ireland Laboratory of Developmental Neurobiology, Department of Psychiatry, and Dlx6 (Dlx5/6-LacZ). Subtle shadings of LacZ signal, jointly with pan-DLX immu- UCSF Medical School, San Francisco, noreaction, and several other ancillary protein or RNA markers, including Calb2 and California Nkx2.2 ISH (for the prethalamic eminence, and derivatives of the rostral zona limitans Correspondence shell domain, respectively) were mapped across the prethalamus. The resulting model Luis Puelles, Department of Human Anatomy and Psychobiology, School of Medicine, of the prethalamic region postulates tetrapartite rostrocaudal and dorsoventral subdi- University of Murcia, Murcia 30071, Spain. visions, as well as a tripartite radial stratification, each cell population showing a char- Email: [email protected] acteristic molecular profile. Some novel nuclei are proposed, and some instances of Carmen Díaz, Department of Medical potential tangential cell migration were noted. -

Supplemental Materials ZNF281 Enhances Cardiac Reprogramming
Supplemental Materials ZNF281 enhances cardiac reprogramming by modulating cardiac and inflammatory gene expression Huanyu Zhou, Maria Gabriela Morales, Hisayuki Hashimoto, Matthew E. Dickson, Kunhua Song, Wenduo Ye, Min S. Kim, Hanspeter Niederstrasser, Zhaoning Wang, Beibei Chen, Bruce A. Posner, Rhonda Bassel-Duby and Eric N. Olson Supplemental Table 1; related to Figure 1. Supplemental Table 2; related to Figure 1. Supplemental Table 3; related to the “quantitative mRNA measurement” in Materials and Methods section. Supplemental Table 4; related to the “ChIP-seq, gene ontology and pathway analysis” and “RNA-seq” and gene ontology analysis” in Materials and Methods section. Supplemental Figure S1; related to Figure 1. Supplemental Figure S2; related to Figure 2. Supplemental Figure S3; related to Figure 3. Supplemental Figure S4; related to Figure 4. Supplemental Figure S5; related to Figure 6. Supplemental Table S1. Genes included in human retroviral ORF cDNA library. Gene Gene Gene Gene Gene Gene Gene Gene Symbol Symbol Symbol Symbol Symbol Symbol Symbol Symbol AATF BMP8A CEBPE CTNNB1 ESR2 GDF3 HOXA5 IL17D ADIPOQ BRPF1 CEBPG CUX1 ESRRA GDF6 HOXA6 IL17F ADNP BRPF3 CERS1 CX3CL1 ETS1 GIN1 HOXA7 IL18 AEBP1 BUD31 CERS2 CXCL10 ETS2 GLIS3 HOXB1 IL19 AFF4 C17ORF77 CERS4 CXCL11 ETV3 GMEB1 HOXB13 IL1A AHR C1QTNF4 CFL2 CXCL12 ETV7 GPBP1 HOXB5 IL1B AIMP1 C21ORF66 CHIA CXCL13 FAM3B GPER HOXB6 IL1F3 ALS2CR8 CBFA2T2 CIR1 CXCL14 FAM3D GPI HOXB7 IL1F5 ALX1 CBFA2T3 CITED1 CXCL16 FASLG GREM1 HOXB9 IL1F6 ARGFX CBFB CITED2 CXCL3 FBLN1 GREM2 HOXC4 IL1F7 -

SUPPLEMENTARY MATERIAL Bone Morphogenetic Protein 4 Promotes
www.intjdevbiol.com doi: 10.1387/ijdb.160040mk SUPPLEMENTARY MATERIAL corresponding to: Bone morphogenetic protein 4 promotes craniofacial neural crest induction from human pluripotent stem cells SUMIYO MIMURA, MIKA SUGA, KAORI OKADA, MASAKI KINEHARA, HIROKI NIKAWA and MIHO K. FURUE* *Address correspondence to: Miho Kusuda Furue. Laboratory of Stem Cell Cultures, National Institutes of Biomedical Innovation, Health and Nutrition, 7-6-8, Saito-Asagi, Ibaraki, Osaka 567-0085, Japan. Tel: 81-72-641-9819. Fax: 81-72-641-9812. E-mail: [email protected] Full text for this paper is available at: http://dx.doi.org/10.1387/ijdb.160040mk TABLE S1 PRIMER LIST FOR QRT-PCR Gene forward reverse AP2α AATTTCTCAACCGACAACATT ATCTGTTTTGTAGCCAGGAGC CDX2 CTGGAGCTGGAGAAGGAGTTTC ATTTTAACCTGCCTCTCAGAGAGC DLX1 AGTTTGCAGTTGCAGGCTTT CCCTGCTTCATCAGCTTCTT FOXD3 CAGCGGTTCGGCGGGAGG TGAGTGAGAGGTTGTGGCGGATG GAPDH CAAAGTTGTCATGGATGACC CCATGGAGAAGGCTGGGG MSX1 GGATCAGACTTCGGAGAGTGAACT GCCTTCCCTTTAACCCTCACA NANOG TGAACCTCAGCTACAAACAG TGGTGGTAGGAAGAGTAAAG OCT4 GACAGGGGGAGGGGAGGAGCTAGG CTTCCCTCCAACCAGTTGCCCCAAA PAX3 TTGCAATGGCCTCTCAC AGGGGAGAGCGCGTAATC PAX6 GTCCATCTTTGCTTGGGAAA TAGCCAGGTTGCGAAGAACT p75 TCATCCCTGTCTATTGCTCCA TGTTCTGCTTGCAGCTGTTC SOX9 AATGGAGCAGCGAAATCAAC CAGAGAGATTTAGCACACTGATC SOX10 GACCAGTACCCGCACCTG CGCTTGTCACTTTCGTTCAG Suppl. Fig. S1. Comparison of the gene expression profiles of the ES cells and the cells induced by NC and NC-B condition. Scatter plots compares the normalized expression of every gene on the array (refer to Table S3). The central line -

Dlx5 and Dlx6 Expression in Gabaergic Neurons Controls Behavior, Metabolism, Healthy Aging and Lifespan
Dlx5 and Dlx6 expression in GABAergic neurons controls behavior, metabolism, healthy aging and lifespan. Camille de Lombares, Eglantine Heude, Gladys Alfama, Anastasia Fontaine, Rim Hassouna, Cecile Vernochet, Fabrice de Chaumont, Christophe Olivo-Marin, Elodie Ey, Sebastien Parnaudeau, et al. To cite this version: Camille de Lombares, Eglantine Heude, Gladys Alfama, Anastasia Fontaine, Rim Hassouna, et al.. Dlx5 and Dlx6 expression in GABAergic neurons controls behavior, metabolism, healthy aging and lifespan.. Aging, Impact Journals, 2019, 11 (17), pp.6638-6656. 10.18632/aging.102141. mnhn- 02295464 HAL Id: mnhn-02295464 https://hal-mnhn.archives-ouvertes.fr/mnhn-02295464 Submitted on 4 Feb 2020 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. Distributed under a Creative Commons Attribution| 4.0 International License www.aging-us.com AGING 2019, Vol. 11, No. 17 Research Paper Dlx5 and Dlx6 expression in GABAergic neurons controls behavior, metabolism, healthy aging and lifespan Camille de Lombares1, Eglantine Heude1, Gladys Alfama1, Anastasia Fontaine1, -

Supplemental Material.Pdf
Supplemental Material ZNF750 Interacts with KLF4 and RCOR1, KDM1A, and CTBP1/2 Chromatin Regulators to Repress Epidermal Progenitor Genes and Induce Differentiation Genes Lisa D. Boxer, Brook Barajas, Shiying Tao, Jiajing Zhang, and Paul Khavari Supplemental Inventory Figure S1. This figure supports Figure 1 and shows the Gene Ontology terms for ZNF750-bound but unaffected genes, the genomic enrichment of ZNF750 ChIP-seq peaks, and the percentage of peaks that contain the ZNF750 motif. Figure S2. This figure supports Figure 2 and shows gene expression changes with depletion of ZNF750-interacting proteins, confirms the knock-down of ZNF750-interacting proteins, and shows the quantification of Ki67 in ZNF750-interacting protein depleted organotypic tissue. Figure S3. This figure supports Figure 3 and shows the quantification of ZNF750 and interacting protein co-IPs, and IPs and Far western blots demonstrating competition between KLF4 and KDM1A for binding to ZNF750. Figure S4. This figure supports Figure 4 and shows the expression of ZNF750 mutant proteins and the effects of full-length or mutant ZNF750 on differentiation in organotypic tissue and on clonogenic growth. Figure S5. This figure supports Figure 5 and shows the effects of mutagenesis of ZNF750 and KLF4 motifs on reporter activity, and the changes in histone marks with depletion of ZNF750 and interacting proteins. Figure S6. This figure supports Figure 6 and shows the changes in mRNA and protein expression of ZNF750-interacting proteins during keratinocyte differentiation, and the expression of ZNF750-interacting proteins with ZNF750 depletion. Table S1. Supports Figure 1 and shows the genomic coordinates of ZNF750 ChIP-seq peaks. -

The Tumor Suppressor HHEX Inhibits Axon Growth When Prematurely Expressed in Developing Central Nervous System Neurons
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by epublications@Marquette Marquette University e-Publications@Marquette Biological Sciences Faculty Research and Biological Sciences, Department of Publications 9-1-2015 The umorT Suppressor HHEX Inhibits Axon Growth when Prematurely Expressed in Developing Central Nervous System Neurons Matthew .T Simpson Marquette University Ishwariya Venkatesh Marquette University Ben L. Callif Marquette University Laura K. Thiel Marquette University Denise M. Coley Marquette University See next page for additional authors Accepted version. Molecular and Cellular Neuroscience, Vol 68 )September 2015): 272-283. DOI. © 2015 Elsevier Inc. Used with permission. NOTICE: this is the author’s version of a work that was accepted for publication in Molecular and Cellular Neuroscience. Changes resulting from the publishing process, such as peer review, editing, corrections, structural formatting, and other quality control mechanisms may not be reflected in this document. Changes may have been made to this work since it was submitted for publication. A definitive version was subsequently published in Molecular and Cellular Neuroscience, Vol 68 )September 2015): 272-283. DOI. Authors Matthew T. Simpson, Ishwariya Venkatesh, Ben L. Callif, Laura K. Thiel, Denise M. Coley, Kristen N. Winsor, Zimei Wang, Audra A. Kramer, Jessica K. Lerch, and Murray G. Blackmore This article is available at e-Publications@Marquette: https://epublications.marquette.edu/bio_fac/515 NOT THE PUBLISHED VERSION; this is the author’s final, peer-reviewed manuscript. The published version may be accessed by following the link in the citation at the bottom of the page. The Tumor Suppressor HHEX Inhibits Axon Growth When Prematurely Expressed in Developing Central Nervous System Neurons Matthew T. -

DLX Genes: Roles in Development and Cancer
cancers Review DLX Genes: Roles in Development and Cancer Yinfei Tan 1,* and Joseph R. Testa 1,2,* 1 Genomics Facility, Fox Chase Cancer Center, Philadelphia, PA 19111, USA 2 Cancer Signaling and Epigenetics Program, Fox Chase Cancer Center, Philadelphia, PA 19111, USA * Correspondence: [email protected] (Y.T.); [email protected] (J.R.T.) Simple Summary: DLX homeobox family genes encode transcription factors that have indispensable roles in embryonic and postnatal development. These genes are critically linked to the morphogene- sis of craniofacial structures, branchial arches, forebrain, and sensory organs. DLX genes are also involved in postnatal homeostasis, particularly hematopoiesis and, when dysregulated, oncogen- esis. DLX1/2, DLX3/4, and DLX5/6 exist as bigenes on different chromosomes, sharing intergenic enhancers between gene pairs, which allows orchestrated spatiotemporal expression. Genomic alterations of human DLX gene enhancers or coding sequences result in congenital disorders such as split-hand/foot malformation. Aberrant postnatal expression of DLX genes is associated with hematological malignancies, including leukemias and lymphomas. In several mouse models of T-cell lymphoma, Dlx5 has been shown to act as an oncogene by cooperating with activated Akt, Notch1/3, and/or Wnt to drive tumor formation. In humans, DLX5 is aberrantly expressed in lung and ovarian carcinomas and holds promise as a therapeutic target. Abstract: Homeobox genes control body patterning and cell-fate decisions during development. The homeobox genes consist of many families, only some of which have been investigated regarding a possible role in tumorigenesis. Dysregulation of HOX family genes have been widely implicated in cancer etiology. -

Transcriptional Profiling of Mesc-Derived Tendon And
ARTICLE https://doi.org/10.1038/s41467-021-24535-5 OPEN Transcriptional profiling of mESC-derived tendon and fibrocartilage cell fate switch ✉ Deepak A. Kaji1, Angela M. Montero1, Roosheel Patel2 & Alice H. Huang 1 The transcriptional regulators underlying induction and differentiation of dense connective tissues such as tendon and related fibrocartilaginous tissues (meniscus and annulus fibrosus) remain largely unknown. Using an iterative approach informed by developmental cues and 1234567890():,; single cell RNA sequencing (scRNA-seq), we establish directed differentiation models to generate tendon and fibrocartilage cells from mouse embryonic stem cells (mESCs) by activation of TGFβ and hedgehog pathways, achieving 90% induction efficiency. Transcrip- tional signatures of the mESC-derived cells recapitulate embryonic tendon and fibrocartilage signatures from the mouse tail. scRNA-seq further identify retinoic acid signaling as a critical regulator of cell fate switch between TGFβ-induced tendon and fibrocartilage lineages. Tra- jectory analysis by RNA sequencing define transcriptional modules underlying tendon and fibrocartilage fate induction and identify molecules associated with lineage-specific differ- entiation. Finally, we successfully generate 3-dimensional engineered tissues using these differentiation protocols and show activation of mechanotransduction markers with dynamic tensile loading. These findings provide a serum-free approach to generate tendon and fibrocartilage cells and tissues at high efficiency for modeling development and -
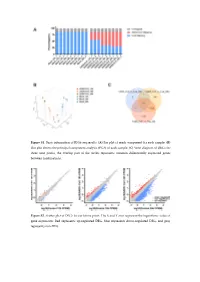
Figure S1. Basic Information of RNA-Seq Results. (A) Bar Plot of Reads Component for Each Sample
Figure S1. Basic information of RNA-seq results. (A) Bar plot of reads component for each sample. (B) Dot plot shows the principal component analysis (PCA) of each sample. (C) Venn diagram of DEGs for three time points, the overlap part of the circles represents common differentially expressed genes between combinations. Figure S2. Scatter plot of DEGs for each time point. The X and Y axes represent the logarithmic value of gene expression. Red represents up-regulated DEG, blue represents down-regulated DEG, and gray represents non-DEG. Table S1. Primers used for quantitative real-time PCR analysis of DEGs. Gene Primer Sequence Forward 5’-CTACGAGTGGATGGTCAAGAGC-3’ FOXO1 Reverse 5’-CCAGTTCCTTCATTCTGCACACG-3’ Forward 5’-GACGTCCGGCATCAGAGAAA-3’ IRS2 Reverse 5’-TCCACGGCTAATCGTCACAG-3’ Forward 5’-CACAACCAGGACCTCACACC-3’ IRS1 Reverse 5’-CTTGGCACGATAGAGAGCGT-3’ Forward 5’-AGGATACCACTCCCAACAGACCT-3’ IL6 Reverse 5’-CAAGTGCATCATCGTTGTTCATAC-3’ Forward 5’-TCACGTTGTACGCAGCTACC-3’ CCL5 Reverse 5’-CAGTCCTCTTACAGCCTTTGG-3’ Forward 5’-CTGTGCAGCCGCAGTGCCTACC-3’ BMP7 Reverse 5’-ATCCCTCCCCACCCCACCATCT-3’ Forward 5’-CTCTCCCCCTCGACTTCTGA-3’ BCL2 Reverse 5’-AGTCACGCGGAACACTTGAT-3’ Forward 5’-CTGTCGAACACAGTGGTACCTG-3’ FGF7 Reverse 5’-CCAACTGCCACTGTCCTGATTTC-3’ Forward 5’-GGGAGCCAAAAGGGTCATCA-3’ GAPDH Reverse 5’-CGTGGACTGTGGTCATGAGT-3’ Supplementary material: Differentially expressed genes log2(SADS-CoV_12h/ Qvalue (SADS-CoV _12h/ Gene Symbol Control_12h) Control_12h) PTGER4 -1.03693 6.79E-04 TMEM72 -3.08132 3.66E-04 IFIT2 -1.02918 2.11E-07 FRAT2 -1.09282 4.66E-05 -

Restoration of Sp4 in Forebrain Gabaergic Neurons Rescues Hypersensitivity to Ketamine in Sp4 Hypomorphic Mice Kerin K
International Journal of Neuropsychopharmacology, 2015, 1–9 doi:10.1093/ijnp/pyv063 Research Article research article Restoration of Sp4 in Forebrain GABAergic Neurons Rescues Hypersensitivity to Ketamine in Sp4 Hypomorphic Mice Kerin K. Higa, MS; Baohu Ji, PhD; Mahalah R. Buell, BA; Victoria B. Risbrough, PhD; Susan B. Powell, PhD; Jared W. Young, PhD; Mark A. Geyer, PhD; and Xianjin Zhou, PhD Department of Psychiatry, University of California San Diego, La Jolla, CA (Ms Higa, Drs Ji, Risbrough, Powell, Young, Geyer, and Zhou, and Ms Buell); Research Service, VA San Diego Healthcare System, La Jolla, CA (Drs Risbrough, Powell, Young, Geyer, and Zhou, and Ms Buell); Neurosciences Graduate Program, University of California San Diego, La Jolla, CA (Ms Higa). Correspondence: Xianjin Zhou, PhD, Department of Psychiatry, University of California San Diego, La Jolla, CA ([email protected]). Abstract Background: Ketamine produces schizophrenia-like behavioral phenotypes in healthy people. Prolonged ketamine effects and exacerbation of symptoms after the administration of ketamine have been observed in patients with schizophrenia. More recently, ketamine has been used as a potent antidepressant to treat patients with major depression. The genes and neurons that regulate behavioral responses to ketamine, however, remain poorly understood. Sp4 is a transcription factor for which gene expression is restricted to neuronal cells in the brain. Our previous studies demonstrated that Sp4 hypomorphic mice display several behavioral phenotypes relevant to psychiatric disorders, consistent with human SP4 gene associations with schizophrenia, bipolar disorder, and major depression. Among those behavioral phenotypes, hypersensitivity to ketamine- induced hyperlocomotion has been observed in Sp4 hypomorphic mice. Methods: In the present study, we used the Cre-LoxP system to restore Sp4 gene expression, specifically in either forebrain excitatory or GABAergic inhibitory neurons in Sp4 hypomorphic mice.