Mpiled Body of Work in Field Epidemiology
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
Districts of Ethiopia
Region District or Woredas Zone Remarks Afar Region Argobba Special Woreda -- Independent district/woredas Afar Region Afambo Zone 1 (Awsi Rasu) Afar Region Asayita Zone 1 (Awsi Rasu) Afar Region Chifra Zone 1 (Awsi Rasu) Afar Region Dubti Zone 1 (Awsi Rasu) Afar Region Elidar Zone 1 (Awsi Rasu) Afar Region Kori Zone 1 (Awsi Rasu) Afar Region Mille Zone 1 (Awsi Rasu) Afar Region Abala Zone 2 (Kilbet Rasu) Afar Region Afdera Zone 2 (Kilbet Rasu) Afar Region Berhale Zone 2 (Kilbet Rasu) Afar Region Dallol Zone 2 (Kilbet Rasu) Afar Region Erebti Zone 2 (Kilbet Rasu) Afar Region Koneba Zone 2 (Kilbet Rasu) Afar Region Megale Zone 2 (Kilbet Rasu) Afar Region Amibara Zone 3 (Gabi Rasu) Afar Region Awash Fentale Zone 3 (Gabi Rasu) Afar Region Bure Mudaytu Zone 3 (Gabi Rasu) Afar Region Dulecha Zone 3 (Gabi Rasu) Afar Region Gewane Zone 3 (Gabi Rasu) Afar Region Aura Zone 4 (Fantena Rasu) Afar Region Ewa Zone 4 (Fantena Rasu) Afar Region Gulina Zone 4 (Fantena Rasu) Afar Region Teru Zone 4 (Fantena Rasu) Afar Region Yalo Zone 4 (Fantena Rasu) Afar Region Dalifage (formerly known as Artuma) Zone 5 (Hari Rasu) Afar Region Dewe Zone 5 (Hari Rasu) Afar Region Hadele Ele (formerly known as Fursi) Zone 5 (Hari Rasu) Afar Region Simurobi Gele'alo Zone 5 (Hari Rasu) Afar Region Telalak Zone 5 (Hari Rasu) Amhara Region Achefer -- Defunct district/woredas Amhara Region Angolalla Terana Asagirt -- Defunct district/woredas Amhara Region Artuma Fursina Jile -- Defunct district/woredas Amhara Region Banja -- Defunct district/woredas Amhara Region Belessa -- -

Nigella Sativa) at the Oromia Regional State, Ethiopia
Asian Journal of Agricultural Extension, Economics & Sociology 31(3): 1-12, 2019; Article no.AJAEES.47315 ISSN: 2320-7027 Assessment of Production and Utilization of Black Cumin (Nigella sativa) at the Oromia Regional State, Ethiopia Wubeshet Teshome1 and Dessalegn Anshiso2* 1Ethiopian Biodiversity Institute, Horticulture and Crop Biodiversity Directorate, P.O.Box 30726; Addis Ababa, Ethiopia. 2College of Economics and Management, Huazhong Agricultural University, No. 1 Shizishan Street, Hongshan District, Wuhan, 430070, Hubei, P.R. China. Authors’ contributions This work was carried out in collaboration between both authors. Author WT managed the literature searches and participated in data collection. Author DA designed the study, performed the statistical analysis, wrote the protocol and wrote the first draft of the manuscript. Both authors read and approved the final manuscript. Article Information DOI: 10.9734/AJAEES/2019/v31i330132 Editor(s): (1) Prof. Fotios Chatzitheodoridis, Department of Agricultural Technology-Division of Agricultural Economics, Technological Education Institute of Western Macedonia, Greece. Reviewers: (1) Lawal Mohammad Anka, Development Project Samaru Gusau Zamfara State, Nigeria. (2) İsmail Ukav, Adiyaman University, Turkey. Complete Peer review History: http://www.sdiarticle3.com/review-history/47315 Received 14 November 2018 Accepted 09 February 2019 Original Research Article Published 06 April 2019 ABSTRACT Background and Objective: Black cuminseed for local consumption and other importance, such as oil and oil rosin for medicinal purposes, export market, crop diversification, income generation, reducing the risk of crop failure and others made it as a best alternative crop under Ethiopian smaller land holdings. The objectives of this study were to examine factors affecting farmer perception of the Black cumin production importance, and assess the crop utilization purpose by smallholder farmers and its income potential for the farmers in two Districts of Bale zone of Oromia regional state in Ethiopia. -

Hum Ethio Manitar Opia Rian Re Espons E Fund D
Hum anitarian Response Fund Ethiopia OCHA, 2011 OCHA, 2011 Annual Report 2011 Office for the Coordination of Humanitarian Affairs Humanitarian Response Fund – Ethiopia Annual Report 2011 Table of Contents Note from the Humanitarian Coordinator ................................................................................................ 2 Acknowledgements ................................................................................................................................. 3 1. Executive Summary ............................................................................................................................ 4 1.1 2011 Humanitarian Context ........................................................................................................... 4 1.2 Map - 2011 HRF Supported Projects ............................................................................................. 6 2. Information on Contributors ................................................................................................................ 7 2.1 Donor Contributions to HRF .......................................................................................................... 7 3. Fund Overview .................................................................................................................................... 8 3.1 Summary of HRF Allocations in 2011 ............................................................................................ 8 3.1.1 HRF Allocation by Sector ....................................................................................................... -

ETHIOPIA Food Security Outlook Update November 2011
ETHIOPIA Food Security Outlook Update November 2011 Good rains likely to stabilize food security in the south The October to December Deyr rains are performing well Figure 1. Most-likely food security outcomes (October in most parts of the southern and southeastern pastoral to December 2011) and agropastoral areas, easing the shortage of pastoral resources. This, coupled with ongoing humanitarian assistance, will continue to stabilize food security among poor and very poor households in these areas. Nonetheless, about 4 million people will continue to require humanitarian assistance through the end of 2011 across the country. Prices of staple foods have generally started declining following the fresh Meher harvest, although they remain higher than the five‐year average. This will continue to constrain access to food over the coming months among the rural and urban poor who heavily depend on purchase to fulfill their minimum food requirements. For more information on FEWS NET’s Food Insecurity Severity Scale, During the January to March 2012 period, Crisis level food please see: www.fews.net/FoodInsecurityScale insecurity will extend to the dominantly Belg producing Source: FEWS NET Ethiopia and WFP zones in the northeastern highlands as well as into some marginal Meher cropping areas due to the below normal Figure 2. Most‐likely food security outcomes (January to 2011 harvests. Similarly, as the long dry season March 2012) (December to March) progresses, deterioration in food security is likely in some southern pastoral and agropastoral woredas which were severely affected by the recent drought. Updated food security outlook through March 2012 Food security in most parts of the country has stabilized as a result of improved market supply and declining prices following the Meher harvest, ongoing humanitarian assistance, and the current good Deyr/Hageya rains in the southern and southeast pastoral and agropastoral. -

Aalborg Universitet Restructuring State and Society Ethnic
Aalborg Universitet Restructuring State and Society Ethnic Federalism in Ethiopia Balcha, Berhanu Publication date: 2007 Document Version Publisher's PDF, also known as Version of record Link to publication from Aalborg University Citation for published version (APA): Balcha, B. (2007). Restructuring State and Society: Ethnic Federalism in Ethiopia. SPIRIT. Spirit PhD Series No. 8 General rights Copyright and moral rights for the publications made accessible in the public portal are retained by the authors and/or other copyright owners and it is a condition of accessing publications that users recognise and abide by the legal requirements associated with these rights. ? Users may download and print one copy of any publication from the public portal for the purpose of private study or research. ? You may not further distribute the material or use it for any profit-making activity or commercial gain ? You may freely distribute the URL identifying the publication in the public portal ? Take down policy If you believe that this document breaches copyright please contact us at [email protected] providing details, and we will remove access to the work immediately and investigate your claim. Downloaded from vbn.aau.dk on: November 29, 2020 SPIRIT Doctoral Programme Aalborg University Kroghstraede 3-3.237 DK-9220 Aalborg East Phone: +45 9940 9810 Mail: [email protected] Restructuring State and Society: Ethnic Federalism in Ethiopia Berhanu Gutema Balcha SPIRIT PhD Series Thesis no. 8 ISSN: 1903-7783 © 2007 Berhanu Gutema Balcha Restructuring State and Society: Ethnic Federalism in Ethiopia SPIRIT – Doctoral Programme Aalborg University Denmark SPIRIT PhD Series Thesis no. -

11 HS 000 ETH 013013 A4.Pdf (English)
ETHIOPIA:Humanitarian Concern Areas Map (as of 04 February 2013) Eritrea > !ª !ª> Note: The following newly created woreda boundaries are not Tahtay !ª E available in the geo-database; hence not represented in this Nutrition Hotspot Priority Laelay Erob R R !ª Adiyabo Mereb Ahferom !ª Tahtay Gulomekeda !ª I E map regardless of their nutrition hot spot priority 1 & 2: Adiyabo Leke T D Adiyabo Adwa Saesie Dalul Priority one Asgede Tahtay R S Kafta Werei Tsaedaemba E E Priority 1: Dawa Sarar (Bale zone), Goro Dola (Guji zone), Abichu Tsimbila Maychew !ª A Humera Leke Hawzen Berahle A Niya( North Showa zone) and Burka Dintu (West Hararge Priority two > T I GR AY > Koneba Central Berahle zone) of Oromia region, Mekoy (Nuer zone) of Gambella Western Naeder Kola Ke>lete Awelallo Priority three Tselemti Adet Temben region, Kersadula and Raso (Afder zone), Ararso, Birkod, Tanqua > Enderta !ª Daror and Yo'ale (Degahabour zone), Kubi (Fik zone), Addi Tselemt Zone 2 No Priority given Arekay Abergele Southern Ab Ala Afdera Mersin (Korahe zone), Dhekasuftu and Mubarek (Liben Beyeda Saharti Erebti Debark Hintalo !ª zone), Hadigala (Shinille zone) and Daratole (Warder Abergele Samre > Megale Erebti Bidu Wejirat zone) of Somali region. Dabat Janamora > Bidu International Boundary Alaje Raya North Lay Sahla Azebo > Wegera Endamehoni > > Priority 2: Saba Boru (Guji zone) of Oromia region and Ber'ano Regional Boundary Gonder Armacho Ziquala > A FA R !ª East Sekota Raya Yalo Teru (Gode zone) and Tulu Guled (Jijiga zone) of Somali region. Ofla Kurri Belesa -

Oromia Region Administrative Map(As of 27 March 2013)
ETHIOPIA: Oromia Region Administrative Map (as of 27 March 2013) Amhara Gundo Meskel ! Amuru Dera Kelo ! Agemsa BENISHANGUL ! Jangir Ibantu ! ! Filikilik Hidabu GUMUZ Kiremu ! ! Wara AMHARA Haro ! Obera Jarte Gosha Dire ! ! Abote ! Tsiyon Jars!o ! Ejere Limu Ayana ! Kiremu Alibo ! Jardega Hose Tulu Miki Haro ! ! Kokofe Ababo Mana Mendi ! Gebre ! Gida ! Guracha ! ! Degem AFAR ! Gelila SomHbo oro Abay ! ! Sibu Kiltu Kewo Kere ! Biriti Degem DIRE DAWA Ayana ! ! Fiche Benguwa Chomen Dobi Abuna Ali ! K! ara ! Kuyu Debre Tsige ! Toba Guduru Dedu ! Doro ! ! Achane G/Be!ret Minare Debre ! Mendida Shambu Daleti ! Libanos Weberi Abe Chulute! Jemo ! Abichuna Kombolcha West Limu Hor!o ! Meta Yaya Gota Dongoro Kombolcha Ginde Kachisi Lefo ! Muke Turi Melka Chinaksen ! Gne'a ! N!ejo Fincha!-a Kembolcha R!obi ! Adda Gulele Rafu Jarso ! ! ! Wuchale ! Nopa ! Beret Mekoda Muger ! ! Wellega Nejo ! Goro Kulubi ! ! Funyan Debeka Boji Shikute Berga Jida ! Kombolcha Kober Guto Guduru ! !Duber Water Kersa Haro Jarso ! ! Debra ! ! Bira Gudetu ! Bila Seyo Chobi Kembibit Gutu Che!lenko ! ! Welenkombi Gorfo ! ! Begi Jarso Dirmeji Gida Bila Jimma ! Ketket Mulo ! Kersa Maya Bila Gola ! ! ! Sheno ! Kobo Alem Kondole ! ! Bicho ! Deder Gursum Muklemi Hena Sibu ! Chancho Wenoda ! Mieso Doba Kurfa Maya Beg!i Deboko ! Rare Mida ! Goja Shino Inchini Sululta Aleltu Babile Jimma Mulo ! Meta Guliso Golo Sire Hunde! Deder Chele ! Tobi Lalo ! Mekenejo Bitile ! Kegn Aleltu ! Tulo ! Harawacha ! ! ! ! Rob G! obu Genete ! Ifata Jeldu Lafto Girawa ! Gawo Inango ! Sendafa Mieso Hirna -

Ethiopia: Administrative Map (August 2017)
Ethiopia: Administrative map (August 2017) ERITREA National capital P Erob Tahtay Adiyabo Regional capital Gulomekeda Laelay Adiyabo Mereb Leke Ahferom Red Sea Humera Adigrat ! ! Dalul ! Adwa Ganta Afeshum Aksum Saesie Tsaedaemba Shire Indasilase ! Zonal Capital ! North West TigrayTahtay KoraroTahtay Maychew Eastern Tigray Kafta Humera Laelay Maychew Werei Leke TIGRAY Asgede Tsimbila Central Tigray Hawzen Medebay Zana Koneba Naeder Adet Berahile Region boundary Atsbi Wenberta Western Tigray Kelete Awelallo Welkait Kola Temben Tselemti Degua Temben Mekele Zone boundary Tanqua Abergele P Zone 2 (Kilbet Rasu) Tsegede Tselemt Mekele Town Special Enderta Afdera Addi Arekay South East Ab Ala Tsegede Mirab Armacho Beyeda Woreda boundary Debark Erebti SUDAN Hintalo Wejirat Saharti Samre Tach Armacho Abergele Sanja ! Dabat Janamora Megale Bidu Alaje Sahla Addis Ababa Ziquala Maychew ! Wegera Metema Lay Armacho Wag Himra Endamehoni Raya Azebo North Gondar Gonder ! Sekota Teru Afar Chilga Southern Tigray Gonder City Adm. Yalo East Belesa Ofla West Belesa Kurri Dehana Dembia Gonder Zuria Alamata Gaz Gibla Zone 4 (Fantana Rasu ) Elidar Amhara Gelegu Quara ! Takusa Ebenat Gulina Bugna Awra Libo Kemkem Kobo Gidan Lasta Benishangul Gumuz North Wello AFAR Alfa Zone 1(Awsi Rasu) Debre Tabor Ewa ! Fogera Farta Lay Gayint Semera Meket Guba Lafto DPubti DJIBOUTI Jawi South Gondar Dire Dawa Semen Achefer East Esite Chifra Bahir Dar Wadla Delanta Habru Asayita P Tach Gayint ! Bahir Dar City Adm. Aysaita Guba AMHARA Dera Ambasel Debub Achefer Bahirdar Zuria Dawunt Worebabu Gambela Dangura West Esite Gulf of Aden Mecha Adaa'r Mile Pawe Special Simada Thehulederie Kutaber Dangila Yilmana Densa Afambo Mekdela Tenta Awi Dessie Bati Hulet Ej Enese ! Hareri Sayint Dessie City Adm. -

Ethiopia: 3W - Health Cluster Ongoing Activities Map (December 2016)
Ethiopia: 3W - Health Cluster Ongoing Activities map (December 2016) ERITREA 8 Total Number of Partners Ahferom CCM CCM GOAL GOAL Erob CCM Adwa GOAL Red Sea GOAL Werei CCM Leke GOAL Koneba GOAL Hawzen GOAL CCM SUDAN TIGRAY GOAL Ab Ala GOAL AMHARA Megale Gulf of GOAL Aden DCA IMC Kobo AFAR Lay DCA Meket DCA Gayint IMC IMC Tach Gayint DCA Guba Lafto GOAL BENESHANGUL Dera IMC Worebabu Simada GOAL GOAL GOAL GOAL GUMU IMC Thehulederie Sirba DJIBOUTI Abay Telalak Afambo GOAL GOAL IRC Tenta GOAL Sayint GOAL GOAL IRC GOAL GOAL Were Ilu Ayisha IRC IRC GOAL Dewa Sherkole Legehida Harewa Kurmuk GOAL IMC Menge Kelela Artuma IRC Yaso Fursi IMC Erer IRC IRC IRC Jille Menz IMC Timuga Dembel Wara Afdem Bilidigilu IRC Mama Assosa IRC Jarso IMC Tarema IMC Midir Ber IRC IRC Agalometi Gerar IMC Jarso Kamashi IMC Bambasi GOAL DIRE Chinaksen IMC IMC IRC IMC DAWA IMC Bio Jiganifado Ankober Meta IRC GOAL IRC IMC IMC Aleltu Deder HARERI GOAL GOAL Gursum IRC IRC IRC GOAL Midega SOMALIA IRC IMC Goba SOUTH SUDAN Tola ACF Koricha Anfilo IMC Gashamo Anchar GOAL Daro Lebu Boke Golo Oda IRC Wantawo GOAL Meyu IMC IRC IRC IRC GOAL GOAL IMC Aware SCI IMC Fik IRC IRC Kokir Sire Jikawo IRC Gedbano Adami IMC GOAL Tulu Jido Degehabur GOAL SCI GOAL Sude Akobo Selti Kombolcha IRC IRC Lanfero Hamero Gunagado Mena Dalocha IMC GAMBELA GOAL Arsi IMC Shekosh GOAL Gololcha GOAL Negele Bale IMC Soro GOAL IMC IRC GOAL IMC Agarfa IRC Tembaro IRC IRC GOAL SCI GOAL GOAL IMC IMC Ginir CCM GOAL GOAL IRC IMC IMC GOAL GOAL IRC GOAL Sinana IMC IRC IRC Dinsho GOAL Goba IRC IMC GOAL IRC GOAL IRC Adaba CCM GOAL Berbere IMC Humbo GOAL SOMALI IMC Hulla IRC GOAL CCM GOAL GOAL GOAL PIN IRC Zala IMC IRC IRC Abaya PIN IRC Wenago Ubadebretsehay Mirab Gelana Abaya IRC GOAL GOAL SCI IRC IRC SCI Amaro OROMIA SNNPR IRC SCI CCM Bonke GOAL IRC Meda CCM SCI Welabu Legend SCI Konso IMC SCI International boundary Filtu Hudet INDIAN Agencies' locOaCtiEoAnNs and Regional boundary SCI Arero Dolobay Dolo Odo area of interventions are IMC No. -
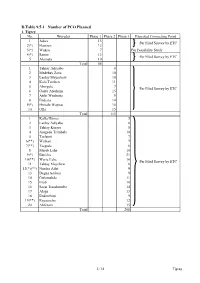
D.Table 9.5-1 Number of PCO Planned 1
D.Table 9.5-1 Number of PCO Planned 1. Tigrey No. Woredas Phase 1 Phase 2 Phase 3 Expected Connecting Point 1 Adwa 13 Per Filed Survey by ETC 2(*) Hawzen 12 3(*) Wukro 7 Per Feasibility Study 4(*) Samre 13 Per Filed Survey by ETC 5 Alamata 10 Total 55 1 Tahtay Adiyabo 8 2 Medebay Zana 10 3 Laelay Mayechew 10 4 Kola Temben 11 5 Abergele 7 Per Filed Survey by ETC 6 Ganta Afeshum 15 7 Atsbi Wenberta 9 8 Enderta 14 9(*) Hintalo Wajirat 16 10 Ofla 15 Total 115 1 Kafta Humer 5 2 Laelay Adiyabo 8 3 Tahtay Koraro 8 4 Asegede Tsimbela 10 5 Tselemti 7 6(**) Welkait 7 7(**) Tsegede 6 8 Mereb Lehe 10 9(*) Enticho 21 10(**) Werie Lehe 16 Per Filed Survey by ETC 11 Tahtay Maychew 8 12(*)(**) Naeder Adet 9 13 Degua temben 9 14 Gulomahda 11 15 Erob 10 16 Saesi Tsaedaemba 14 17 Alage 13 18 Endmehoni 9 19(**) Rayaazebo 12 20 Ahferom 15 Total 208 1/14 Tigrey D.Table 9.5-1 Number of PCO Planned 2. Affar No. Woredas Phase 1 Phase 2 Phase 3 Expected Connecting Point 1 Ayisaita 3 2 Dubti 5 Per Filed Survey by ETC 3 Chifra 2 Total 10 1(*) Mile 1 2(*) Elidar 1 3 Koneba 4 4 Berahle 4 Per Filed Survey by ETC 5 Amibara 5 6 Gewane 1 7 Ewa 1 8 Dewele 1 Total 18 1 Ere Bti 1 2 Abala 2 3 Megale 1 4 Dalul 4 5 Afdera 1 6 Awash Fentale 3 7 Dulecha 1 8 Bure Mudaytu 1 Per Filed Survey by ETC 9 Arboba Special Woreda 1 10 Aura 1 11 Teru 1 12 Yalo 1 13 Gulina 1 14 Telalak 1 15 Simurobi 1 Total 21 2/14 Affar D.Table 9.5-1 Number of PCO Planned 3. -

Periodic Monitoring Report Working 2016 Humanitarian Requirements Document – Ethiopia Group
DRMTechnical Periodic Monitoring Report Working 2016 Humanitarian Requirements Document – Ethiopia Group Covering 1 Jan to 31 Dec 2016 Prepared by Clusters and NDRMC Introduction The El Niño global climactic event significantly affected the 2015 meher/summer rains on the heels of failed belg/ spring rains in 2015, driving food insecurity, malnutrition and serious water shortages in many parts of the country. The Government and humanitarian partners issued a joint 2016 Humanitarian Requirements Document (HRD) in December 2015 requesting US$1.4 billion to assist 10.2 million people with food, health and nutrition, water, agriculture, shelter and non-food items, protection and emergency education responses. Following the delay and erratic performance of the belg/spring rains in 2016, a Prioritization Statement was issued in May 2016 with updated humanitarian requirements in nutrition (MAM), agriculture, shelter and non-food items and education.The Mid-Year Review of the HRD identified 9.7 million beneficiaries and updated the funding requirements to $1.2 billion. The 2016 HRD is 69 per cent funded, with contributions of $1.08 billion from international donors and the Government of Ethiopia (including carry-over resources from 2015). Under the leadership of the Government of Ethiopia delivery of life-saving and life- sustaining humanitarian assistance continues across the sectors. However, effective humanitarian response was challenged by shortage of resources, limited logistical capacities and associated delays, and weak real-time information management. This Periodic Monitoring Report (PMR) provides a summary of the cluster financial inputs against outputs and achievements against cluster objectives using secured funding since the launch of the 2016 HRD. -

Ethiopia Administrative Map As of 2013
(as of 27 March 2013) ETHIOPIA:Administrative Map R E Legend E R I T R E A North D Western \( Erob \ Tahtay Laelay National Capital Mereb Ahferom Gulomekeda Adiyabo Adiyabo Leke Central Ganta S Dalul P Afeshum Saesie Tahtay Laelay Adwa E P Tahtay Tsaedaemba Regional Capital Kafta Maychew Maychew Koraro Humera Asgede Werei Eastern A Leke Hawzen Tsimbila Medebay Koneba Zana Kelete Berahle Western Atsbi International Boundary Welkait Awelallo Naeder Tigray Wenberta Tselemti Adet Kola Degua Tsegede Temben Mekele Temben P Zone 2 Undetermined Boundary Addi Tselemt Tanqua Afdera Abergele Enderta Arekay Ab Ala Tsegede Beyeda Mirab Armacho Debark Hintalo Abergele Saharti Erebti Regional Boundary Wejirat Tach Samre Megale Bidu Armacho Dabat Janamora Alaje Lay Sahla Zonal Boundary Armacho Wegera Southern Ziquala Metema Sekota Endamehoni Raya S U D A N North Wag Azebo Chilga Yalo Amhara East Ofla Teru Woreda Boundary Gonder West Belesa Himra Kurri Gonder Dehana Dembia Belesa Zuria Gaz Alamata Zone 4 Quara Gibla Elidar Takusa I Libo Ebenat Gulina Lake Kemkem Bugna Kobo Awra Afar T Lake Tana Lasta Gidan (Ayna) Zone 1 0 50 100 200 km Alfa Ewa U Fogera North Farta Lay Semera ¹ Meket Guba Lafto Semen Gayint Wollo P O Dubti Jawi Achefer Bahir Dar East Tach Wadla Habru Chifra B G U L F O F A D E N Delanta Aysaita Creation date:27 Mar.2013 P Dera Esite Gayint I Debub Bahirdar Ambasel Dawunt Worebabu Map Doc Name:21_ADM_000_ETH_032713_A0 Achefer Zuria West Thehulederie J Dangura Simada Tenta Sources:CSA (2007 population census purpose) and Field Pawe Mecha