Milnacipran for Pain in Fibromyalgia in Adults
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Formulary and Prescribing Guidelines
Formulary and Prescribing Guidelines SECTION 1: TREATMENT OF DEPRESSION Section 1. Treatment of depression 1.1 Introduction This guidance should be considered as part of a stepped care approach in the management of depressive disorders. Antidepressants are not routinely recommended for persistent sub-threshold depressive symptoms or mild depression but can be considered in these categories where there is a past history of moderate or severe depression, initial presentation of sub-threshold depressive symptoms for at least 2 years, and persistence of either mild or sub-threshold depression after other interventions1 have failed. The most current NICE guidance should be consulted wherever possible to obtain the most up to date information. For individuals with moderate or severe depression, a combination of antidepressant medication and a high intensity psychological intervention (CBT or IPT) is recommended. When depression is accompanied by symptoms of anxiety, usually treat the depression first. If the person has an anxiety disorder and co-morbid depression or depressive symptoms, consider treating the anxiety first. Also consider offering advice on sleep hygiene, by way of establishing regular sleep and wake times; avoiding excess eating, avoiding smoking and drinking alcohol before sleep; and taking regular physical exercise if possible. Detailed information on the treatment of depression in children and adolescents can be found in section 12. Further guidance on prescribing for older adults and for antenatal/postnatal service users can be found in section 11 and section 20, respectively. 1.2 Approved Drugs for the treatment of Depression in Adults For licensing indications, see Annex 1 Drug5 Formulation5 Comments2,3,4 Caps 20mg, 60mg Selective serotonin reuptake inhibitor (SSRI) Fluoxetine Liquid 20mg/5ml 1st line SSRI (based on acquisition cost) Tabs 10mg, 25mg, 50mg Amitriptyline Tricyclic (TCA). -

Levomilnacipran for the Treatment of Major Depressive Disorder
Out of the Pipeline Levomilnacipran for the treatment of major depressive disorder Matthew Macaluso, DO, Hala Kazanchi, MD, and Vikram Malhotra, MD An SNRI with n July 2013, the FDA approved levomil- Table 1 once-daily dosing, nacipran for the treatment of major de- Levomilnacipran: Fast facts levomilnacipran pressive disorder (MDD) in adults.1 It is I Brand name: Fetzima decreased core available in a once-daily, extended-release formulation (Table 1).1 The drug is the fifth Class: Serotonin-norepinephrine reuptake symptoms of inhibitor serotonin-norepinephrine reuptake inhibi- MDD and was well Indication: Treatment of major depressive tor (SNRI) to be sold in the United States disorder in adults tolerated in clinical and the fourth to receive FDA approval for FDA approval date: July 26, 2013 trials treating MDD. Availability date: Fourth quarter of 2013 Levomilnacipran is believed to be the Manufacturer: Forest Pharmaceuticals more active enantiomer of milnacipran, Dosage forms: Extended–release capsules in which has been available in Europe for 20 mg, 40 mg, 80 mg, and 120 mg strengths years and was approved by the FDA in Recommended dosage: 40 mg to 120 mg 2009 for treating fibromyalgia. Efficacy of capsule once daily with or without food levomilnacipran for treating patients with Source: Reference 1 MDD was established in three 8-week ran- domized controlled trials (RCTs).1 cial and occupational functioning in addi- Clinical implications tion to improvement in the core symptoms Levomilnacipran is indicated for treating of depression.5 -

Milnacipran Remediates Impulsive Deficits in Rats with Lesions of the Ventromedial Prefrontal Cortex
Title Milnacipran Remediates Impulsive Deficits in Rats with Lesions of the Ventromedial Prefrontal Cortex Author(s) Tsutsui-Kimura, Iku; Yoshida, Takayuki; Ohmura, Yu; Izumi, Takeshi; Yoshioka, Mitsuhiro International journal of neuropsychopharmacology, 18(5), pyu083 Citation https://doi.org/10.1093/ijnp/pyu083 Issue Date 2015-03 Doc URL http://hdl.handle.net/2115/59283 Rights(URL) http://creativecommons.org/licenses/by-nc/4.0 Type article File Information pyu083.full.pdf Instructions for use Hokkaido University Collection of Scholarly and Academic Papers : HUSCAP International Journal of Neuropsychopharmacology, 2015, 1–14 doi:10.1093/ijnp/pyu083 Research Article research article Milnacipran Remediates Impulsive Deficits in Rats with Lesions of the Ventromedial Prefrontal Cortex Iku Tsutsui-Kimura, PhD; Takayuki Yoshida, PhD; Yu Ohmura, PhD; Takeshi Izumi, MD, PhD; Mitsuhiro Yoshioka, MD, PhD Department of Neuropharmacology, Hokkaido University Graduate School of Medicine, Sapporo, Japan (Drs Tsutsui-Kimura, Yoshida, Ohmura, Izumi, and Yoshioka); Japan Society for the Promotion of Science, Tokyo, Japan (Dr Tsutsui-Kimura); Department of Neuropsychiatry, Keio University School of Medicine, Tokyo, Japan (Dr Tsutsui-Kimura). Correspondence: Yu Ohmura, PhD, Department of Neuropharmacology, Hokkaido University Graduate School of Medicine, N15 W7, Kita-ku, Sapporo, 060-8638, Japan ([email protected]). Abstract Background: Deficits in impulse control are often observed in psychiatric disorders in which abnormalities of the prefrontal cortex are observed, including attention-deficit/hyperactivity disorder and bipolar disorder. We recently found that milnacipran, a serotonin/noradrenaline reuptake inhibitor, could suppress impulsive action in normal rats. However, whether milnacipran could suppress elevated impulsive action in rats with lesions of the ventromedial prefrontal cortex, which is functionally comparable with the human prefrontal cortex, remains unknown. -

Duloxetine for Chronic Pain Relief
Patient Information Leaflet Duloxetine for Chronic Pain Relief Produced By: Chronic Pain Service April 2014 Review due April 2017 1 If you require this leaflet in another language, large print or another format, please contact the Quality Team, telephone 01983 534850, who will advise you. Duloxetine ( Cymbalta ) is a medicine used to relieve chronic pain and treat low mood. It is often helpful for nerve-related pain or pain sensitivity (also called central sensitisation ). It has a different way of relieving pain than standard pain killers and is often prescribed in combination. If used for pain other than diabetes-related neuropathic pain it is formally used outside it’s product license as per individual decision by your pain specialist/doctor. As it affects the central nervous system in complex ways, the effect often takes a while to be felt and requires regular intake . We recommend a trial of at least four weeks to judge the effect. If good, it should be taken on a regular long-term basis; if there is no distinct relief it should be discontinued after the trial period. If you suffer with seizures/epilepsy, glaucoma and poorly controlled high blood pressure, Duloxetine should be used with caution and at a lower dose (up to 60mg per day). The most common side effect is drowsiness/dizziness therefore it is best time to take it in the evening. There are many other possible side effects, but most are uncommon, rare or very rare (please take a look at the product leaflet) We suggest that you avoid driving for at least 2 days after starting or increasing the dose of this medication. -
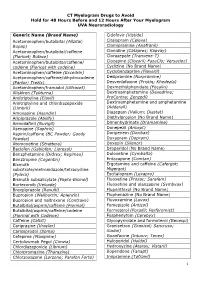
CT Myelogram Drugs to Avoid Hold for 48 Hours Before and 12 Hours After Your Myelogram UVA Neuroradiology
CT Myelogram Drugs to Avoid Hold for 48 Hours Before and 12 Hours After Your Myelogram UVA Neuroradiology Generic Name (Brand Name) Cidofovir (Vistide) Acetaminophen/butalbital (Allzital; Citalopram (Celexa) Bupap) Clomipramine (Anafranil) Acetaminophen/butalbital/caffeine Clonidine (Catapres; Kapvay) (Fioricet; Butace) Clorazepate (Tranxene-T) Acetaminophen/butalbital/caffeine/ Clozapine (Clozaril; FazaClo; Versacloz) codeine (Fioricet with codeine) Cyclizine (No Brand Name) Acetaminophen/caffeine (Excedrin) Cyclobenzaprine (Flexeril) Acetaminophen/caffeine/dihydrocodeine Desipramine (Norpramine) (Panlor; Trezix) Desvenlafaxine (Pristiq; Khedezla) Acetaminophen/tramadol (Ultracet) Dexmethylphenidate (Focalin) Aliskiren (Tekturna) Dextroamphetamine (Dexedrine; Amitriptyline (Elavil) ProCentra; Zenzedi) Amitriptyline and chlordiazepoxide Dextroamphetamine and amphetamine (Limbril) (Adderall) Amoxapine (Asendin) Diazepam (Valium; Diastat) Aripiprazole (Abilify) Diethylpropion (No Brand Name) Armodafinil (Nuvigil) Dimenhydrinate (Dramamine) Asenapine (Saphris) Donepezil (Aricept) Aspirin/caffeine (BC Powder; Goody Doripenem (Doribax) Powder) Doxapram (Dopram) Atomoxetine (Strattera) Doxepin (Silenor) Baclofen (Gablofen; Lioresal) Droperidol (No Brand Name) Benzphetamine (Didrex; Regimex) Duloxetine (Cymbalta) Benztropine (Cogentin) Entacapone (Comtan) Bismuth Ergotamine and caffeine (Cafergot; subcitrate/metronidazole/tetracycline Migergot) (Pylera) Escitalopram (Lexapro) Bismuth subsalicylate (Pepto-Bismol) Fluoxetine (Prozac; Sarafem) -

Adverse Effects of First-Line Pharmacologic Treatments of Major Depression in Older Adults
Draft Comparative Effectiveness Review Number xx Adverse Effects of First-line Pharmacologic Treatments of Major Depression in Older Adults Prepared for: Agency for Healthcare Research and Quality U.S. Department of Health and Human Services 5600 Fishers Lane Rockville, MD 20857 www.ahrq.gov This information is distributed solely for the purposes of predissemination peer review. It has not been formally disseminated by the Agency for Healthcare Research and Quality. The findings are subject to change based on the literature identified in the interim and peer-review/public comments and should not be referenced as definitive. It does not represent and should not be construed to represent an Agency for Healthcare Research and Quality or Department of Health and Human Services (AHRQ) determination or policy. Contract No. 290-2015-00012I Prepared by: Will be included in the final report Investigators: Will be included in the final report AHRQ Publication No. xx-EHCxxx <Month, Year> ii Purpose of the Review To assess adverse events of first-line antidepressants in the treatment of major depressive disorder in adults 65 years or older. Key Messages • Acute treatment (<12 weeks) with o Serotonin norepinephrine reuptake inhibitors (SNRIs) (duloxetine, venlafaxine), but not selective serotonin reuptake inhibitors (SSRIs) (escitalopram, fluoxetine) led to a greater number of adverse events compared with placebo. o SSRIs (citalopram, escitalopram and fluoxetine) and SNRIs (duloxetine and venlafaxine) led to a greater number of patients withdrawing from studies due to adverse events compared with placebo o Details of the contributing adverse events in RCTs were rarely reported to more clearly characterize what adverse events to expect. -

Efficacy of Milnacipran in Patients with Fibromyalgia R
Efficacy of Milnacipran in Patients with Fibromyalgia R. MICHAEL GENDREAU, MICHAEL D. THORN, JUDY F. GENDREAU, JAY D. KRANZLER, SAULO RIBEIRO, RICHARD H. GRACELY, DAVID A. WILLIAMS, PHILIP J. MEASE, SAMUEL A. McLEAN, and DANIEL J. CLAUW ABSTRACT. Objective. Fibromyalgia (FM) is a common musculoskeletal condition characterized by widespread pain, tenderness, and a variety of other somatic symptoms. Current treatments are modestly effec- tive. Arguably, the best studied and most effective compounds are tricyclic antidepressants (TCA). Milnacipran, a nontricyclic compound that inhibits the reuptake of both serotonin and norepineph- rine, may provide many of the beneficial effects of TCA with a superior side effect profile. Methods. One hundred twenty-five patients with FM were randomly assigned in a 3:3:2 ratio to receive milnacipran twice daily, milnacipran once daily, or placebo for 3 months in a double-blind dose-escalation trial; 92% of twice-daily and 81% of once-daily participants achieved dose escala- tion to the target milnacipran dose of 200 mg. Results. The primary endpoint was reduction of pain. Both the once- and twice-daily groups showed statistically significant improvements in pain, as well as improvements in global well being, fatigue, and other domains. Response rates for patients receiving milnacipran were equal in patients with and without comorbid depression, but placebo response rates were considerably higher in depressed patients, leading to significantly greater overall efficacy in the nondepressed group. Conclusion. In this Phase II study, milnacipran led to statistically significant improvements in pain and other symptoms of FM. The effect sizes were equal to those previously found with TCA, and the drug was generally well tolerated. -

Cymbalta (Duloxetine Hcl) Prior Authorization of Benefits Form
Cymbalta (Duloxetine HCl) Prior Authorization of Benefits Form CONTAINS CONFIDENTIAL PATIENT INFORMATION Complete form in its entirety and fax to: Prior Authorization of Benefits Center at 844-512-9005 for retail pharmacy or 844-512-7027 for medical injectable. 1. Patient information 2. Physician information Patient name: _________________________________ Prescribing physician: _______________________________ Patient ID #: __________________________________ Physician address: _________________________________ Patient DOB: _________________________________ Physician phone #: _________________________________ Date of Rx: ___________________________________ Physician fax #: ____________________________________ Patient phone #: _______________________________ Physician specialty: _________________________________ Patient email address: __________________________ Physician DEA: ____________________________________ Physician NPI #: ___________________________________ Physician email address: ____________________________ 3. Medication 4. Strength 5. Directions 6. Quantity per 30 days Cymbalta (Duloxetine HCl) ______________________ _____________________ Specify: ________________________ 7. Diagnosis: ____________________________________________________________________________________ 8. Approval criteria: (Check all boxes that apply. Note: Any areas not filled out are considered not applicable to your patient and may affect the outcome of this request.) Major depressive disorder (MDD), depressive disorder or dysthymia: □ Yes □ No Patient has -

View Rome, Italy Final Program
8TH INTERNATIONAL CONGRESS OF PARKINSON’S DISEASE AND MOVEMENT DISORDERS CONGRESS OF PARKINSON’S DISEASE 8TH INTERNATIONAL TABLE OF CONTENTS Invitation . .2 Acknowledgements . .4 Organization . .5 MDS Committees and Task Forces . .8 International Congress Registration and Venue . 11 International Congress Information . .12 Continuing Medical Education . .12 Evaluations . .12 Press Room . .13 Social Events . .14 Program at a Glance . .17 Scientific Sessions . .18 Faculty . .38 Committee and Task Force Meetings . .41 Exhibitor Information and Directory . .42 Floor Plan and Meeting Space . .46 Map of Rome . .51 Poster Session 1 . .52 Poster Session 2 . .63 Poster Session 3 . .73 Poster Session 4 . .83 THE CONGRESS IS UNDER THE AUSPICES OF: The President of the Italian Republic The Prime Minister of the Italian government The National Institute of Health (ISS) The National Research Council (CNR) The City of Rome The School of Medicine and Surgery, University of Rome “La Sapienza” The Italian Society of Neurology 1 THE MOVEMENT DISORDER SOCIETY INVITATION Dear Colleagues, Rome, Italy On behalf of the Officers and International Executive Committee of The Movement Disorder Society, welcome to the 8th International Congress of Parkinson’s Disease and Movement Disorders. I would like to thank the faculty and the members of the Congress Scientific Program Committee for this exemplary scientific program and for their contribution to the International Congress. The International Congress week begins with a variety of Kickoff Seminars, which are supported through unrestricted educational grants from industry. The week continues with a wide array of plenary sessions, parallel sessions, seminars and video dinners. Poster sessions are unopposed and, to further serve our participants, time has also been allotted for 16 poster platform presentations. -

Anticonvulsants Antipsychotics Benzodiazepines/Anxiolytics ADHD
Anticonvulsants Antidepressants Generic Name Brand Name Generic Name Brand Name Carbamazepine Tegretol SSRI Divalproex Depakote Citalopram Celexa Lamotrigine Lamictal Escitalopram Lexapro Topirimate Topamax Fluoxetine Prozac Fluvoxamine Luvox Paroxetine Paxil Antipsychotics Sertraline Zoloft Generic Name Brand Name SNRI Typical Desvenlafaxine Pristiq Chlorpromazine Thorazine Duloextine Cymbalta Fluphenazine Prolixin Milnacipran Savella Haloperidol Haldol Venlafaxine Effexor Perphenazine Trilafon SARI Atypical Nefazodone Serzone Aripiprazole Abilify Trazodone Desyrel Clozapine Clozaril TCA Lurasidone Latuda Clomimpramine Enafranil Olanzapine Zyprexa Despiramine Norpramin Quetiapine Seroquel Nortriptyline Pamelor Risperidone Risperal MAOI Ziprasidone Geodon Phenylzine Nardil Selegiline Emsam Tranylcypromine Parnate Benzodiazepines/Anxiolytics DNRI Generic Name Brand Name Bupropion Wellbutrin Alprazolam Xanax Clonazepam Klonopin Sedative‐Hypnotics Lorazepam Ativan Generic Name Brand Name Diazepam Valium Clonidine Kapvay Buspirone Buspar Eszopiclone Lunesta Pentobarbital Nembutal Phenobarbital Luminal ADHD Medicines Zaleplon Sonata Generic Name Brand Name Zolpidem Ambien Stimulant Amphetamine Adderall Others Dexmethylphenidate Focalin Generic Name Brand Name Dextroamphetamine Dexedrine Antihistamine Methylphenidate Ritalin Non‐stimulant Diphenhydramine Bendryl Atomoxetine Strattera Anti‐tremor Guanfacine Intuniv Benztropine Cogentin Mood stabilizer (bipolar treatment) Lithium Lithane Never Mix, Never Worry: What Clinicians Need to Know about -

Medication Guide Duloxetine Delayed-Release Capsules, USP (Doo-LOX
Medication Guide Duloxetine Delayed-Release Capsules, USP (doo-LOX-e-teen) Read this Medication Guide before you start taking duloxetine delayed-release capsules and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or treatment. Talk to your healthcare provider about: • all risks and benefits of treatment with antidepressant medicines • all treatment choices for depression or other serious mental illness What is the most important information I should know about antidepressant medicines, depression, other serious mental illnesses, and suicidal thoughts or actions? 1. Duloxetine delayed-release capsules and other antidepressant medicines may increase suicidal thoughts or actions in some children, teenagers, or young adults within the first few months of treatment or when the dose is changed. 2. Depression and other serious mental illnesses are the most important causes of suicidal thoughts or actions. Some people may have a particularly high risk of having suicidal thoughts or actions. These include people who have (or have a family history of) bipolar illness (also called manic-depressive illness). 3. How can I watch for and try to prevent suicidal thoughts and actions? • Pay close attention to any changes in mood, behavior, actions, thoughts, or feelings, especially sudden changes. This is very important when an antidepressant medicine is started or when the dose is changed. • Call your healthcare provider right away to report new or sudden changes in mood, behavior, thoughts, or feelings. • Keep all follow-up visits with your healthcare provider as scheduled. -

(12) Patent Application Publication (10) Pub. No.: US 2015/0202317 A1 Rau Et Al
US 20150202317A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2015/0202317 A1 Rau et al. (43) Pub. Date: Jul. 23, 2015 (54) DIPEPTDE-BASED PRODRUG LINKERS Publication Classification FOR ALPHATIC AMNE-CONTAINING DRUGS (51) Int. Cl. A647/48 (2006.01) (71) Applicant: Ascendis Pharma A/S, Hellerup (DK) A638/26 (2006.01) A6M5/9 (2006.01) (72) Inventors: Harald Rau, Heidelberg (DE); Torben A 6LX3/553 (2006.01) Le?mann, Neustadt an der Weinstrasse (52) U.S. Cl. (DE) CPC ......... A61K 47/48338 (2013.01); A61 K3I/553 (2013.01); A61 K38/26 (2013.01); A61 K (21) Appl. No.: 14/674,928 47/48215 (2013.01); A61M 5/19 (2013.01) (22) Filed: Mar. 31, 2015 (57) ABSTRACT The present invention relates to a prodrug or a pharmaceuti Related U.S. Application Data cally acceptable salt thereof, comprising a drug linker conju (63) Continuation of application No. 13/574,092, filed on gate D-L, wherein D being a biologically active moiety con Oct. 15, 2012, filed as application No. PCT/EP2011/ taining an aliphatic amine group is conjugated to one or more 050821 on Jan. 21, 2011. polymeric carriers via dipeptide-containing linkers L. Such carrier-linked prodrugs achieve drug releases with therapeu (30) Foreign Application Priority Data tically useful half-lives. The invention also relates to pharma ceutical compositions comprising said prodrugs and their use Jan. 22, 2010 (EP) ................................ 10 151564.1 as medicaments. US 2015/0202317 A1 Jul. 23, 2015 DIPEPTDE-BASED PRODRUG LINKERS 0007 Alternatively, the drugs may be conjugated to a car FOR ALPHATIC AMNE-CONTAINING rier through permanent covalent bonds.