Computational Drug Design Challenges and Opportunities VRB
Total Page:16
File Type:pdf, Size:1020Kb

Load more
Recommended publications
-

User Guide of Computational Facilities
Physical Chemistry Unit Departament de Quimica User Guide of Computational Facilities Beta Version System Manager: Marc Noguera i Julian January 20, 2009 Contents 1 Introduction 1 2 Computer resources 2 3 Some Linux tools 2 4 The Cluster 2 4.1 Frontend servers . 2 4.2 Filesystems . 3 4.3 Computational nodes . 3 4.4 User’s environment . 4 4.5 Disk space quota . 4 4.6 Data Backup . 4 4.7 Generation of SSH keys for automatic SSH login . 4 4.8 Submitting jobs to the cluster . 5 4.8.1 Typical queue system commands . 6 4.8.2 HomeMade scripts . 6 4.8.3 Create your own submit script . 6 4.8.4 Submitting Parallel Calculations . 7 5 Available Software 9 5.1 Chemisty software . 9 5.2 Development Software . 10 5.3 How to use the software . 11 5.4 Non-default environments . 11 6 Your Linux Workstation 11 6.1 Disk space on your workstation . 12 6.2 Workstation Data Backup . 12 6.3 Running virtual Windows XP . 13 6.3.1 The Windows XP Environment . 13 6.4 Submitting calculations to your dekstop . 13 6.5 Network dependent structure . 14 7 Your WindowsXP Workstation 14 7.1 Workstation Data Backup . 14 8 Frequently asked questions 14 1 Introduction This User Guide is aimed to the standard user of the computer facilities in the ”Unitat de Quimica Fisica” in the Universitat Autnoma de Barcelona. It is not assumed that you have a knowledge of UNIX/Linux. However, you are 1 encouraged to take a close look to the linux guides that will be pointed out. -

Open Data, Open Source, and Open Standards in Chemistry: the Blue Obelisk Five Years On" Journal of Cheminformatics Vol
Oral Roberts University Digital Showcase College of Science and Engineering Faculty College of Science and Engineering Research and Scholarship 10-14-2011 Open Data, Open Source, and Open Standards in Chemistry: The lueB Obelisk five years on Andrew Lang Noel M. O'Boyle Rajarshi Guha National Institutes of Health Egon Willighagen Maastricht University Samuel Adams See next page for additional authors Follow this and additional works at: http://digitalshowcase.oru.edu/cose_pub Part of the Chemistry Commons Recommended Citation Andrew Lang, Noel M O'Boyle, Rajarshi Guha, Egon Willighagen, et al.. "Open Data, Open Source, and Open Standards in Chemistry: The Blue Obelisk five years on" Journal of Cheminformatics Vol. 3 Iss. 37 (2011) Available at: http://works.bepress.com/andrew-sid-lang/ 19/ This Article is brought to you for free and open access by the College of Science and Engineering at Digital Showcase. It has been accepted for inclusion in College of Science and Engineering Faculty Research and Scholarship by an authorized administrator of Digital Showcase. For more information, please contact [email protected]. Authors Andrew Lang, Noel M. O'Boyle, Rajarshi Guha, Egon Willighagen, Samuel Adams, Jonathan Alvarsson, Jean- Claude Bradley, Igor Filippov, Robert M. Hanson, Marcus D. Hanwell, Geoffrey R. Hutchison, Craig A. James, Nina Jeliazkova, Karol M. Langner, David C. Lonie, Daniel M. Lowe, Jerome Pansanel, Dmitry Pavlov, Ola Spjuth, Christoph Steinbeck, Adam L. Tenderholt, Kevin J. Theisen, and Peter Murray-Rust This article is available at Digital Showcase: http://digitalshowcase.oru.edu/cose_pub/34 Oral Roberts University From the SelectedWorks of Andrew Lang October 14, 2011 Open Data, Open Source, and Open Standards in Chemistry: The Blue Obelisk five years on Andrew Lang Noel M O'Boyle Rajarshi Guha, National Institutes of Health Egon Willighagen, Maastricht University Samuel Adams, et al. -

Real-Time Pymol Visualization for Rosetta and Pyrosetta
Real-Time PyMOL Visualization for Rosetta and PyRosetta Evan H. Baugh1, Sergey Lyskov1, Brian D. Weitzner1, Jeffrey J. Gray1,2* 1 Department of Chemical and Biomolecular Engineering, The Johns Hopkins University, Baltimore, Maryland, United States of America, 2 Program in Molecular Biophysics, The Johns Hopkins University, Baltimore, Maryland, United States of America Abstract Computational structure prediction and design of proteins and protein-protein complexes have long been inaccessible to those not directly involved in the field. A key missing component has been the ability to visualize the progress of calculations to better understand them. Rosetta is one simulation suite that would benefit from a robust real-time visualization solution. Several tools exist for the sole purpose of visualizing biomolecules; one of the most popular tools, PyMOL (Schro¨dinger), is a powerful, highly extensible, user friendly, and attractive package. Integrating Rosetta and PyMOL directly has many technical and logistical obstacles inhibiting usage. To circumvent these issues, we developed a novel solution based on transmitting biomolecular structure and energy information via UDP sockets. Rosetta and PyMOL run as separate processes, thereby avoiding many technical obstacles while visualizing information on-demand in real-time. When Rosetta detects changes in the structure of a protein, new coordinates are sent over a UDP network socket to a PyMOL instance running a UDP socket listener. PyMOL then interprets and displays the molecule. This implementation also allows remote execution of Rosetta. When combined with PyRosetta, this visualization solution provides an interactive environment for protein structure prediction and design. Citation: Baugh EH, Lyskov S, Weitzner BD, Gray JJ (2011) Real-Time PyMOL Visualization for Rosetta and PyRosetta. -

A Study on Cheminformatics and Its Applications on Modern Drug Discovery
Available online at www.sciencedirect.com Procedia Engineering 38 ( 2012 ) 1264 – 1275 Internatio na l Conference on Modeling Optimisatio n and Computing (ICMOC 2012) A Study on Cheminformatics and its Applications on Modern Drug Discovery B.Firdaus Begama and Dr. J.Satheesh Kumarb aResearch Scholar, Bharathiar University, Coimbatore, India, [email protected] bAssistant Professor, Bharathiar University, Coimbatore, India, [email protected] Abstract Discovering drugs to a disease is still a challenging task for medical researchers due to the complex structures of biomolecules which are responsible for disease such as AIDS, Cancer, Autism, Alzimear etc. Design and development of new efficient anti-drugs for the disease without any side effects are becoming mandatory in the recent history of human life cycle due to changes in various factors which includes food habit, environmental and migration in human life style. Cheminformaticds deals with discovering drugs based in modern drug discovery techniques which in turn rectifies complex issues in traditional drug discovery system. Cheminformatics tools, helps medical chemist for better understanding of complex structures of chemical compounds. Cheminformatics is a new emerging interdisciplinary field which primarily aims to discover Novel Chemical Entities [NCE] which ultimately results in design of new molecule [chemical data]. It also plays an important role for collecting, storing and analysing the chemical data. This paper focuses on cheminformatics and its applications on drug discovery and modern drug discovery techniques which helps chemist and medical researchers for finding solution to the complex disease. © 2012 Published by Elsevier Ltd. Selection and/or peer-review under responsibility of Noorul Islam Centre for Higher Education. -

Molecular Structure Input on the Web Peter Ertl
Ertl Journal of Cheminformatics 2010, 2:1 http://www.jcheminf.com/content/2/1/1 REVIEW Open Access Molecular structure input on the web Peter Ertl Abstract A molecule editor, that is program for input and editing of molecules, is an indispensable part of every cheminfor- matics or molecular processing system. This review focuses on a special type of molecule editors, namely those that are used for molecule structure input on the web. Scientific computing is now moving more and more in the direction of web services and cloud computing, with servers scattered all around the Internet. Thus a web browser has become the universal scientific user interface, and a tool to edit molecules directly within the web browser is essential. The review covers a history of web-based structure input, starting with simple text entry boxes and early molecule editors based on clickable maps, before moving to the current situation dominated by Java applets. One typical example - the popular JME Molecule Editor - will be described in more detail. Modern Ajax server-side molecule editors are also presented. And finally, the possible future direction of web-based molecule editing, based on tech- nologies like JavaScript and Flash, is discussed. Introduction this trend and input of molecular structures directly A program for the input and editing of molecules is an within a web browser is therefore of utmost importance. indispensable part of every cheminformatics or molecu- In this overview a history of entering molecules into lar processing system. Such a program is known as a web applications will be covered, starting from simple molecule editor, molecular editor or structure sketcher. -

A Nano-Visualization Software for Education and Research
A Nano-Visualization Software for Education and Research Lillian C. Oetting Department of Computer Science, Stanford University, Stanford, CA 94305 USA West High School, Iowa City, IA 52246 USA Tehseen Z. Raza Department of Physics and Astronomy, University of Iowa, Iowa City, IA 52242 USA Hassan Raza Department of Electrical and Computer Engineering, University of Iowa, Iowa City, IA 52242 USA Centre for Fundamental Research, Islamabad, Pakistan Abstract: We report the development of a user-friendly nano-visualization software program which can acquaint high-school students with nanotechnology. The visual introduction to atoms and molecules, which are the building blocks of this technology, is an effective way to introduce the key concepts in this area. The software’s graphical user interface enables multidimensional atomic visualization by using ball and stick schematics. Additionally, the software provides the option of wavefunction visualization for arbitrary nanomaterials and nanostructures by using extended Hückel theory. The software is instructive, application oriented and may be useful not only in high school education but also for the undergraduate research and teaching. 1 I. Introduction: The ability to accurately depict atomic, molecular and electronic structures has been a key factor in the advancement of nanotechnology. In this context, it is imperative to provide teaching and research platforms to motivate students towards this novel technology [1-3], while keeping the societal implications in perspective [4]. Nano-visualization appeals well due to its simplistic, yet elegant approach towards the visual representation of detailed concepts about quantum mechanics, quantum chemistry and linear algebra. Additionally, the conflux of quantum mechanics, numerical computation, graphical design, and computer programming gives exposure to the multi-disciplinary aspect of this technology [5-8]. -

Mannhold Methods and Principles in Medicinal Chemistry
Molecular Drug Properties Edited by Raimund Mannhold Methods and Principles in Medicinal Chemistry Edited by R. Mannhold, H. Kubinyi, G. Folkers Editorial Board H. Timmerman, J. Vacca, H. van de Waterbeemd, T. Wieland Previous Volumes of this Series: G. Cruciani (ed.) T. Langer, R. D. Hofmann (eds.) Molecular Interaction Fields Pharmacophores and Vol. 27 Pharmacophore Searches 2006, ISBN 978-3-527-31087-6 Vol. 32 2006, ISBN 978-3-527-31250-4 M. Hamacher, K. Marcus, K. Stühler, A. van Hall, B. Warscheid, H. E. Meyer (eds.) E. Francotte, W. Lindner (eds.) Proteomics in Drug Research Chirality in Drug Research Vol. 28 Vol. 33 2006, ISBN 978-3-527-31226-9 2006, ISBN 978-3-527-31076-0 D. J. Triggle, M. Gopalakrishnan, W. Jahnke, D. A. Erlanson (eds.) D. Rampe, W. Zheng (eds.) Fragment-based Approaches Voltage-Gated Ion Channels in Drug Discovery as Drug Targets Vol. 34 Vol. 29 2006, ISBN 978-3-527-31291-7 2006, ISBN 978-3-527-31258-0 D. Rognan (ed.) J. Hüser (ed.) Ligand Design for G High-Throughput Screening Protein-coupled Receptors in Drug Discovery Vol. 30 Vol. 35 2006, ISBN 978-3-527-31284-9 2006, ISBN 978-3-527-31283-2 D. A. Smith, H. van de Waterbeemd, K. Wanner, G. Höfner (eds.) D. K. Walker Mass Spectrometry in Pharmacokinetics and Medicinal Chemistry Metabolism in Drug Design, Vol. 36 2nd Ed. 2007, ISBN 978-3-527-31456-0 Vol. 31 2006, ISBN 978-3-527-31368-6 Molecular Drug Properties Measurement and Prediction Edited by Raimund Mannhold Series Editors All books published by Wiley-VCH are carefully produced. -

Python Tools in Computational Chemistry (And Biology)
Python Tools in Computational Chemistry (and Biology) Andrew Dalke Dalke Scientific, AB Göteborg, Sweden EuroSciPy, 26-27 July, 2008 “Why does ‘import numpy’ take 0.4 seconds? Does it need to import 228 libraries?” - My first Numpy-discussion post (paraphrased) Your use case isn't so typical and so suffers on the import time end of the balance. - Response from Robert Kern (Others did complain. Import time down to 0.28s.) 52,000 structures PDB doubles every 2½ years HEADER PHOTORECEPTOR 23-MAY-90 1BRD 1BRD 2 COMPND BACTERIORHODOPSIN 1BRD 3 SOURCE (HALOBACTERIUM $HALOBIUM) 1BRD 4 EXPDTA ELECTRON DIFFRACTION 1BRD 5 AUTHOR R.HENDERSON,J.M.BALDWIN,T.A.CESKA,F.ZEMLIN,E.BECKMANN, 1BRD 6 AUTHOR 2 K.H.DOWNING 1BRD 7 REVDAT 3 15-JAN-93 1BRDB 1 SEQRES 1BRDB 1 REVDAT 2 15-JUL-91 1BRDA 1 REMARK 1BRDA 1 .. ATOM 54 N PRO 8 20.397 -15.569 -13.739 1.00 20.00 1BRD 136 ATOM 55 CA PRO 8 21.592 -15.444 -12.900 1.00 20.00 1BRD 137 ATOM 56 C PRO 8 21.359 -15.206 -11.424 1.00 20.00 1BRD 138 ATOM 57 O PRO 8 21.904 -15.930 -10.563 1.00 20.00 1BRD 139 ATOM 58 CB PRO 8 22.367 -14.319 -13.591 1.00 20.00 1BRD 140 ATOM 59 CG PRO 8 22.089 -14.564 -15.053 1.00 20.00 1BRD 141 ATOM 60 CD PRO 8 20.647 -15.054 -15.103 1.00 20.00 1BRD 142 ATOM 61 N GLU 9 20.562 -14.211 -11.095 1.00 20.00 1BRD 143 ATOM 62 CA GLU 9 20.192 -13.808 -9.737 1.00 20.00 1BRD 144 ATOM 63 C GLU 9 19.567 -14.935 -8.932 1.00 20.00 1BRD 145 ATOM 64 O GLU 9 19.815 -15.104 -7.724 1.00 20.00 1BRD 146 ATOM 65 CB GLU 9 19.248 -12.591 -9.820 1.00 99.00 1 1BRD 147 ATOM 66 CG GLU 9 19.902 -11.351 -10.387 1.00 99.00 1 1BRD 148 ATOM 67 CD GLU 9 19.243 -10.169 -10.980 1.00 99.00 1 1BRD 149 ATOM 68 OE1 GLU 9 18.323 -10.191 -11.782 1.00 99.00 1 1BRD 150 ATOM 69 OE2 GLU 9 19.760 -9.089 -10.597 1.00 99.00 1 1BRD 151 ATOM 70 N TRP 10 18.764 -15.737 -9.597 1.00 20.00 1BRD 152 ATOM 71 CA TRP 10 18.034 -16.884 -9.090 1.00 20.00 1BRD 153 ATOM 72 C TRP 10 18.843 -17.908 -8.318 1.00 20.00 1BRD 154 ATOM 73 O TRP 10 18.376 -18.310 -7.230 1.00 20.00 1BRD 155 . -

Chemdoodle Web Components: HTML5 Toolkit for Chemical Graphics, Interfaces, and Informatics Melanie C Burger1,2*
Burger. J Cheminform (2015) 7:35 DOI 10.1186/s13321-015-0085-3 REVIEW Open Access ChemDoodle Web Components: HTML5 toolkit for chemical graphics, interfaces, and informatics Melanie C Burger1,2* Abstract ChemDoodle Web Components (abbreviated CWC, iChemLabs, LLC) is a light-weight (~340 KB) JavaScript/HTML5 toolkit for chemical graphics, structure editing, interfaces, and informatics based on the proprietary ChemDoodle desktop software. The library uses <canvas> and WebGL technologies and other HTML5 features to provide solutions for creating chemistry-related applications for the web on desktop and mobile platforms. CWC can serve a broad range of scientific disciplines including crystallography, materials science, organic and inorganic chemistry, biochem- istry and chemical biology. CWC is freely available for in-house use and is open source (GPL v3) for all other uses. Keywords: ChemDoodle Web Components, Chemical graphics, Animations, Cheminformatics, HTML5, Canvas, JavaScript, WebGL, Structure editor, Structure query Introduction Mobile browsers did support HTML5, which opened How we communicate chemical information is increas- the door to web applications built with only HTML, ingly technology driven. Learning management systems, CSS and JavaScript (JS), such as the ChemDoodle Web virtual classrooms and MOOCs are a few examples where Components. chemistry educators need forward compatible tools for digital natives. Companies that implement emerg- Review ing web technologies can find efficiencies and benefit The ChemDoodle Web Components technology stack from competitive advantages. The first chemical graph- and features ics toolkit for the web, MDL Chime, was introduced in The ChemDoodle Web Components library, released in 1996 [1]. Based on the molecular visualization program 2009, is the first chemistry toolkit for structure viewing RasMol, Chime was developed as a plugin for Netscape and editing that is originally built using only web stand- and later for Internet Explorer and Firefox. -

64-194 Projekt Parallelrechnerevaluation Abschlussbericht
64-194 Projekt Parallelrechnerevaluation Abschlussbericht Automatisierte Build-Tests für Spack Sarah Lubitz Malte Fock [email protected] [email protected] Studiengang: LA Berufliche Schulen – Informatik Studiengang LaGym - Informatik Matr.-Nr. 6570465 Matr.-Nr. 6311966 Inhaltsverzeichnis i Inhaltsverzeichnis 1 Einleitung1 1.1 Motivation.....................................1 1.2 Vorbereitungen..................................2 1.2.1 Spack....................................2 1.2.2 Python...................................2 1.2.3 Unix-Shell.................................3 1.2.4 Slurm....................................3 1.2.5 GitHub...................................4 2 Code 7 2.1 Automatisiertes Abrufen und Installieren von Spack-Paketen.......7 2.1.1 Python Skript: install_all_packets.py..................7 2.2 Batch-Jobskripte..................................9 2.2.1 Test mit drei Paketen........................... 10 2.3 Analyseskript................................... 11 2.3.1 Test mit 500 Paketen........................... 12 2.3.2 Code zur Analyse der Error-Logs, forts................. 13 2.4 Hintereinanderausführung der Installationen................. 17 2.4.1 Wrapper Skript.............................. 19 2.5 E-Mail Benachrichtigungen........................... 22 3 Testlauf 23 3.1 Durchführung und Ergebnisse......................... 23 3.2 Auswertung und Schlussfolgerungen..................... 23 3.3 Fazit und Ausblick................................ 24 4 Schulische Relevanz 27 Literaturverzeichnis -
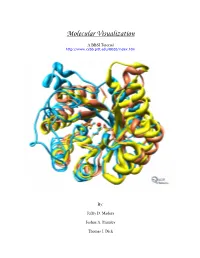
Instructions for PDB Downloading (From Either Website)
Molecular Visualization A BBSI Tutorial http://www.ccbb.pitt.edu/BBSI/index.htm By: Jeffry D. Madura Joshua A. Plumley Thomas J. Dick Table of Contents Instructions for PDB downloading..........................................................2 RasMol Tutorial ...........................................................................................3 VMD Tutorial ...............................................................................................5 CAChe Tutuorial..........................................................................................8 MOE Tutorial ............................................................................................. 10 Chimera Tutorial ....................................................................................... 13 Exercises .................................................................................................... 16 1 Instructions for PDB downloading (from either website) -Go to: - Go to: www.rcsb.org/pdb/ www.pdb.bu.edu/oca-bin/pdblite -Type in name of protein (examples at - Type in name of Protein / bottom of the page). Macromolecule ( examples at bottom of page). - Click on Name icon (first name in purple box). - Click on Retrieve Released Data Matching Your Query icon. -On the left side of the screen, click on Download/Display Structure - Highlight any of the molecule name and click on the View/ Analyze/ -Under Download the Structure File, Save Macro Molecule icon. right click on the X where the PDB(top) meets with none, under - Under the Data Retrieval section, -

How to Print a Crystal Structure Model in 3D Teng-Hao Chen,*A Semin Lee, B Amar H
Electronic Supplementary Material (ESI) for CrystEngComm. This journal is © The Royal Society of Chemistry 2014 How to Print a Crystal Structure Model in 3D Teng-Hao Chen,*a Semin Lee, b Amar H. Flood b and Ognjen Š. Miljanić a a Department of Chemistry, University of Houston, 112 Fleming Building, Houston, Texas 77204-5003, United States b Department of Chemistry, Indiana University, 800 E. Kirkwood Avenue, Bloomington, Indiana 47405, United States Supporting Information Instructions for Printing a 3D Model of an Interlocked Molecule: Cyanostars .................................... S2 Instructions for Printing a "Molecular Spinning Top" in 3D: Triazolophanes ..................................... S3 References ....................................................................................................................................................................... S4 S1 Instructions for Printing a 3D Model of an Interlocked Molecule: Cyanostars Mechanically interlocked molecules such as rotaxanes and catenanes can also be easily printed in 3D. As long as the molecule is correctly designed and modeled, there is no need to assemble and glue the components after printing—they are printed inherently as mechanically interlocked molecules. We present the preparation of a cyanostar- based [3]rotaxane as an example. A pair of sandwiched cyanostars S1 (CS ) derived from the crystal structure was "cleaned up" in the molecular computation software Spartan ʹ10, i.e. disordered atoms were removed. A linear molecule was created and threaded through the cavity of two cyanostars (Figure S1a). Using a space filling view of the molecule, the three components were spaced sufficiently far apart to ensure that they did not make direct contact Figure S1 . ( a) Side and top view of two cyanostars ( CS ) with each other when they are printed. threaded with a linear molecule. ( b) Side and top view of a Bulky stoppers were added to each [3]rotaxane.