Early Evolution of the Vascular Plant Body Plan — the Missing Mechanisms
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
History Department Botany
THE HISTORY OF THE DEPARTMENT OF BOTANY 1889-1989 UNIVERSITY OF MINNESOTA SHERI L. BARTLETT I - ._-------------------- THE HISTORY OF THE DEPARTMENT OF BOTANY 1889-1989 UNIVERSITY OF MINNESOTA SHERI L. BARTLETT TABLE OF CONTENTS Preface 1-11 Chapter One: 1889-1916 1-18 Chapter Two: 1917-1935 19-38 Chapter Three: 1936-1954 39-58 Chapter Four: 1955-1973 59-75 Epilogue 76-82 Appendix 83-92 Bibliography 93-94 -------------------------------------- Preface (formerly the College of Science, Literature and the Arts), the College of Agriculture, or The history that follows is the result some other area. Eventually these questions of months ofresearch into the lives and work were resolved in 1965 when the Department of the Botany Department's faculty members joined the newly established College of and administrators. The one-hundred year Biological Sciences (CBS). In 1988, The overview focuses on the Department as a Department of Botany was renamed the whole, and the decisions that Department Department of Plant Biology, and Irwin leaders made to move the field of botany at Rubenstein from the Department of Genetics the University of Minnesota forward in a and Cell Biology became Plant Biology's dynamic and purposeful manner. However, new head. The Department now has this is not an effort to prove that the administrative ties to both the College of Department's history was linear, moving Biological Sciences and the College of forward in a pre-determined, organized Agriculture. fashion at every moment. Rather I have I have tried to recognize the attempted to demonstrate the complexities of accomplishments and individuality of the the personalities and situations that shaped Botany Department's faculty while striving to the growth ofthe Department and made it the describe the Department as one entity. -

Gymnosperms the MESOZOIC: ERA of GYMNOSPERM DOMINANCE
Chapter 24 Gymnosperms THE MESOZOIC: ERA OF GYMNOSPERM DOMINANCE THE VASCULAR SYSTEM OF GYMNOSPERMS CYCADS GINKGO CONIFERS Pinaceae Include the Pines, Firs, and Spruces Cupressaceae Include the Junipers, Cypresses, and Redwoods Taxaceae Include the Yews, but Plum Yews Belong to Cephalotaxaceae Podocarpaceae and Araucariaceae Are Largely Southern Hemisphere Conifers THE LIFE CYCLE OF PINUS, A REPRESENTATIVE GYMNOSPERM Pollen and Ovules Are Produced in Different Kinds of Structures Pollination Replaces the Need for Free Water Fertilization Leads to Seed Formation GNETOPHYTES GYMNOSPERMS: SEEDS, POLLEN, AND WOOD THE ECOLOGICAL AND ECONOMIC IMPORTANCE OF GYMNOSPERMS The Origin of Seeds, Pollen, and Wood Seeds and Pollen Are Key Reproductive SUMMARY Innovations for Life on Land Seed Plants Have Distinctive Vegetative PLANTS, PEOPLE, AND THE Features ENVIRONMENT: The California Coast Relationships among Gymnosperms Redwood Forest 1 KEY CONCEPTS 1. The evolution of seeds, pollen, and wood freed plants from the need for water during reproduction, allowed for more effective dispersal of sperm, increased parental investment in the next generation and allowed for greater size and strength. 2. Seed plants originated in the Devonian period from a group called the progymnosperms, which possessed wood and heterospory, but reproduced by releasing spores. Currently, five lineages of seed plants survive--the flowering plants plus four groups of gymnosperms: cycads, Ginkgo, conifers, and gnetophytes. Conifers are the best known and most economically important group, including pines, firs, spruces, hemlocks, redwoods, cedars, cypress, yews, and several Southern Hemisphere genera. 3. The pine life cycle is heterosporous. Pollen strobili are small and seasonal. Each sporophyll has two microsporangia, in which microspores are formed and divide into immature male gametophytes while still retained in the microsporangia. -

"National List of Vascular Plant Species That Occur in Wetlands: 1996 National Summary."
Intro 1996 National List of Vascular Plant Species That Occur in Wetlands The Fish and Wildlife Service has prepared a National List of Vascular Plant Species That Occur in Wetlands: 1996 National Summary (1996 National List). The 1996 National List is a draft revision of the National List of Plant Species That Occur in Wetlands: 1988 National Summary (Reed 1988) (1988 National List). The 1996 National List is provided to encourage additional public review and comments on the draft regional wetland indicator assignments. The 1996 National List reflects a significant amount of new information that has become available since 1988 on the wetland affinity of vascular plants. This new information has resulted from the extensive use of the 1988 National List in the field by individuals involved in wetland and other resource inventories, wetland identification and delineation, and wetland research. Interim Regional Interagency Review Panel (Regional Panel) changes in indicator status as well as additions and deletions to the 1988 National List were documented in Regional supplements. The National List was originally developed as an appendix to the Classification of Wetlands and Deepwater Habitats of the United States (Cowardin et al.1979) to aid in the consistent application of this classification system for wetlands in the field.. The 1996 National List also was developed to aid in determining the presence of hydrophytic vegetation in the Clean Water Act Section 404 wetland regulatory program and in the implementation of the swampbuster provisions of the Food Security Act. While not required by law or regulation, the Fish and Wildlife Service is making the 1996 National List available for review and comment. -

STEM Disciplines
STEM Disciplines In order to be applicable to the many types of institutions that participate in the HERI Faculty Survey, this list is intentionally broad and comprehensive in its definition of STEM disciplines. It includes disciplines in the life sciences, physical sciences, engineering, mathematics, computer science, and the health sciences. Agriculture/Natural Resources Health Professions 0101 Agriculture and related sciences 1501 Alternative/complementary medicine/sys 0102 Natural resources and conservation 1503 Clinical/medical lab science/allied 0103 Agriculture/natural resources/related, other 1504 Dental support services/allied 1505 Dentistry Biological and Biomedical Sciences 1506 Health & medical administrative services 0501 Biochem/biophysics/molecular biology 1507 Allied health and medical assisting services 0502 Botany/plant biology 1508 Allied health diagnostic, intervention, 0503 Genetics treatment professions 0504 Microbiological sciences & immunology 1509 Medicine, including psychiatry 0505 Physiology, pathology & related sciences 1511 Nursing 0506 Zoology/animal biology 1512 Optometry 0507 Biological & biomedical sciences, other 1513 Osteopathic medicine/osteopathy 1514 Pharmacy/pharmaceutical sciences/admin Computer/Info Sciences/Support Tech 1515 Podiatric medicine/podiatry 0801 Computer/info tech administration/mgmt 1516 Public health 0802 Computer programming 1518 Veterinary medicine 0803 Computer science 1519 Health/related clinical services, other 0804 Computer software and media applications 0805 Computer systems -

Anthocerotophyta
Glime, J. M. 2017. Anthocerotophyta. Chapt. 2-8. In: Glime, J. M. Bryophyte Ecology. Volume 1. Physiological Ecology. Ebook 2-8-1 sponsored by Michigan Technological University and the International Association of Bryologists. Last updated 5 June 2020 and available at <http://digitalcommons.mtu.edu/bryophyte-ecology/>. CHAPTER 2-8 ANTHOCEROTOPHYTA TABLE OF CONTENTS Anthocerotophyta ......................................................................................................................................... 2-8-2 Summary .................................................................................................................................................... 2-8-10 Acknowledgments ...................................................................................................................................... 2-8-10 Literature Cited .......................................................................................................................................... 2-8-10 2-8-2 Chapter 2-8: Anthocerotophyta CHAPTER 2-8 ANTHOCEROTOPHYTA Figure 1. Notothylas orbicularis thallus with involucres. Photo by Michael Lüth, with permission. Anthocerotophyta These plants, once placed among the bryophytes in the families. The second class is Leiosporocerotopsida, a Anthocerotae, now generally placed in the phylum class with one order, one family, and one genus. The genus Anthocerotophyta (hornworts, Figure 1), seem more Leiosporoceros differs from members of the class distantly related, and genetic evidence may even present -

The Analysis of Pinus Pinaster Snrks Reveals Clues of the Evolution of This Family and a New Set of Abiotic Stress Resistance Biomarkers
agronomy Article The Analysis of Pinus pinaster SnRKs Reveals Clues of the Evolution of This Family and a New Set of Abiotic Stress Resistance Biomarkers Francisco Javier Colina * , María Carbó , Ana Álvarez , Luis Valledor * and María Jesús Cañal Plant Physiology, Department of Organisms and Systems Biology and University Institute of Biotechnology (IUBA), University of Oviedo, 33006 Oviedo, Spain; [email protected] (M.C.); [email protected] (A.Á.); [email protected] (M.J.C.) * Correspondence: [email protected] (F.J.C.); [email protected] (L.V.) Received: 15 January 2020; Accepted: 17 February 2020; Published: 19 February 2020 Abstract: Climate change is increasing the intensity and incidence of environmental stressors, reducing the biomass yields of forestry species as Pinus pinaster. Selection of new stress-tolerant varieties is thus required. Many genes related to plant stress signaling pathways have proven useful for this purpose with sucrose non-fermenting related kinases (SnRK), conserved across plant evolution and connected to different phosphorylation cascades within ABA- and Ca2+-mediated signaling pathways, as a good example. The modulation of SnRKs and/or the selection of specific SnRK alleles have proven successful strategies to increase plant stress resistance. Despite this, SnRKs have been barely studied in gymnosperms. In this work P. pinaster SnRK sequences (PpiSnRK) were identified through a homology- and domain-based sequence analysis using Arabidopsis SnRK sequences as query. Moreover, PpiSnRKs links to the gymnosperm stress response were modeled out of the known interactions of PpiSnRKs orthologs from other species with different signaling complexity. This approach successfully identified the pine SnRK family and predicted their central role into the gymnosperm stress response, linking them to ABA, Ca2+, sugar/energy and possibly ethylene signaling. -

The Big Bloom—How Flowering Plants Changed the World
The Big Bloom—How Flowering Plants Changed the World Written by Michael Klesius Republished from the pages of National Geographic magazine -- July 2002 In the summer of 1973 sunflowers appeared in my father's vegetable garden. They seemed to sprout overnight in a few rows he had lent that year to new neighbors from California. Only six years old at the time, I was at first put off by these garish plants. Such strange and vibrant flowers seemed out of place among the respectable beans, peppers, spinach, and other vegetables we had always grown. Gradually, however, the brilliance of the sunflowers won me over. Their fiery halos relieved the green monotone that by late summer ruled the garden. I marveled at birds that clung upside down to the shaggy, gold disks, wings fluttering, looting the seeds. Sunflowers defined flowers for me that summer and changed my view of the world. Flowers have a way of doing that. They began changing the way the world looked almost as soon as they appeared on Earth about 130 million years ago, during the Cretaceous period. That's relatively recent in geologic time: If all Earth's history were compressed into an hour, flowering plants would exist for only the last 90 seconds. But once they took firm root about 100 million years ago, they swiftly diversified in an explosion of varieties that established most of the flowering plant families of the modern world. Today flowering plant species outnumber by twenty to one those of ferns and cone-bearing trees, or conifers, which had thrived for 200 million years before the first bloom appeared. -

Ordovician Land Plants and Fungi from Douglas Dam, Tennessee
PROOF The Palaeobotanist 68(2019): 1–33 The Palaeobotanist 68(2019): xxx–xxx 0031–0174/2019 0031–0174/2019 Ordovician land plants and fungi from Douglas Dam, Tennessee GREGORY J. RETALLACK Department of Earth Sciences, University of Oregon, Eugene, OR 97403, USA. *Email: gregr@uoregon. edu (Received 09 September, 2019; revised version accepted 15 December, 2019) ABSTRACT The Palaeobotanist 68(1–2): Retallack GJ 2019. Ordovician land plants and fungi from Douglas Dam, Tennessee. The Palaeobotanist 68(1–2): xxx–xxx. 1–33. Ordovician land plants have long been suspected from indirect evidence of fossil spores, plant fragments, carbon isotopic studies, and paleosols, but now can be visualized from plant compressions in a Middle Ordovician (Darriwilian or 460 Ma) sinkhole at Douglas Dam, Tennessee, U. S. A. Five bryophyte clades and two fungal clades are represented: hornwort (Casterlorum crispum, new form genus and species), liverwort (Cestites mirabilis Caster & Brooks), balloonwort (Janegraya sibylla, new form genus and species), peat moss (Dollyphyton boucotii, new form genus and species), harsh moss (Edwardsiphyton ovatum, new form genus and species), endomycorrhiza (Palaeoglomus strotheri, new species) and lichen (Prototaxites honeggeri, new species). The Douglas Dam Lagerstätte is a benchmark assemblage of early plants and fungi on land. Ordovician plant diversity now supports the idea that life on land had increased terrestrial weathering to induce the Great Ordovician Biodiversification Event in the sea and latest Ordovician (Hirnantian) -

Prescribed Fire Decreases Lichen and Bryophyte Biomass and Alters Functional Group Composition in Pacific Northwest Prairies Author(S): Lalita M
Prescribed Fire Decreases Lichen and Bryophyte Biomass and Alters Functional Group Composition in Pacific Northwest Prairies Author(s): Lalita M. Calabria, Kate Petersen, Sarah T. Hamman and Robert J. Smith Source: Northwest Science, 90(4):470-483. Published By: Northwest Scientific Association DOI: http://dx.doi.org/10.3955/046.090.0407 URL: http://www.bioone.org/doi/full/10.3955/046.090.0407 BioOne (www.bioone.org) is a nonprofit, online aggregation of core research in the biological, ecological, and environmental sciences. BioOne provides a sustainable online platform for over 170 journals and books published by nonprofit societies, associations, museums, institutions, and presses. Your use of this PDF, the BioOne Web site, and all posted and associated content indicates your acceptance of BioOne’s Terms of Use, available at www.bioone.org/page/terms_of_use. Usage of BioOne content is strictly limited to personal, educational, and non-commercial use. Commercial inquiries or rights and permissions requests should be directed to the individual publisher as copyright holder. BioOne sees sustainable scholarly publishing as an inherently collaborative enterprise connecting authors, nonprofit publishers, academic institutions, research libraries, and research funders in the common goal of maximizing access to critical research. Lalita M. Calabria1, Kate Petersen,The Evergreen State College, 2700 Evergreen Parkway NW, Olympia, Washington 98505 Sarah T. Hamman, The Center for Natural Lands Management, 120 Union Ave SE #215, Olympia, Washington 98501 and Robert J. Smith, Department of Botany and Plant Pathology, 2082 Cordley Hall, Oregon State University, Corvallis, Oregon 97331 Prescribed Fire Decreases Lichen and Bryophyte Biomass and Alters Functional Group Composition in Pacific Northwest Prairies Abstract The reintroduction of fire to Pacific Northwest prairies has been useful for removing non-native shrubs and supporting habitat for fire-adapted plant and animal species. -
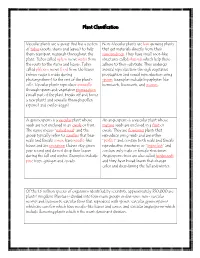
Plant Classification
Plant Classification Vascular plants are a group that has a system Non-Vascular plants are low growing plants of tubes (roots, stems and leaves) to help that get materials directly from their them transport materials throughout the surroundings. They have small root-like plant. Tubes called xylem move water from structures called rhizoids which help them the roots to the stems and leaves. Tubes adhere to their substrate. They undergo called phloem move food from the leaves asexual reproduction through vegetative (where sugar is made during propagation and sexual reproduction using photosynthesis) to the rest of the plant’s spores. Examples include bryophytes like cells. Vascular plants reproduce asexually hornworts, liverworts, and mosses. through spores and vegetative propagation (small part of the plant breaks off and forms a new plant) and sexually through pollen (sperm) and ovules (eggs). A gymnosperm is a vascular plant whose An angiosperm is a vascular plant whose seeds are not enclosed in an ovule or fruit. mature seeds are enclosed in a fruit or The name means “naked seed” and the ovule. They are flowering plants that group typically refers to conifers that bear reproduce using seeds and are either male and female cones, have needle-like “perfect” and contain both male and female leaves and are evergreen (leaves stay green reproductive structures or “imperfect” and year round and do not drop their leaves contain only male or female structures. during the fall and winter. Examples include Angiosperm trees are also called hardwoods pine trees, ginkgos and cycads. and they have broad leaves that change color and drop during the fall and winter. -

Bryophyte Life Cycle
MARCH 2016LEARNING | VELD & FLORA ABOUT BIODIVERSITY Veld & Flora MARCHFACTSHEET: 2016 | VELD MOSS & FLORA 24 25 3. BRYOPHYTE LIFE CYCLE Gametophyte (n) UNDERSTANDING THE GERMINATION MALE ALTERNATION OF GENERATIONS The way that almost all land plants reproduce is by means of two distinct, alternating life forms, a sexual phase that produces and releases gametes or sex cells and allows fertilisation, and a dispersal phase – both of which are adaptations to an essentially waterless environment. The sexual phase is known as the GAMETOPHYTE or haploid spores (n) generation and the dispersal phase is the SPOROPHYTE or diploid (2n) generation. Mature gametophyte plants produce haploid sex cells (egg and sperm) in sex organs (the male antheridia and female archegonia). These sex cells (also called gametes) fuse during fertilisation to form a diploid (2n) zygote which COPING OUT OF WATER grows, by means of mitosis (that results in two daughter cells each having Sperm (n) the same number and kind of chromosomes as the parent cell), into a new released Bryophytes, which include moss, are primitive plants that give us some idea sporophyte plant. The diploid sporophyte produces haploid (n) spores (i.e. from male of how the first plants that ventured onto land coped with their new waterless FEMALE each spore has a single set of chromosomes) by means of the process environment. They share many features with other plants, but differ in some of cell division called meiosis. Meiosis results in four daughter cells each ways – such as the lack of an effective vascular system (specialised tissue for with half the number of chromosomes of the parent cell. -

The Timescale of Early Land Plant Evolution PNAS PLUS
The timescale of early land plant evolution PNAS PLUS Jennifer L. Morrisa,1, Mark N. Putticka,b,1, James W. Clarka, Dianne Edwardsc, Paul Kenrickb, Silvia Presseld, Charles H. Wellmane, Ziheng Yangf,g, Harald Schneidera,d,h,2, and Philip C. J. Donoghuea,2 aSchool of Earth Sciences, University of Bristol, Bristol BS8 1TQ, United Kingdom; bDepartment of Earth Sciences, Natural History Museum, London SW7 5BD, United Kingdom; cSchool of Earth and Ocean Sciences, Cardiff University, Cardiff CF10, United Kingdom; dDepartment of Life Sciences, Natural History Museum, London SW7 5BD, United Kingdom; eDepartment of Animal and Plant Sciences, University of Sheffield, Sheffield S10 2TN, United Kingdom; fDepartment of Genetics, Evolution and Environment, University College London, London WC1E 6BT, United Kingdom; gRadclie Institute for Advanced Studies, Harvard University, Cambridge, MA 02138; and hCenter of Integrative Conservation, Xishuangbanna Tropical Botanical Garden, Chinese Academy of Sciences, Yunnan 666303, China Edited by Peter R. Crane, Oak Spring Garden Foundation, Upperville, VA, and approved January 17, 2018 (received for review November 10, 2017) Establishing the timescale of early land plant evolution is essential recourse but to molecular clock methodology, employing the for testing hypotheses on the coevolution of land plants and known fossil record to calibrate and constrain molecular evolu- Earth’s System. The sparseness of early land plant megafossils and tion to time. Unfortunately, the relationships among the four stratigraphic controls on their distribution make the fossil record principal lineages of land plants, namely, hornworts, liverworts, an unreliable guide, leaving only the molecular clock. However, mosses, and tracheophytes, are unresolved, with almost every the application of molecular clock methodology is challenged by possible solution currently considered viable (14).