Jérôme KIEFFER
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Supporting Information
Electronic Supplementary Material (ESI) for RSC Advances. This journal is © The Royal Society of Chemistry 2020 Supporting Information How to Select Ionic Liquids as Extracting Agent Systematically? Special Case Study for Extractive Denitrification Process Shurong Gaoa,b,c,*, Jiaxin Jina,b, Masroor Abroc, Ruozhen Songc, Miao Hed, Xiaochun Chenc,* a State Key Laboratory of Alternate Electrical Power System with Renewable Energy Sources, North China Electric Power University, Beijing, 102206, China b Research Center of Engineering Thermophysics, North China Electric Power University, Beijing, 102206, China c Beijing Key Laboratory of Membrane Science and Technology & College of Chemical Engineering, Beijing University of Chemical Technology, Beijing 100029, PR China d Office of Laboratory Safety Administration, Beijing University of Technology, Beijing 100124, China * Corresponding author, Tel./Fax: +86-10-6443-3570, E-mail: [email protected], [email protected] 1 COSMO-RS Computation COSMOtherm allows for simple and efficient processing of large numbers of compounds, i.e., a database of molecular COSMO files; e.g. the COSMObase database. COSMObase is a database of molecular COSMO files available from COSMOlogic GmbH & Co KG. Currently COSMObase consists of over 2000 compounds including a large number of industrial solvents plus a wide variety of common organic compounds. All compounds in COSMObase are indexed by their Chemical Abstracts / Registry Number (CAS/RN), by a trivial name and additionally by their sum formula and molecular weight, allowing a simple identification of the compounds. We obtained the anions and cations of different ILs and the molecular structure of typical N-compounds directly from the COSMObase database in this manuscript. -

Quantitative Evaluation of Dissociation Mechanisms in Phenolphthalein and the Related Compounds
J. Comput. Chem. Jpn., Vol. 15, No. 1, pp. 13–21 (2016) ©2016 Society of Computer Chemistry, Japan Technical Paper Quantitative Evaluation of Dissociation Mechanisms in Phenolphthalein and the Related Compounds Toshihiko HANAI Health Research Foundation, Research Institute for Production Development 4F, 15 Shimogamo-morimoto-cho, Sakyo-ku, Kyoto 606-0805, Japan e-mail: [email protected] (Received: October 11, 2015; Accepted for publication: April 14, 2016; Online publication: May 6, 2016) Computational chemistry programs were evaluated as aids to teaching qualitative analytical chemistry. Compu- tational chemical calculations can predict absorption spectra, thus enabling the modeling of indicator dissociation mechanisms by different computational chemical programs using a personal computer. An updated MNDO program among 51 programs was found to be the best predictor to explain the dissociation mechanisms of isobenzofuranones and sulfonephthaleins. Unknown dissociation constants were predicted from atomic partial charges instead of Ham- mett's constants. Keywords: Quantitative analysis of dissociation mechanisms, Isobenzofuranone, Sulfonephthalein, Computational chemistry 1 Introduction the absorption wavelength and electron density changes were not well described. The dissociation mechanisms, maximum How to quantitatively teach qualitative analytical chemistry is wavelengths, and electron density maps of isobenzofuranones a very important subject for analytical chemists. Previously, a and sulfonephthaleins were, therefore, evaluated by in silico method to teach molecular interaction mechanisms in chroma- analysis despite the anticipated poor precision. The experimen- tography was quantitatively achieved using molecular mechan- tally measured dissociation of phenolphthalein is described ics and MOPAC programs [1]. Furthermore, the reaction mech- using four dissociation structures, where the ionization of two anisms of highly sensitive detections were also quantitatively phenolic hydroxyl groups converts the neutral molecular form described [2,3]. -

Computer-Assisted Catalyst Development Via Automated Modelling of Conformationally Complex Molecules
www.nature.com/scientificreports OPEN Computer‑assisted catalyst development via automated modelling of conformationally complex molecules: application to diphosphinoamine ligands Sibo Lin1*, Jenna C. Fromer2, Yagnaseni Ghosh1, Brian Hanna1, Mohamed Elanany3 & Wei Xu4 Simulation of conformationally complicated molecules requires multiple levels of theory to obtain accurate thermodynamics, requiring signifcant researcher time to implement. We automate this workfow using all open‑source code (XTBDFT) and apply it toward a practical challenge: diphosphinoamine (PNP) ligands used for ethylene tetramerization catalysis may isomerize (with deleterious efects) to iminobisphosphines (PPNs), and a computational method to evaluate PNP ligand candidates would save signifcant experimental efort. We use XTBDFT to calculate the thermodynamic stability of a wide range of conformationally complex PNP ligands against isomeriation to PPN (ΔGPPN), and establish a strong correlation between ΔGPPN and catalyst performance. Finally, we apply our method to screen novel PNP candidates, saving signifcant time by ruling out candidates with non‑trivial synthetic routes and poor expected catalytic performance. Quantum mechanical methods with high energy accuracy, such as density functional theory (DFT), can opti- mize molecular input structures to a nearby local minimum, but calculating accurate reaction thermodynamics requires fnding global minimum energy structures1,2. For simple molecules, expert intuition can identify a few minima to focus study on, but an alternative approach must be considered for more complex molecules or to eventually fulfl the dream of autonomous catalyst design 3,4: the potential energy surface must be frst surveyed with a computationally efcient method; then minima from this survey must be refned using slower, more accurate methods; fnally, for molecules possessing low-frequency vibrational modes, those modes need to be treated appropriately to obtain accurate thermodynamic energies 5–7. -
Biovia Materials Studio Visualizer Datasheet
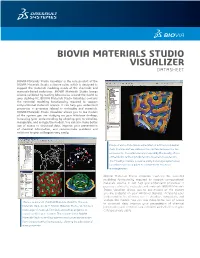
BIOVIA MATERIALS STUDIO VISUALIZER DATASHEET BIOVIA Materials Studio Visualizer is the core product of the BIOVIA Materials Studio software suite, which is designed to support the materials modeling needs of the chemicals and materials-based industries. BIOVIA Materials Studio brings science validated by leading laboratories around the world to your desktop PC. BIOVIA Materials Studio Visualizer contains the essential modeling functionality required to support computational materials science. It can help you understand properties or processes related to molecules and materials. BIOVIA Materials Studio Visualizer allows you to see models of the system you are studying on your Windows desktop, increasing your understanding by allowing you to visualize, manipulate, and analyze the models. You can also make better use of access to structural data, improve your presentation of chemical information, and communicate problems and solutions to your colleagues very easily. Image of early-stage phase segregation in a diblock copolymer melt. The blue surface indicates the interface between the two components. The volume is colormapped by the density of one of the blocks, red being high density, blue being low-density. The MesoDyn module is used to study these large systems over long-times such as required to observe these structural rearrangements. BIOVIA Materials Studio Visualizer contains the essential modeling functionality required to support computational materials science. It can help you understand properties or processes related to molecules and materials. BIOVIA Materials Studio Visualizer allows you to see models of the system you are studying on your Windows desktop, increasing your understanding by allowing you to visualize, manipulate, and analyze the models. -

Download Author Version (PDF)
PCCP Accepted Manuscript This is an Accepted Manuscript, which has been through the Royal Society of Chemistry peer review process and has been accepted for publication. Accepted Manuscripts are published online shortly after acceptance, before technical editing, formatting and proof reading. Using this free service, authors can make their results available to the community, in citable form, before we publish the edited article. We will replace this Accepted Manuscript with the edited and formatted Advance Article as soon as it is available. You can find more information about Accepted Manuscripts in the Information for Authors. Please note that technical editing may introduce minor changes to the text and/or graphics, which may alter content. The journal’s standard Terms & Conditions and the Ethical guidelines still apply. In no event shall the Royal Society of Chemistry be held responsible for any errors or omissions in this Accepted Manuscript or any consequences arising from the use of any information it contains. www.rsc.org/pccp Page 1 of 11 PhysicalPlease Chemistry do not adjust Chemical margins Physics PCCP PAPER Effect of nanosize on surface properties of NiO nanoparticles for adsorption of Quinolin-65 ab a a Received 00th January 20xx, Nedal N. Marei, Nashaat N. Nassar* and Gerardo Vitale Accepted 00th January 20xx Using Quinolin-65 (Q-65) as a model-adsorbing compound for polar heavy hydrocarbons, the nanosize effect of NiO Manuscript DOI: 10.1039/x0xx00000x nanoparticles on adsorption of Q-65 was investigated. Different-sized NiO nanoparticles with sizes between 5 and 80 nm were prepared by controlled thermal dehydroxylation of Ni(OH)2. -

A Comparative Study of the Efficiency of HCV NS3/4A Protease Drugs
Life Sciences 217 (2019) 176–184 Contents lists available at ScienceDirect Life Sciences journal homepage: www.elsevier.com/locate/lifescie A comparative study of the efficiency of HCV NS3/4A protease drugs against different HCV genotypes using in silico approaches T ⁎ Ahmed A. Ezat , Wael M. Elshemey Biophysics Department, Faculty of Science, Cairo University, 12613 Giza, Egypt ARTICLE INFO ABSTRACT Keywords: Aims: To investigate the efficacy of Direct Acting Antivirals (DAAs) in the treatment of different Hepatitis C HCV Virus (HCV) genotypes. NS3/4A protease Main methods: Homology modeling is used to predict the 3D structures of different genotypes while molecular Molecular docking docking is employed to predict genotype – drug interactions (Binding Mode) and binding free energy (Docking Homology modeling Score). Key findings: Simeprevir (TMC435) and to a lesser degree MK6325 are the best drugs among the studied drugs. The predicted affinity of drugs against genotype 1a is always better than other genotypes. P2–P4 macrocyclic drugs show better performance against genotypes 2, 3 and 5. Macrocyclic drugs are better than linear drugs. Significance: HCV is one of the major health problems worldwide. Until the discovery of DAAs, HCV treatment faced many failures. DAAs target key functional machines of the virus life cycle and shut it down. NS3/4A protease is an important target and several drugs have been designed to inhibit its functions. There are several NS3/4A protease drugs approved by Food and Drug Administration (FDA). Unfortunately, the virus exhibits resistance against these drugs. This study is significant in elucidating that no one drug is able to treat different genotypes with the same efficiency. -

Supporting Information
Electronic Supplementary Material (ESI) for Chemical Science. This journal is © The Royal Society of Chemistry 2015 Supporting Information A Single Crystalline Porphyrinic Titanium Metal−Organic Framework Shuai Yuana†, Tian-Fu Liua†, Dawei Fenga, Jian Tiana, Kecheng Wanga, Junsheng Qina, a a a a b Qiang Zhang , Ying-Pin Chen , Mathieu Bosch , Lanfang Zou , Simon J. Teat, Scott J. c a Dalgarno and Hong-Cai Zhou * a Department of Chemistry, Texas A&M University, College Station, Texas 77842-3012, USA b Advanced Light Source, Lawrence Berkeley National Laboratory Berkeley, CA 94720, USA c Institute of Chemical Sciences, Heriot-Watt University Riccarton, Edinburgh EH14 4AS, U.K. † Equal contribution to this work *To whom correspondence should be addressed. Email: [email protected] Tel: +1 (979) 845-4034; Fax: +1 (979) 845-1595 S1 Contents S1. Ligand Synthesis..............................................................................................................3 S2. Syntheses of PCN-22.......................................................................................................5 S3. X-ray Crystallography .....................................................................................................6 S4. Topological Analyses ......................................................................................................9 S5. N2 Sorption Isotherm .....................................................................................................10 S6. Simulation of the Accessible Surface Area ...................................................................12 -

Reactive Molecular Dynamics Study of the Thermal Decomposition of Phenolic Resins
Article Reactive Molecular Dynamics Study of the Thermal Decomposition of Phenolic Resins Marcus Purse 1, Grace Edmund 1, Stephen Hall 1, Brendan Howlin 1,* , Ian Hamerton 2 and Stephen Till 3 1 Department of Chemistry, Faculty of Engineering and Physical Sciences, University of Surrey, Guilford, Surrey GU2 7XH, UK; [email protected] (M.P.); [email protected] (G.E.); [email protected] (S.H.) 2 Bristol Composites Institute (ACCIS), Department of Aerospace Engineering, School of Civil, Aerospace, and Mechanical Engineering, University of Bristol, Bristol BS8 1TR, UK; [email protected] 3 Defence Science and Technology Laboratory, Porton Down, Salisbury SP4 0JQ, UK; [email protected] * Correspondence: [email protected]; Tel.: +44-1483-686-248 Received: 6 March 2019; Accepted: 23 March 2019; Published: 28 March 2019 Abstract: The thermal decomposition of polyphenolic resins was studied by reactive molecular dynamics (RMD) simulation at elevated temperatures. Atomistic models of the polyphenolic resins to be used in the RMD were constructed using an automatic method which calls routines from the software package Materials Studio. In order to validate the models, simulated densities and heat capacities were compared with experimental values. The most suitable combination of force field and thermostat for this system was the Forcite force field with the Nosé–Hoover thermostat, which gave values of heat capacity closest to those of the experimental values. Simulated densities approached a final density of 1.05–1.08 g/cm3 which compared favorably with the experimental values of 1.16–1.21 g/cm3 for phenol-formaldehyde resins. -

Dmol Guide to Select a Dmol3 Task 1
DMOL3 GUIDE MATERIALS STUDIO 8.0 Copyright Notice ©2014 Dassault Systèmes. All rights reserved. 3DEXPERIENCE, the Compass icon and the 3DS logo, CATIA, SOLIDWORKS, ENOVIA, DELMIA, SIMULIA, GEOVIA, EXALEAD, 3D VIA, BIOVIA and NETVIBES are commercial trademarks or registered trademarks of Dassault Systèmes or its subsidiaries in the U.S. and/or other countries. All other trademarks are owned by their respective owners. Use of any Dassault Systèmes or its subsidiaries trademarks is subject to their express written approval. Acknowledgments and References To print photographs or files of computational results (figures and/or data) obtained using BIOVIA software, acknowledge the source in an appropriate format. For example: "Computational results obtained using software programs from Dassault Systèmes Biovia Corp.. The ab initio calculations were performed with the DMol3 program, and graphical displays generated with Materials Studio." BIOVIA may grant permission to republish or reprint its copyrighted materials. Requests should be submitted to BIOVIA Support, either through electronic mail to [email protected], or in writing to: BIOVIA Support 5005 Wateridge Vista Drive, San Diego, CA 92121 USA Contents DMol3 1 Setting up a molecular dynamics calculation20 Introduction 1 Choosing an ensemble 21 Further Information 1 Defining the time step 21 Tasks in DMol3 2 Defining the thermostat control 21 Energy 3 Constraints during dynamics 21 Setting up the calculation 3 Setting up a transition state calculation 22 Dynamics 4 Which method to use? -

EPSRC Service Level Agreement with STFC for Computational Science Support
CoSeC Computational Science Centre for Research Communities EPSRC Service Level Agreement with STFC for Computational Science Support FY 2016/17 Report and Update on FY 2017/18 Work Plans This document contains the 2016/17 plans, 2016/17 summary reports, and 2017/18 plans for the programme in support of CCP and HEC communities delivered by STFC and funded by EPSRC through a Service Level Agreement. Notes in blue are in-year updates on progress to the tasks included in the 2016/17 plans. Text highlighted in yellow shows changes to the draft 2017/18 plans that we submitted in January 2017. Contents CCP5 – Computer Simulation of Condensed Phases .......................................................................... 4 CCP5 – 2016 / 17 Plans (1 April 2016 – 31 March 2017) ...................................................... 4 CCP5 – Summary Report (1 April 2016 – 31 March 2017) .................................................... 7 CCP5 –2017 / 18 Plans (1 April 2017 – 31 March 2018) ....................................................... 8 CCP9 – Electronic Structure of Solids .................................................................................................. 9 CCP9 – 2016 / 17 Plans (1 April 2016 – 31 March 2017) ...................................................... 9 CCP9 – Summary Report (1 April 2016 – 31 March 2017) .................................................. 11 CCP9 – 2017 / 18 Plans (1 April 2017 – 31 March 2018) .................................................... 12 CCP-mag – Computational Multiscale -

Kingston University London the Antibiotic Resistance Growth Plate
Kingston University London The Antibiotic Resistance Growth Plate (ARGP) as an experimental evolution tool to explore the phenotypic and genotypic mutational pathways underlying the emergence of antimicrobial resistance in Escherichia coli. A thesis submitted in partial fulfilment for the degree of Doctor of Philosophy By Lucky Bonnie Lucia CULLEN Faculty of Science, Engineering and Computing February 2019 Declaration This thesis entitled ‘The Antibiotic Resistance Growth Plate (ARGP)’ as an experimental evolution tool to explore the phenotypic and genotypic mutational pathways underlying the emergence of antimicrobial resistance in Escherichia coli’ is based upon the work conducted in the Faculty of Science, Engineering and Computing at Kingston University London and in collaboration with Dr Philip Aldridge at Newcastle University and Katie Hopkins and Neil Woodford from Public Health England. All the work described is the candidate’s own original work unless otherwise stated. None of the work presented has been submitted for another degree internally or externally. i Acknowledgements At the start of this PhD I had a dream, a dream which could have never been accomplished without the support of many people. Firstly, I would like to express my sincere appreciation to my director of studies Professor Mark Fielder, your provision, guidance and unconditional belief in my potential has contributed significantly to the completion of this thesis. I would also like to acknowledge the overwhelming support received from, Dr Scott Lawton, Camilla Eldridge and Ben Jones, your wealth of knowledge and expertise in the field of evolutionary biology and bioinformatics has assisted greatly in concluding the work within this PhD. I would also like to praise the incredible multidisciplinary support received at Kingston University, specifically that from Dr Gary-Forster Wilkins, Dr Adam Le Gresley, Dr Brian Rooney, Dr James Denholm-Price and Richard Giddens. -

Conformational Analysis with Scigress
CONFORMATIONAL ANALYSIS WITH SCIGRESS During this laboratory period you will use two computational chemistry methods to carry out conformational analysis experiments on butane, cyclohexanol and cis-1,3-cyclohexanediol. Mechanics is a method which uses classical Newtonian mechanics to compute the energies of molecules. Bonds are treated as springs and atoms as balls attached to these springs. MOPAC is a method which uses quantum mechanics to calculate the energies of molecules and determine their optimum geometries and other properties; MOPAC is an acronym for Molecular Orbital PACkage Conformational Energy of Butane 1. Build and beautify a molecule of butane. Save your drawing with the name "butane." 2. Highlight (select) the four carbon atoms (in order). All other atoms should be grayed out. Choose Adjust→Dihedral Angle from the menu bar. Select “Define Geometry Label” and “Search”. Apply and then click OK. The workspace containing the butane molecule will reappear with the dihedral angle written in blue across it. The letter S will appear next to the value of the dihedral angle indicating that it is a search label. Re-save the drawing 3. Choose Experiment→New from the menu bar; the experiment dialog box should appear. Choose chemical sample conformations (CAChe 5.0 experiments) from the “Property of” box. Choose optimized map in the “Property” box. Click Start (if an error appears click OK and re-try). 4. Two adjacent windows should appear presenting the results of the calculation. The left window is a plot of potential energy versus dihedral angle for the butane molecule. The graph colors represent the relative energies of the butane molecule with various dihedral angles.