Studies of the ABO and FORS Histo-Blood Group Systems: Focus on Flow Cytometric and Genetic Analysis
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Biological and Clinical Aspects of ABO Blood Group System
174 REVIEW Biological and clinical aspects of ABO blood group system Eiji Hosoi Department of Cells and Immunity Analytics, Institute of Health Biosciences, the University of Tokushima Graduate School, Tokushima, Japan Abstract : The ABO blood group was discovered in 1900 by Austrian scientist, Karl Land- steiner. At present, the International Society of Blood Transfusion (ISBT) approves as 29 human blood group systems. The ABO blood group system consists of four antigens (A, B, O and AB). These antigens are known as oligosaccharide antigens, and widely ex- pressed on the membranes of red cell and tissue cells as well as, in the saliva and body fluid. The ABO blood group antigens are one of the most important issues in transfusion medicine to evaluate the adaptability of donor blood cells with bone marrow transplan- tations, and lifespan of the hemocytes. This article reviews the serology, biochemistry and genetic characteristics, and clini- cal application of ABO antigens. J. Med. Invest. 55 : 174-182, August, 2008 Keywords : ABO blood group, glycosyltransferase, ABO allele, cisAB allele, PASA : PCR amplification of spe- cific alleles INTRODUCTION The genes of ABO blood group has been deter- mined at chromosome locus 9 (6-9), and Yamamoto, The ABO blood group system was discovered by et al. cloned and determined the structures. It has Austrian scientist, Karl Landsteiner, who found made it possible to analyze genetically ABO blood three different blood types (A, B and O) in 1900 group antigens using molecular biology techniques from serological differences in blood called the Land- (7, 10 - 18). steiner Law (1). In 1902, DesCasterllo and Sturli dis- covered the fourth type, AB (2). -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

Análise Integrativa De Perfis Transcricionais De Pacientes Com
UNIVERSIDADE DE SÃO PAULO FACULDADE DE MEDICINA DE RIBEIRÃO PRETO PROGRAMA DE PÓS-GRADUAÇÃO EM GENÉTICA ADRIANE FEIJÓ EVANGELISTA Análise integrativa de perfis transcricionais de pacientes com diabetes mellitus tipo 1, tipo 2 e gestacional, comparando-os com manifestações demográficas, clínicas, laboratoriais, fisiopatológicas e terapêuticas Ribeirão Preto – 2012 ADRIANE FEIJÓ EVANGELISTA Análise integrativa de perfis transcricionais de pacientes com diabetes mellitus tipo 1, tipo 2 e gestacional, comparando-os com manifestações demográficas, clínicas, laboratoriais, fisiopatológicas e terapêuticas Tese apresentada à Faculdade de Medicina de Ribeirão Preto da Universidade de São Paulo para obtenção do título de Doutor em Ciências. Área de Concentração: Genética Orientador: Prof. Dr. Eduardo Antonio Donadi Co-orientador: Prof. Dr. Geraldo A. S. Passos Ribeirão Preto – 2012 AUTORIZO A REPRODUÇÃO E DIVULGAÇÃO TOTAL OU PARCIAL DESTE TRABALHO, POR QUALQUER MEIO CONVENCIONAL OU ELETRÔNICO, PARA FINS DE ESTUDO E PESQUISA, DESDE QUE CITADA A FONTE. FICHA CATALOGRÁFICA Evangelista, Adriane Feijó Análise integrativa de perfis transcricionais de pacientes com diabetes mellitus tipo 1, tipo 2 e gestacional, comparando-os com manifestações demográficas, clínicas, laboratoriais, fisiopatológicas e terapêuticas. Ribeirão Preto, 2012 192p. Tese de Doutorado apresentada à Faculdade de Medicina de Ribeirão Preto da Universidade de São Paulo. Área de Concentração: Genética. Orientador: Donadi, Eduardo Antonio Co-orientador: Passos, Geraldo A. 1. Expressão gênica – microarrays 2. Análise bioinformática por module maps 3. Diabetes mellitus tipo 1 4. Diabetes mellitus tipo 2 5. Diabetes mellitus gestacional FOLHA DE APROVAÇÃO ADRIANE FEIJÓ EVANGELISTA Análise integrativa de perfis transcricionais de pacientes com diabetes mellitus tipo 1, tipo 2 e gestacional, comparando-os com manifestações demográficas, clínicas, laboratoriais, fisiopatológicas e terapêuticas. -

Iraqi Academic Scientific Journals
Baghdad Science Journal Vol.11(2)2014 Gene frequencies of ABO and rhesus blood groups in Sabians (Mandaeans), Iraq Alia E. M. Alubadi* Asmaa M. Salih** Maisam B. N. Alkhamesi** Noor J.Ali*** Received 20, December, 2012 Accepted 3, March, 2014 Abstract: The present study aimed to determine the frequency of ABO and Rh blood group antigens among Sabians (Mandaeans) population. This paper document the frequency of ABO and Rh blood groups among the Sabians (Mandaeans) population of Iraq.There is no data available on the ABO/Rh (D) frequencies in the Sabians (Mandaeans) population. Total 341 samples analyzed; phenotype O blood type has the highest frequency 49.9%, followed by A 28.7%, and B 13.8% whereas the lowest prevalent blood group was AB 7.6%. The overall phenotypic frequencies of ABO blood groups were O>A>B>AB. The allelic frequencies of O, A, and B alleles were 0.687, 0.2 and 0.1122 respectively. Rhesus study showed that with a percentage of 96.2% Rh (D) positive is by far the most prevalent, while Rh (d) negative is present only in 3.8% of the total population. The Sabians (Mandaeans) ethnic group showed the same distribution of ABO and Rh blood groups with others ethnic groups in Iraqi population. Key words: Gene, ABO, rhesus blood groups, Sabians, Gene frequencies Introduction: We are very thankful to Shakoori of pre-Arab and pre-Islamic origin. Farhan dakhel and Nisreen Iehad Badri They are Semites and speak a dialect for their support and guidelines in of Eastern Aramaic known as Mandaic. -

ABO in the Context of Blood Transfusion and Beyond
1 ABO in the Context of Blood Transfusion and Beyond Emili Cid, Sandra de la Fuente, Miyako Yamamoto and Fumiichiro Yamamoto Institut de Medicina Predictiva i Personalitzada del Càncer (IMPPC), Badalona, Barcelona, Spain 1. Introduction ABO histo-blood group system is widely acknowledged as one of the antigenic systems most relevant to blood transfusion, but also cells, tissues and organs transplantation. This chapter will illustrate a series of subjects related to blood transfusion but will also give an overview of ABO related topics such as its genetics, biochemistry and its association to human disease as well as a historical section. We decided not to include much detail about the related Lewis oligosaccharide antigens which have been reviewed extensively elsewhere (Soejima & Koda 2005) in order to focus on ABO and allow the inclusion of novel and exciting developments. A/B antigens on ABO group Anti-A/-B in serum Genotype red blood cells O None Anti-A and Anti-B O/O A A Anti-B A/A or A/O B B Anti-A B/B or B/O AB A and B None A/B Table 1. Simple classification of ABO phenotypes and their corresponding genotypes. As its simplest, the ABO system is dictated by a polymorphic gene (ABO) whose different alleles encode for a glycosyltransferase (A or B) that adds a monosaccharide (N-acetyl-D- galactosamine or D-galactose, respectively) to a specific glycan chain, except for the protein O which is not active. The 3 main alleles: A, B and O are inherited in a classical codominant Mendelian fashion (with O being recessive) and produce, when a pair of them are combined in a diploid cell, the very well known four phenotypic groups (see Table 1). -

US 2017/0020926 A1 Mata-Fink Et Al
US 20170020926A1 (19) United States (12) Patent Application Publication (10) Pub. No.: US 2017/0020926 A1 Mata-Fink et al. (43) Pub. Date: Jan. 26, 2017 (54) METHODS AND COMPOSITIONS FOR 62/006,825, filed on Jun. 2, 2014, provisional appli MMUNOMODULATION cation No. 62/006,829, filed on Jun. 2, 2014, provi sional application No. 62/006,832, filed on Jun. 2, (71) Applicant: RUBIUS THERAPEUTICS, INC., 2014, provisional application No. 61/991.319, filed Cambridge, MA (US) on May 9, 2014, provisional application No. 61/973, 764, filed on Apr. 1, 2014, provisional application No. (72) Inventors: Jordi Mata-Fink, Somerville, MA 61/973,763, filed on Apr. 1, 2014. (US); John Round, Cambridge, MA (US); Noubar B. Afeyan, Lexington, (30) Foreign Application Priority Data MA (US); Avak Kahvejian, Arlington, MA (US) Nov. 12, 2014 (US) ................. PCT/US2O14/0653O4 (21) Appl. No.: 15/301,046 Publication Classification (22) PCT Fed: Mar. 13, 2015 (51) Int. Cl. A6II 35/28 (2006.01) (86) PCT No.: PCT/US2O15/02O614 CI2N 5/078 (2006.01) (52) U.S. Cl. S 371 (c)(1), CPC ............. A61K 35/28 (2013.01); C12N5/0641 (2) Date: Sep. 30, 2016 (2013.01): CI2N 5/0644 (2013.01); A61 K Related U.S. Application Data 2035/122 (2013.01) (60) Provisional application No. 62/059,100, filed on Oct. (57) ABSTRACT 2, 2014, provisional application No. 62/025,367, filed on Jul. 16, 2014, provisional application No. 62/006, Provided are cells containing exogenous antigen and uses 828, filed on Jun. 2, 2014, provisional application No. -

Investigation of Adiposity Phenotypes in AA Associated with GALNT10 & Related Pathway Genes
Investigation of Adiposity Phenotypes in AA Associated With GALNT10 & Related Pathway Genes By Mary E. Stromberg A Dissertation Submitted to the Graduate Faculty of WAKE FOREST UNIVERSITY GRADUATE SCHOOL OF ARTS AND SCIENCES in Partial Fulfillment of the Requirements for the Degree of DOCTOR OF PHILOSOPHY In Molecular Genetics and Genomics December 2018 Winston-Salem, North Carolina Approved by: Donald W. Bowden, Ph.D., Advisor Maggie C.Y. Ng, Ph.D., Advisor Timothy D. Howard, Ph.D., Chair Swapan Das, Ph.D. John P. Parks, Ph.D. Acknowledgements I would first like to thank my mentors, Dr. Bowden and Dr. Ng, for guiding my learning and growth during my years at Wake Forest University School of Medicine. Thank you Dr. Ng for spending so much time ensuring that I learn every detail of every protocol, and supporting me through personal difficulties over the years. Thank you Dr. Bowden for your guidance in making me a better scientist and person. I would like to thank my committee for their patience and the countless meetings we have had in discussing this project. I would like to say thank you to the members of our lab as well as the Parks lab for their support and friendship as well as their contributions to my project. Special thanks to Dean Godwin for his support and understanding. The umbrella program here at WFU has given me the chance to meet some of the best friends I could have wished for. I would like to also thank those who have taught me along the way and helped me to get to this point of my life, with special thanks to the late Dr. -

Identification, Characterization, and Utilization of Glycosyltransferases
Identification, Characterization, and Utilization of Glycosyltransferases DISSERTATION Presented in Partial Fulfillment of the Requirements for the Degree Doctor of Philosophy in the Graduate School of The Ohio State University By Nicholas Roman Pettit Graduate Program in Chemistry The Ohio State University 2011 Dissertation Committee: Dr. Peng George Wang, Advisor Dr. Zucai Suo Dr. Karin Musier-Forsyth Copyright by Nicholas Pettit 2011 3 ABSTRACT Among the many biomolecules found in nature, such as amino acids and DNA, carbohydrates are among the most important and abundant. The assembly of carbohydrates to build various oligosaccharides or glycans is of considerable interest in the glycobiology community, as understanding, controlling, and manipulating such assemblies can yield many interesting insights. This is because glycans are directly involved with many critical biological processes such as molecular recognition, cell-to- cell communication, protein folding and stability, as well as being involved many unique disease state phenotypes. The assembly of these glycans is commonly accomplished by a class of enzymes called glycosyltransferases, and it is these enzymes which have been exploited in vitro for the synthesis of attractive oligosaccharides. However, the identification, characterization, and subsequent exploitation of a glycosyltransferase is a very tedious and long process which stems from the root problem that knowing the primary function of a glycosyltransferase must be determined experimentally. This is because glycans -

Molecular Evolution of the Glycosyltransferase 6 Gene Family in Primates
Hindawi Publishing Corporation Biochemistry Research International Volume 2016, Article ID 9051727, 6 pages http://dx.doi.org/10.1155/2016/9051727 Research Article Molecular Evolution of the Glycosyltransferase 6 Gene Family in Primates Eliane Evanovich,1 Patricia Jeanne de Souza Mendonça-Mattos,1 and Maria Lúcia Harada2 1 Laboratorio´ de Genetica´ Humana e Medica,´ Instituto de Cienciasˆ Biologicas,´ Universidade Federal do Para,´ Belem,´ PA, Brazil 2Laboratorio´ de Biologia Molecular “Francisco Mauro Salzano”, Instituto de Cienciasˆ Biologicas,´ Universidade Federal do Para,´ Belem,´ PA, Brazil Correspondence should be addressed to Eliane Evanovich; [email protected] Received 12 July 2016; Accepted 20 October 2016 Academic Editor: Stefano Pascarella Copyright © 2016 Eliane Evanovich et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Glycosyltransferase 6 gene family includes ABO, Ggta1, iGb3S, and GBGT1 genes and by three putative genes restricted to mammals, GT6m6, GTm6, and GT6m7, only the latter is found in primates. GT6 genes may encode functional and nonfunctional proteins. Ggta1 and GBGT1 genes, for instance, are pseudogenes in catarrhine primates, while iGb3S gene is only inactive in human, bonobo, and chimpanzee. Even inactivated, these genes tend to be conversed in primates. As some of the GT6 genes are related to the susceptibility or resistance to parasites, we investigated (i) the selective pressure on the GT6 paralogs genes in primates; (ii) the basis of the conservation of iGb3S in human, chimpanzee, and bonobo; and (iii) the functional potential of the GBGT1 and GT6m7 in catarrhines. -

Genetic Characterisation of Human ABO Blood Group Variants with a Focus on Subgroups and Hybrid Alleles Hosseini Maaf, Bahram
Genetic Characterisation of Human ABO Blood Group Variants with a Focus on Subgroups and Hybrid Alleles Hosseini Maaf, Bahram 2007 Link to publication Citation for published version (APA): Hosseini Maaf, B. (2007). Genetic Characterisation of Human ABO Blood Group Variants with a Focus on Subgroups and Hybrid Alleles. Division of Hematology and Transfusion Medicine, Department of Laboratory Medicine, Lund University. Total number of authors: 1 General rights Unless other specific re-use rights are stated the following general rights apply: Copyright and moral rights for the publications made accessible in the public portal are retained by the authors and/or other copyright owners and it is a condition of accessing publications that users recognise and abide by the legal requirements associated with these rights. • Users may download and print one copy of any publication from the public portal for the purpose of private study or research. • You may not further distribute the material or use it for any profit-making activity or commercial gain • You may freely distribute the URL identifying the publication in the public portal Read more about Creative commons licenses: https://creativecommons.org/licenses/ Take down policy If you believe that this document breaches copyright please contact us providing details, and we will remove access to the work immediately and investigate your claim. LUND UNIVERSITY PO Box 117 221 00 Lund +46 46-222 00 00 Genetic Characterisation of Human ABO Blood Group Variants with a Focus on Subgroups and Hybrid -

Human Social Genomics in the Multi-Ethnic Study of Atherosclerosis
Getting “Under the Skin”: Human Social Genomics in the Multi-Ethnic Study of Atherosclerosis by Kristen Monét Brown A dissertation submitted in partial fulfillment of the requirements for the degree of Doctor of Philosophy (Epidemiological Science) in the University of Michigan 2017 Doctoral Committee: Professor Ana V. Diez-Roux, Co-Chair, Drexel University Professor Sharon R. Kardia, Co-Chair Professor Bhramar Mukherjee Assistant Professor Belinda Needham Assistant Professor Jennifer A. Smith © Kristen Monét Brown, 2017 [email protected] ORCID iD: 0000-0002-9955-0568 Dedication I dedicate this dissertation to my grandmother, Gertrude Delores Hampton. Nanny, no one wanted to see me become “Dr. Brown” more than you. I know that you are standing over the bannister of heaven smiling and beaming with pride. I love you more than my words could ever fully express. ii Acknowledgements First, I give honor to God, who is the head of my life. Truly, without Him, none of this would be possible. Countless times throughout this doctoral journey I have relied my favorite scripture, “And we know that all things work together for good, to them that love God, to them who are called according to His purpose (Romans 8:28).” Secondly, I acknowledge my parents, James and Marilyn Brown. From an early age, you two instilled in me the value of education and have been my biggest cheerleaders throughout my entire life. I thank you for your unconditional love, encouragement, sacrifices, and support. I would not be here today without you. I truly thank God that out of the all of the people in the world that He could have chosen to be my parents, that He chose the two of you. -
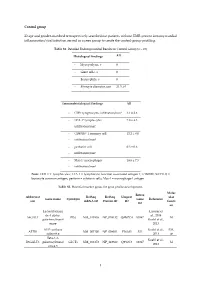
1 Control Group 23 Age and Gender-Matched
Control group 23 age and gender-matched retrospectively searched out patients without EMB-proven intramyocardial inflammation/viral infection served as a peer group to create the control-group profiling. Table S1. Detailed Endomyocardial Results in Control Group (n = 23) Histological findings All - Myocytolysis, n 0 - Giant cells, n 0 - Eosinophils, n 0 - Myocyte diameter, µm 21.3 ± 0 Immunohistological findings All - CD3+ lymphocytes infiltration/mm² 3.1 ± 2.6 - LFA-1+ lymphocytes 7.8 ± 4.3 infiltration/mm² - CD45R0+ T memory cell 12.2 ± 8.0 - infiltration/mm² - perforin+ cell 0.3 ± 0.6 - infiltration/mm² - Mac-1+ macrophages 18.6 ± 7.5 - infiltration/mm² Note: CD3 = T lymphocytes; LFA-1 = lymphocyte function-associated antigen-1; CD45R0 (UCHL1) = leucocyte common antigen; perforin = cytotoxic cells; Mac-1 = macrophage-1 antigen. Table S2. Potential marker genes for gene profile development. Molec Entrez Abbreviat RefSeq RefSeq Uniprot ular Gene name Synonym Gene Reference ion mRNA ID Protein ID ID functi ID on Lactosylcerami Lassner et de 4-alpha- al., 2014 A4GALT P(k) NM_017436 NP_059132 Q9NPC4 53947 M galactosyltransf Kuehl et al., erase 2013 ATP synthase Kuehl et al., EM, ATP5I NM_007100 NP_009031 P56385 521 subunit g 2013 ce Beta-1,3- Kuehl et al., B3GALT5 galactosyltransf GLCT5 NM_033172 NP_149362 Q9Y2C3 10317 M 2013 erase 5 1 BAG family molecular Kuehl et al., BAG5 NM_004873 NP_004864 Q9UL15 9529 CC chaperone 2013 regulator 5 Lassner et CC-chemokine al., 2014 CCL20 ST38 NM_004591 NP_004582 P78556 6364 IR ligand 20 Kuehl et al.,