ARISTADA™ Safely and Effectively
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Aripiprazole As an Adjunct to Anti-Depressants for Major Depressive Disorder: Clinical Effectiveness, Cost-Effectiveness, and Guidelines
TITLE: Aripiprazole as an Adjunct to Anti-Depressants for Major Depressive Disorder: Clinical Effectiveness, Cost-Effectiveness, and Guidelines DATE: 04 May 2016 RESEARCH QUESTIONS 1. What is the clinical efficacy of aripiprazole as an adjunct to antidepressants for the treatment of patients with major depressive disorder who have had an inadequate response to prior antidepressant treatments? 2. What is the cost-effectiveness of aripiprazole as an adjunct to antidepressants for the treatment of patients with major depressive disorder who have had an inadequate response to prior antidepressant treatments? 3. What are the evidence-based guidelines for the use of aripiprazole for the treatment of patients with major depressive disorder who have had an inadequate response to prior antidepressant treatments? KEY FINDINGS Three systematic reviews with meta-analyses, two randomized controlled trials, and two evidence-based guidelines were identified regarding the use of aripiprazole as an adjunct to antidepressants for the treatment of patients with major depressive disorder who have had an inadequate response to prior antidepressant treatments. METHODS A limited literature search was conducted on key resources including PubMed, Ovid PsychINFO, Ovid Embase, The Cochrane Library, University of York Centre for Reviews and Dissemination (CRD) databases, Canadian and major international health technology agencies, as well as a focused Internet search. No methodological filters were used to limit retrieval by study type. Where possible, retrieval was limited to the human population. The search was also limited to English language documents published between January 1, 2014 and April 29, 2016. Disclaimer: The Rapid Response Service is an information service for those involved in planning and providing health care in Canada. -

Aristada™ (Aripiprazole Lauroxil)
Aristada™ (aripiprazole lauroxil) – New Drug Approval • On October 5, 2015, Alkermes’ announced the FDA approval of Aristada (aripiprazole lauroxil) extended-release injection, an atypical antipsychotic, for the treatment of schizophrenia. • Schizophrenia is a chronic, severe and disabling brain disorder affecting an estimated 2.4 million Americans. Typically, symptoms include hearing voices, believing other people are reading their minds or controlling their thoughts, and being suspicious or withdrawn. • Aristada’s approval was based on data from a double-blind, placebo-controlled 12-week trial involving 622 patients with schizophrenia. In addition, the efficacy of Aristada was established, in part, on the basis of efficacy data from trials with oral aripiprazole. — Aristada significantly improved symptoms of schizophrenia compared to placebo at day 85. • Similar to other atypical antipsychotics, Aristada carries a boxed warning for increased mortality in elderly patients with dementia-related psychosis. • Other warnings and precautions for Aristada include cerebrovascular adverse reactions, including stroke; neuroleptic malignant syndrome; tardive dyskinesia; metabolic changes; orthostatic hypotension; leukopenia, neutropenia, and agranulocytosis; seizures; potential for cognitive and motor impairment; body temperature regulation; and dysphagia. • The most common adverse reaction (≥ 5% and at least twice that for placebo) with Aristada use was akathisia. • Aristada is administered by intramuscular injection in the deltoid (441 mg dose only) or gluteal (441 mg, 662 mg, or 882 mg) muscle by a healthcare professional. — Aristada can be initiated at a monthly dose (441 mg, 662 mg or 882 mg) or every 6 week dose (882 mg). — For patients naïve to aripiprazole, tolerability should be established with oral aripiprazole prior to initiating treatment with Aristada. -

SIGHI-Leaflet Histamine Elimination Diet Simplified Histamine Elimination Diet for Histamine Intolerance (DAO Degradation Disorder)
Version 2020-07-20 SIGHI-Leaflet Histamine Elimination Diet Simplified histamine elimination diet for histamine intolerance (DAO degradation disorder) For people with a DAO degradation disorder The compatibility is highly dependent on the in- who have to avoid histamine, other biogenic dividual sensitivity and the amount consumed. amines and DAO inhibitors in their diet. Furthermore, it is temporarily affected by stress, In case of histamine sensitivity due to mast cell hormones and many other factors. First and activation disorders (MCAD) this dietary guide- foremost, the freshness is an important cri- line is not sufficient! If no permanent symptom terion. Everybody has to find out by trial and er- relief can be achieved and maintained with this ror what he/she can tolerate in what quantities. diet, please follow the detailed list, which addi- At the beginning of the experimental diet this list tionally takes histamine liberators into consid- should be followed as consistently as possible. eration as completely as possible. It is available In a later stage, however, the diet should be here: based more on the experiences of the person www.mastzellaktivierung.info concerned rather than following any list. Mast cell activation disorders are often mistaken Always read the list of ingredients to find out for histamine intolerance. whether a food contains incompatible ingredi- ents. References: • Experience reports from among our members and readers • Various patient leaflets from doctors, clinics and hospitals • Experience of other patient organizations, bloggers, forum threads etc. • Scientific publications • Textbooks and cookbooks about histamine intolerance To avoid: ? Risky: Well tolerated: Fermented or microbially ripened Meals from res- Prefer fresh, unprocessed or little pro- products (e.g. -

Download Executive Summary
Comparative Effectiveness Review Number 43 Effective Health Care Program Off-Label Use of Atypical Antipsychotics: An Update Executive Summary Background Effective Health Care Program Antipsychotics medications are approved The Effective Health Care Program by the U.S. Food and Drug Administration was initiated in 2005 to provide valid (FDA) for treatment of schizophrenia and evidence about the comparative bipolar disorder. These medications are effectiveness of different medical commonly divided into two classes, interventions. The object is to help reflecting two waves of historical consumers, health care providers, and development: the conventional others in making informed choices antipsychotics and the atypical. The among treatment alternatives. Through conventional antipsychotics served as the its Comparative Effectiveness Reviews, first successful pharmacologic treatment the program supports systematic for primary psychotic disorders such as appraisals of existing scientific schizophrenia. Having been widely used evidence regarding treatments for for decades, the conventional high-priority health conditions. It also antipsychotics also produced various side promotes and generates new scientific effects requiring additional medications, evidence by identifying gaps in which spurred the development of the existing scientific evidence and atypical antipsychotics. supporting new research. The program Currently, nine atypical antipsychotic drugs puts special emphasis on translating have been approved by FDA: aripiprazole, findings into a variety of useful asenapine, clozapine, iloperidone, formats for different stakeholders, olanzapine, paliperidone, quetiapine, including consumers. risperidone, and ziprasidone. These drugs The full report and this summary are have been used off-label (i.e., for available at www.effectivehealthcare. indications not approved by FDA) for the ahrq.gov/reports/final.cfm. treatment of various psychiatric conditions. -

Addyi Generic Name: Flibanserin Manufacturer
Brand Name: Addyi Generic Name: Flibanserin Manufacturer: Sprout Pharmaceuticals Drug Class: Central Nervous System Agent, Serotonin Agonist, Dopamine antagonist Uses: Labeled Uses: Indicated for the treatment of premenopausal women with acquired, generalized hypoactive sexual desire disorder (HSDD) as characterized by low sexual desire that causes marked distress or interpersonal difficulty and is NOT due to: A co-existing medical or psychiatric condition, problems within the relationship, or the effects of a medication or other drug substance. Unlabeled Uses: none. Mechanism of Action: The mechanism of action for flibanserin in the treatment of hypoactive sexual desire disorder is unknown. Flibanserin has high affinity for serotonin (5-hydroxytryptamine or 5-HT) 1A receptors, as an agonist, and 5-HT2A receptors, as an antagonist, and moderate affinity for 5- HT2B, 5-HT2C, and dopamine D4 receptors as an antagonist Pharmacokinetics: Absorption: Tmax 0.75 hours Vd 50L t ½ 11 hours Clearance Not reported Protein binding 98% (albumin) Bioavailability 33% Metabolism: Flibanserin is extensively metabolized primarily by CYP3A4 and, to a lesser extent, CYP2C19 to at least 35 metabolites, with most of the metabolites occurring in low concentrations in plasma. Elimination: Flibanserin is primarily excreted through the kidneys in to urine (44%) and feces (51%). Two metabolites could be characterized that showed plasma concentration similar to that achieved with flibanserin: 6,21-dihydroxy-flibanserin-6,21-disulfate and 6- hydroxy-flibanserin-6-sulfate. These two metabolites are inactive. Efficacy: Katz M, DeRogatis LR, Ackerman R, et al. Efficacy of flibanserin in women with hypoactive sexual desire disorder: results from the BEGONIA trial. J Sex Med. -

Modern Antipsychotic Drugs: a Critical Overview
Review Synthèse Modern antipsychotic drugs: a critical overview David M. Gardner, Ross J. Baldessarini, Paul Waraich Abstract mine was based primarily on the finding that dopamine ago- nists produced or worsened psychosis and that antagonists CONVENTIONAL ANTIPSYCHOTIC DRUGS, used for a half century to treat were clinically effective against psychotic and manic symp- a range of major psychiatric disorders, are being replaced in clini- 5 toms. Blocking dopamine D2 receptors may be a critical or cal practice by modern “atypical” antipsychotics, including ari- even sufficient neuropharmacologic action of most clinically piprazole, clozapine, olanzapine, quetiapine, risperidone and effective antipsychotic drugs, especially against hallucina- ziprasidone among others. As a class, the newer drugs have been promoted as being broadly clinically superior, but the evidence for tions and delusions, but it is not necessarily the only mecha- this is problematic. In this brief critical overview, we consider the nism for antipsychotic activity. Moreover, this activity, and pharmacology, therapeutic effectiveness, tolerability, adverse ef- subsequent pharmacocentric and circular speculations about fects and costs of individual modern agents versus older antipsy- altered dopaminergic function, have not led to a better un- chotic drugs. Because of typically minor differences between derstanding of the pathophysiology or causes of the several agents in clinical effectiveness and tolerability, and because of still idiopathic psychotic disorders, nor have they provided a growing concerns about potential adverse long-term health conse- non-empirical, theoretical basis for the design or discovery quences of some modern agents, it is reasonable to consider both of improved treatments for psychotic disorders. older and newer drugs for clinical use, and it is important to inform The neuropharmacodynamics of specific modern anti- patients of relative benefits, risks and costs of specific choices. -

Comparative Effects of Cimetidine and Famotidine on the Vagally Stimulated Acid Secretion in the Isolated Mouse Whole Stomach
Comparative Effects of Cimetidine and Famotidine on the Vagally Stimulated Acid Secretion in the Isolated Mouse Whole Stomach Kazuo Watanabe1, Shingo Yano1, Masayuki Yamamoto1 and Shoko Kanaoka2 1Laboratory of Chemical Pharmacology, Department of Drug Evaluation and Toxicological Sciences, Faculty of Pharmaceutical Sciences, Chiba University, 1-33, Yayoi-cho, Inage-ku, Chiba 263, Japan 2Research Institute for Wakan-Yaku, Toyama Medical and Pharmaceutical University, Toyama 930-01, Japan Received July 15, 1992 Accepted December 10, 1992 ABSTRACT-We investigated the effects of cimetidine and famotidine on the acid secretory response to elec trical vagal stimulation, bethanechol and histamine in the isolated mouse whole stomach preparation. The acid secretion elicited by electrical vagal stimulation at the position of the esophagus (10 Hz, 0.3 msec, 10 V for 5 min) was reproducible by repeated stimulation in each preparation, and it was abolished by tetrodo toxin, atropine and hexamethonium. This vagally stimulated acid secretion was abolished by cimetidine (3 mM), while it was only partly inhibited by famotidine (10-100 ƒÊM). Histamine (100 ƒÊM)-induced acid secre tion was inhibited by cimetidine and famotidine, and the doses of these drugs required for complete inhibi tion were 3 mM and 10 ƒÊM, respectively. In contrast, bethanechol (10 ƒÊM)-induced acid secretion was slight ly reduced by famotidine (1-100 ƒÊM), but markedly reduced by cimetidine (3 mM). In the guinea pig ileum, millimolar concentrations of cimetidine and famotidine shifted the dose-response curve of the contractile response to acetylcholine rightward. These findings suggest that the inhibitory effect of cimetidine on the vagally stimulated or bethanechol-induced acid secretion is elicited at least partly through mechanisms different from H2-antagonism. -
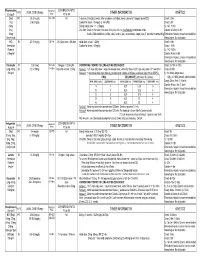
Medication Conversion Chart
Fluphenazine FREQUENCY CONVERSION RATIO ROUTE USUAL DOSE (Range) (Range) OTHER INFORMATION KINETICS Prolixin® PO to IM Oral PO 2.5-20 mg/dy QD - QID NA ↑ dose by 2.5mg/dy Q week. After symptoms controlled, slowly ↓ dose to 1-5mg/dy (dosed QD) Onset: ≤ 1hr 1mg (2-60 mg/dy) Caution for doses > 20mg/dy (↑ risk EPS) Cmax: 0.5hr 2.5mg Elderly: Initial dose = 1 - 2.5mg/dy t½: 14.7-15.3hr 5mg Oral Soln: Dilute in 2oz water, tomato or fruit juice, milk, or uncaffeinated carbonated drinks Duration of Action: 6-8hr 10mg Avoid caffeinated drinks (coffee, cola), tannics (tea), or pectinates (apple juice) 2° possible incompatibilityElimination: Hepatic to inactive metabolites 5mg/ml soln Hemodialysis: Not dialyzable HCl IM 2.5-10 mg/dy Q6-8 hr 1/3-1/2 po dose = IM dose Initial dose (usual): 1.25mg Onset: ≤ 1hr Immediate Caution for doses > 10mg/dy Cmax: 1.5-2hr Release t½: 14.7-15.3hr 2.5mg/ml Duration Action: 6-8hr Elimination: Hepatic to inactive metabolites Hemodialysis: Not dialyzable Decanoate IM 12.5-50mg Q2-3 wks 10mg po = 12.5mg IM CONVERTING FROM PO TO LONG-ACTING DECANOATE: Onset: 24-72hr (4-72hr) Long-Acting SC (12.5-100mg) (1-4 wks) Round to nearest 12.5mg Method 1: 1.25 X po daily dose = equiv decanoate dose; admin Q2-3wks. Cont ½ po daily dose X 1st few mths Cmax: 48-96hr 25mg/ml Method 2: ↑ decanoate dose over 4wks & ↓ po dose over 4-8wks as follows (accelerate taper for sx of EPS): t½: 6.8-9.6dy (single dose) ORAL DECANOATE (Administer Q 2 weeks) 15dy (14-100dy chronic administration) ORAL DOSE (mg/dy) ↓ DOSE OVER (wks) INITIAL DOSE (mg) TARGET DOSE (mg) DOSE OVER (wks) Steady State: 2mth (1.5-3mth) 5 4 6.25 6.25 0 Duration Action: 2wk (1-6wk) Elimination: Hepatic to inactive metabolites 10 4 6.25 12.5 4 Hemodialysis: Not dialyzable 20 8 6.25 12.5 4 30 8 6.25 25 4 40 8 6.25 25 4 Method 3: Admin equivalent decanoate dose Q2-3wks. -

Drug Use Evaluation: Antipsychotic Utilization in Schizophrenia Patients
© Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 | Fax 503-947-1119 Drug Use Evaluation: Antipsychotic Utilization in Schizophrenia Patients Research Questions: 1. How many schizophrenia patients are prescribed recommended first-line second-generation treatments for schizophrenia? 2. How many schizophrenia patients switch to an injectable antipsychotic after stabilization on an oral antipsychotic? 3. How many schizophrenia patients are prescribed 2 or more concomitant antipsychotics? 4. Are claims for long-acting injectable antipsychotics primarily billed as pharmacy or physician administered claims? 5. Does adherence to antipsychotic therapy differ between patients with claims for different routes of administration (oral vs. long-acting injectable)? Conclusions: In total, 4663 schizophrenia patients met inclusion criteria, and approximately 14% of patients (n=685) were identified as treatment naïve without claims for antipsychotics in the year before their first antipsychotic prescription. Approximately 45% of patients identified as treatment naïve had a history of remote antipsychotic use, but it is unclear if antipsychotics were historically prescribed for schizophrenia. Oral second-generation antipsychotics which are recommended as first-line treatment in the MHCAG schizophrenia algorithm were prescribed as initial treatment in 37% of treatment naive patients and 28% of all schizophrenia patients. Recommended agents include risperidone, paliperidone, and aripiprazole. Utilization of parenteral antipsychotics was limited in patients with schizophrenia. Overall only 8% of patients switched from an oral to an injectable therapy within 6 months of their first claim. Approximately, 60% of all schizophrenia patients (n=2512) had claims for a single antipsychotic for at least 12 continuous weeks and may be eligible to transition to a long-acting injectable antipsychotic. -

THE USE of MIRTAZAPINE AS a HYPNOTIC O Uso Da Mirtazapina Como Hipnótico Francisca Magalhães Scoralicka, Einstein Francisco Camargosa, Otávio Toledo Nóbregaa
ARTIGO ESPECIAL THE USE OF MIRTAZAPINE AS A HYPNOTIC O uso da mirtazapina como hipnótico Francisca Magalhães Scoralicka, Einstein Francisco Camargosa, Otávio Toledo Nóbregaa Prescription of approved hypnotics for insomnia decreased by more than 50%, whereas of antidepressive agents outstripped that of hypnotics. However, there is little data on their efficacy to treat insomnia, and many of these medications may be associated with known side effects. Antidepressants are associated with various effects on sleep patterns, depending on the intrinsic pharmacological properties of the active agent, such as degree of inhibition of serotonin or noradrenaline reuptake, effects on 5-HT1A and 5-HT2 receptors, action(s) at alpha-adrenoceptors, and/or histamine H1 sites. Mirtazapine is a noradrenergic and specific serotonergic antidepressive agent that acts by antagonizing alpha-2 adrenergic receptors and blocking 5-HT2 and 5-HT3 receptors. It has high affinity for histamine H1 receptors, low affinity for dopaminergic receptors, and lacks anticholinergic activity. In spite of these potential beneficial effects of mirtazapine on sleep, no placebo-controlled randomized clinical trials of ABSTRACT mirtazapine in primary insomniacs have been conducted. Mirtazapine was associated with improvements in sleep on normal sleepers and depressed patients. The most common side effects of mirtazapine, i.e. dry mouth, drowsiness, increased appetite and increased body weight, were mostly mild and transient. Considering its use in elderly people, this paper provides a revision about studies regarding mirtazapine for sleep disorders. KEYWORDS: sleep; antidepressive agents; sleep disorders; treatment� A prescrição de hipnóticos aprovados para insônia diminuiu em mais de 50%, enquanto de antidepressivos ultrapassou a dos primeiros. -

A Comparative Evaluation of Lafutidine and 2 Rabeprazole in The
1 *Original research paper 2 A comparative evaluation of Lafutidine and 3 Rabeprazole in the treatment of gastritis and 4 peptic ulcer: A double-blind, randomized study 5 in Indian patients. 6 Dr. Sanjay Kumar 1, Dr. Bhupesh Dewan 2*, Deepashri Shah 2 7 8 1Global Liver and Gastroenterology Centre, Bhopal, India 2 9 Medical Department, Zuventus Healthcare Ltd., Mumbai, India 10 11 . 12 ABSTRACT 13 Aims: To assess the efficacy of lafutidine therapy versus rabeprazole in Indian patients with endoscopically and histologically proven gastritis and peptic ulcer. Study design: A double blind, double dummy, randomized, comparative study. Place and Duration of Study: Global Liver and Gastroenterology Centre, Bhopal, India, between March 2010 and October 2010. Methodology: A total of 100 patients were enrolled, including 50 with endoscopically and histologically proven gastritis and other 50 with peptic ulcer (over 5 mm in diameter). Each group was randomized to receive either lafutidine or rabeprazole tablet and their corresponding competitor placebo dummy tablet, for a period of 4 weeks. Gastritis/ulcer cure rates confirmed by endoscopic histology, symptom response and Helicobacter pylori (H. Pylori) eradication were compared among the two drugs Results: Complete cure of gastritis was observed in all the patients (100%) treated with lafutidine and 95.24% [20/21; 95% CI: 76.18 to 99.88%] patients treated with rabeprazole. Complete cure of ulcer was observed in 72.0% (18/25, 95% CI = 50.61 to 87.93%) and 79.16% (19/24, 95% CI = 57.85 to 92.87%) patients treated with lafutidine and rabeprazole respectively. There was no significant difference in gastritis/ulcer cure rate and symptom response rate between the two treatment groups at the end of the study. -

Supplementary Materials
Supplementary Materials Table S1. The significant drug pairs in potential DDIs examined by the two databases. Micromedex Drugs.com List of drugs paired PK-PD Mechanism details 1. Amiodarone— PD Additive QT-interval prolongation Dronedarone 2. Amiodarone— PK CYP3A inhibition by Ketoconazole Ketoconazole 3. Ciprofloxacin— PD Additive QT-interval prolongation Dronedarone 4. Cyclosporine— PK CYP3A inhibition by Cyclosporine Dronedarone 5. Dronedarone— PK CYP3A inhibition by Erythromycin Erythromycin 6. Dronedarone— PD Additive QT-interval prolongation Flecainide 7. Dronedarone— PK CYP3A4 inhibition by Itraconazole Itraconazole 8. Dronedarone— PK Contraindication Major CYP3A inhibition by Ketoconazole Ketoconazole 9. Dronedarone— PD Additive QT-interval prolongation Procainamide PD 10. Dronedarone—Sotalol Additive QT-interval prolongation 11. Felodipine— PK CYP3A inhibition by Itraconazole Itraconazole 12. Felodipine— PK CYP3A inhibition by Ketoconazole Ketoconazole 13. Itraconazole— PK CYP3A inhibition by Itraconazole Nisoldipine 14. Ketoconazole— PK CYP3A inhibition by Ketoconazole Nisoldipine 15. Praziquantel— PK CYP induction by Rifampin Rifampin PD 1. Amikacin—Furosemide Additive or synergistic toxicity 2. Aminophylline— Decreased clearance of PK Ciprofloxacin Theophylline by Ciprofloxacin 3. Aminophylline— PK Decreased hepatic metabolism Mexiletine 4. Amiodarone— PD Additive effects on QT interval Ciprofloxacin 5. Amiodarone—Digoxin PK P-glycoprotein inhibition by Amiodarone 6. Amiodarone— PD, PK Major Major Additive effects on QT Erythromycin prolongation, CYP3A inhibition by Erythromycin 7. Amiodarone— PD, PK Flecainide Antiarrhythmic inhibition by Amiodarone, CYP2D inhibition by Amiodarone 8. Amiodarone— PK CYP3A inhibition by Itraconazole Itraconazole 9. Amiodarone— PD Antiarrhythmic inhibition by Procainamide Amiodarone 10. Amiodarone— PK CYP induction by Rifampin Rifampin PD Additive effects on refractory 11. Amiodarone—Sotalol potential 12. Amiodarone— PK CYP3A inhibition by Verapamil Verapamil 13.