Heme Oxygenase-1 Deficiency and Oxidative Stress
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
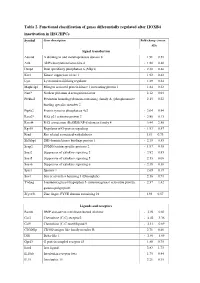
Table 2. Functional Classification of Genes Differentially Regulated After HOXB4 Inactivation in HSC/Hpcs
Table 2. Functional classification of genes differentially regulated after HOXB4 inactivation in HSC/HPCs Symbol Gene description Fold-change (mean ± SD) Signal transduction Adam8 A disintegrin and metalloprotease domain 8 1.91 ± 0.51 Arl4 ADP-ribosylation factor-like 4 - 1.80 ± 0.40 Dusp6 Dual specificity phosphatase 6 (Mkp3) - 2.30 ± 0.46 Ksr1 Kinase suppressor of ras 1 1.92 ± 0.42 Lyst Lysosomal trafficking regulator 1.89 ± 0.34 Mapk1ip1 Mitogen activated protein kinase 1 interacting protein 1 1.84 ± 0.22 Narf* Nuclear prelamin A recognition factor 2.12 ± 0.04 Plekha2 Pleckstrin homology domain-containing. family A. (phosphoinosite 2.15 ± 0.22 binding specific) member 2 Ptp4a2 Protein tyrosine phosphatase 4a2 - 2.04 ± 0.94 Rasa2* RAS p21 activator protein 2 - 2.80 ± 0.13 Rassf4 RAS association (RalGDS/AF-6) domain family 4 3.44 ± 2.56 Rgs18 Regulator of G-protein signaling - 1.93 ± 0.57 Rrad Ras-related associated with diabetes 1.81 ± 0.73 Sh3kbp1 SH3 domain kinase bindings protein 1 - 2.19 ± 0.53 Senp2 SUMO/sentrin specific protease 2 - 1.97 ± 0.49 Socs2 Suppressor of cytokine signaling 2 - 2.82 ± 0.85 Socs5 Suppressor of cytokine signaling 5 2.13 ± 0.08 Socs6 Suppressor of cytokine signaling 6 - 2.18 ± 0.38 Spry1 Sprouty 1 - 2.69 ± 0.19 Sos1 Son of sevenless homolog 1 (Drosophila) 2.16 ± 0.71 Ywhag 3-monooxygenase/tryptophan 5- monooxygenase activation protein. - 2.37 ± 1.42 gamma polypeptide Zfyve21 Zinc finger. FYVE domain containing 21 1.93 ± 0.57 Ligands and receptors Bambi BMP and activin membrane-bound inhibitor - 2.94 ± 0.62 -

Carbon Monoxide Prevents TNF-Α-Induced Enos Downregulation by Inhibiting NF-Κb-Responsive Mir-155-5P Biogenesis
OPEN Experimental & Molecular Medicine (2017) 49, e403; doi:10.1038/emm.2017.193 Official journal of the Korean Society for Biochemistry and Molecular Biology www.nature.com/emm ORIGINAL ARTICLE Carbon monoxide prevents TNF-α-induced eNOS downregulation by inhibiting NF-κB-responsive miR-155-5p biogenesis Seunghwan Choi1, Joohwan Kim1, Ji-Hee Kim1, Dong-Keon Lee1, Wonjin Park1, Minsik Park1, Suji Kim1, Jong Yun Hwang2, Moo-Ho Won3, Yoon Kyung Choi1,4, Sungwoo Ryoo5, Kwon-Soo Ha1, Young-Guen Kwon6 and Young-Myeong Kim1 Heme oxygenase-1-derived carbon monoxide prevents inflammatory vascular disorders. To date, there is no clear evidence that HO-1/CO prevents endothelial dysfunction associated with the downregulation of endothelial NO synthesis in human endothelial cells stimulated with TNF-α. Here, we found that the CO-releasing compound CORM-2 prevented TNF-α-mediated decreases in eNOS expression and NO/cGMP production, without affecting eNOS promoter activity, by maintaining the functional activity of the eNOS mRNA 3′-untranslated region. By contrast, CORM-2 inhibited MIR155HG expression and miR-155-5p biogenesis in TNF-α-stimulated endothelial cells, resulting in recovery of the 3′-UTR activity of eNOS mRNA, a target of miR-155-5p. The beneficial effect of CORM-2 was blocked by an NF-κB inhibitor, a miR-155-5p mimic, a HO-1 inhibitor and siRNA against HO-1, indicating that CO rescues TNF-α-induced eNOS downregulation through NF-κB-responsive miR-155-5p expression via HO-1 induction; similar protective effects of ectopic HO-1 expression and bilirubin were observed in endothelial cells treated with TNF-α. -

Targeting Heme Oxygenase-1 in Cardiovascular and Kidney Disease
antioxidants Review Targeting Heme Oxygenase-1 in Cardiovascular and Kidney Disease Heather A. Drummond 1, Zachary L. Mitchell 1, Nader G. Abraham 2,3 and David E. Stec 1,* 1 Department of Physiology and Biophysics, Mississippi Center for Obesity Research, University of Mississippi Medical Center, Jackson, MI 39216, USA; [email protected] (H.A.D.); [email protected] (Z.L.M.) 2 Departments of Medicine and Pharmacology, New York Medical College, Vahalla, NY 10595, USA; [email protected] 3 Joan C. Edwards School of Medicine, Marshall University, Huntington, VA 25701, USA * Correspondence: [email protected]; Tel.: +1-601-815-1859; Fax: +1-601-984-1817 Received: 3 June 2019; Accepted: 15 June 2019; Published: 18 June 2019 Abstract: Heme oxygenase (HO) plays an important role in the cardiovascular system. It is involved in many physiological and pathophysiological processes in all organs of the cardiovascular system. From the regulation of blood pressure and blood flow to the adaptive response to end-organ injury, HO plays a critical role in the ability of the cardiovascular system to respond and adapt to changes in homeostasis. There have been great advances in our understanding of the role of HO in the regulation of blood pressure and target organ injury in the last decade. Results from these studies demonstrate that targeting of the HO system could provide novel therapeutic opportunities for the treatment of several cardiovascular and renal diseases. The goal of this review is to highlight the important role of HO in the regulation of cardiovascular and renal function and protection from disease and to highlight areas in which targeting of the HO system needs to be translated to help benefit patient populations. -

Relating Metatranscriptomic Profiles to the Micropollutant
1 Relating Metatranscriptomic Profiles to the 2 Micropollutant Biotransformation Potential of 3 Complex Microbial Communities 4 5 Supporting Information 6 7 Stefan Achermann,1,2 Cresten B. Mansfeldt,1 Marcel Müller,1,3 David R. Johnson,1 Kathrin 8 Fenner*,1,2,4 9 1Eawag, Swiss Federal Institute of Aquatic Science and Technology, 8600 Dübendorf, 10 Switzerland. 2Institute of Biogeochemistry and Pollutant Dynamics, ETH Zürich, 8092 11 Zürich, Switzerland. 3Institute of Atmospheric and Climate Science, ETH Zürich, 8092 12 Zürich, Switzerland. 4Department of Chemistry, University of Zürich, 8057 Zürich, 13 Switzerland. 14 *Corresponding author (email: [email protected] ) 15 S.A and C.B.M contributed equally to this work. 16 17 18 19 20 21 This supporting information (SI) is organized in 4 sections (S1-S4) with a total of 10 pages and 22 comprises 7 figures (Figure S1-S7) and 4 tables (Table S1-S4). 23 24 25 S1 26 S1 Data normalization 27 28 29 30 Figure S1. Relative fractions of gene transcripts originating from eukaryotes and bacteria. 31 32 33 Table S1. Relative standard deviation (RSD) for commonly used reference genes across all 34 samples (n=12). EC number mean fraction bacteria (%) RSD (%) RSD bacteria (%) RSD eukaryotes (%) 2.7.7.6 (RNAP) 80 16 6 nda 5.99.1.2 (DNA topoisomerase) 90 11 9 nda 5.99.1.3 (DNA gyrase) 92 16 10 nda 1.2.1.12 (GAPDH) 37 39 6 32 35 and indicates not determined. 36 37 38 39 S2 40 S2 Nitrile hydration 41 42 43 44 Figure S2: Pearson correlation coefficients r for rate constants of bromoxynil and acetamiprid with 45 gene transcripts of ECs describing nucleophilic reactions of water with nitriles. -

Flavin-Containing Monooxygenases: Regulation, Endogenous Roles and Dietary Supplements
Flavin-containing monooxygenases: Regulation, endogenous roles and dietary supplements A thesis submitted to the University of London in partial fulfilment of the requirements for the degree of Doctor of Philosophy by Lyndsey Houseman Department of Biochemistry and Molecular Biology University College London February 2008 1, Lyndsey Houseman, confirm that the work presented in this thesis is my own. Where information has been derived from other sources, I confirm that this has been indicated in the thesis. 1 UMI Number: U59122B All rights reserved INFORMATION TO ALL USERS The quality of this reproduction is dependent upon the quality of the copy submitted. In the unlikely event that the author did not send a complete manuscript and there are missing pages, these will be noted. Also, if material had to be removed, a note will indicate the deletion. Dissertation Publishing UMI U591223 Published by ProQuest LLC 2013. Copyright in the Dissertation held by the Author. Microform Edition © ProQuest LLC. All rights reserved. This work is protected against unauthorized copying under Title 17, United States Code. ProQuest LLC 789 East Eisenhower Parkway P.O. Box 1346 Ann Arbor, Ml 48106-1346 Abstract To investigate the regulation of flavin-containing monooxygenase 5 (FM05) by xenobiotics in mouse liver, a method for isolating primary mouse hepatocytes was designed. Fmo5 expression was up-regulated by hormones (estradiol, progesterone), dietary supplements (lipoic acid) and other xenobiotics (rifampicin, ethanol) in primary mouse hepatocytes and HepG2 cells. Gene reporter assays showed that Fmo5 expression in HepG2 cells is mediated by pregnane-x-receptor (PXR) ligands. To uncover more about the endogenous role of FMOs, two knockout mouse models, Fmol, 2, 4 (-/-) and Fmo5 (-/-), were studied. -

Mechanistic Study of Cysteine Dioxygenase, a Non-Heme
MECHANISTIC STUDY OF CYSTEINE DIOXYGENASE, A NON-HEME MONONUCLEAR IRON ENZYME by WEI LI Presented to the Faculty of the Graduate School of The University of Texas at Arlington in Partial Fulfillment of the Requirements for the Degree of DOCTOR OF PHILOSOPHY THE UNIVERSITY OF TEXAS AT ARLINGTON August 2014 Copyright © by Student Name Wei Li All Rights Reserved Acknowledgements I would like to thank Dr. Pierce for your mentoring, guidance and patience over the five years. I cannot go all the way through this without your help. Your intelligence and determination has been and will always be an example for me. I would like to thank my committee members Dr. Dias, Dr. Heo and Dr. Jonhson- Winters for the directions and invaluable advice. I also would like to thank all my lab mates, Josh, Bishnu ,Andra, Priyanka, Eleanor, you all helped me so I could finish my projects. I would like to thank the Department of Chemistry and Biochemistry for the help with my academic and career. At Last, I would like to thank my lovely wife and beautiful daughter who made my life meaningful and full of joy. July 11, 2014 iii Abstract MECHANISTIC STUDY OF CYSTEINE DIOXYGENASE A NON-HEME MONONUCLEAR IRON ENZYME Wei Li, PhD The University of Texas at Arlington, 2014 Supervising Professor: Brad Pierce Cysteine dioxygenase (CDO) is an non-heme mononuclear iron enzymes that catalyzes the O2-dependent oxidation of L-cysteine (Cys) to produce cysteine sulfinic acid (CSA). CDO controls cysteine levels in cells and is a potential drug target for some diseases such as Parkinson’s and Alzhermer’s. -

FMO3) As a Novel Genetic Determinant of Acetaminophen (APAP) Induced Hepatotoxicity Swetha Rudraiah University of Connecticut - Storrs, [email protected]
University of Connecticut OpenCommons@UConn Doctoral Dissertations University of Connecticut Graduate School 8-4-2014 Characterization of Flavin-containing Monooxygenase-3 (FMO3) as a Novel Genetic Determinant of Acetaminophen (APAP) Induced Hepatotoxicity Swetha Rudraiah University of Connecticut - Storrs, [email protected] Follow this and additional works at: https://opencommons.uconn.edu/dissertations Recommended Citation Rudraiah, Swetha, "Characterization of Flavin-containing Monooxygenase-3 (FMO3) as a Novel Genetic Determinant of Acetaminophen (APAP) Induced Hepatotoxicity" (2014). Doctoral Dissertations. 506. https://opencommons.uconn.edu/dissertations/506 Characterization of Flavin-containing Monooxygenase-3 (FMO3) as a Novel Genetic Determinant of Acetaminophen (APAP) Induced Hepatotoxicity Swetha Rudraiah, Ph.D. University of Connecticut, 2014 Mice pretreated with a mild toxic dose of acetaminophen (APAP) acquire resistance to a second, higher APAP dose. This phenomenon is termed APAP autoprotection and the exact mechanism by which such resistance develops is not clearly known. Given the prevalence of APAP- hepatotoxicity and the human health impact of this potentially hepatotoxic agent, a further understanding of the mechanism(s) involved in such protection are of considerable significance and could lead to new modalities of treatment of acute drug-induced liver injury. The work presented in this thesis investigates FMO3 gene expression during APAP-induced liver injury as well as the functional significance of FMO3 over-expression during APAP- induced liver injury. Furthermore, FMO3 gene regulation during oxidative stress conditions is also examined. Acetaminophen treatment resulted in up-regulation of liver Fmo3 protein in male mice. Female mice express higher liver Fmo3 than males and are highly resistant to APAP hepatotoxicity. -

Adrenocorticotropin Induces Heme Oxygenase-1 Expression in Adrenal Cells
113 Adrenocorticotropin induces heme oxygenase-1 expression in adrenal cells Y Pomeraniec, N Grion, L Gadda, V Pannunzio, E J Podesta and C B Cymeryng Departamento de Bioquí mica Humana, Facultad de Medicina, Universidad de Buenos Aires, Paraguay 2155, 5 piso, C1121 ABG, Buenos Aires, Argentina (Requests for offprints should be addressed to C B Cymeryng; Email: [email protected]) Abstract Heme oxygenase (HO) catalyzes the first and rate- and cycloheximide completely blocked the effect of controlling step of heme catabolism into biliverdin, iron ACTH on HO-1 mRNA expression whereas mRNA and carbon monoxide. Three isoforms of HO have been stability was not affected by ACTH. Permeable analogs identified so far: the inducible HO-1 and the constitutive of cAMP mimicked the effect of ACTH on HO-1 HO-2 and HO-3. Both HO-1 and HO-2 were expressed expression and ACTH induction was prevented by the in zona fasciculata (ZF) adrenal cells and in a mouse protein kinase A (PKA) inhibitor H89. Steroid production adrenocortical cell line (Y1). HO-1 but not HO-2 was significantly increased when both HO-1 and HO-2 expression was upregulated by adrenocorticotropic hor- activities were inhibited by Sn-protoporphyrin IX mone (ACTH) and accumulation of HO-1 protein corre- (SnPPIX). The lipid peroxidation and increase in carbonyl lated with an increase in HO activity in Y1 cells. ACTH content triggered by hydrogen peroxide was prevented by induced HO-1 expression in a time- and dose-dependent treatment of Y1 cells with bilirubin and ACTH. manner with a maximum after 5 h of treatment and a Journal of Endocrinology (2004) 180, 113–124 threshold concentration of 0·1 mIU/ml. -

The Blockade of Cyclooxygenases-1 and -2 Reduces the Effects of Hypoxia on Endothelial Cells
Brazilian Journal of Medical and Biological Research (2006) 39: 1189-1196 Endothelial cell activation and hypoxia 1189 ISSN 0100-879X The blockade of cyclooxygenases-1 and -2 reduces the effects of hypoxia on endothelial cells M.A. Gloria1, 1Laboratório de Imunologia Clínica e Experimental, Divisão de Nefrologia, M.A. Cenedeze1, Universidade Federal de São Paulo, Hospital do Rim e Hipertensão, A. Pacheco-Silva1 Fundação Oswaldo Ramos, São Paulo, SP, Brasil and N.O.S. Câmara1,2 2Laboratório de Imunobiologia de Transplantes, Departamento de Imunologia, Instituto de Ciências Biomédicas IV, Universidade de São Paulo, São Paulo, SP, Brasil Abstract Correspondence Hypoxia activates endothelial cells by the action of reactive oxygen Key words N.O.S. Câmara species generated in part by cyclooxygenases (COX) production • Endothelial cell Laboratório de Imunologia Clínica enhancing leukocyte transmigration. We investigated the effect of • Hypoxia e Experimental specific COX inhibition on the function of endothelial cells exposed • Indomethacin Divisão de Nefrologia, UNIFESP to hypoxia. Mouse immortalized endothelial cells were subjected to • Cyclooxygenase Rua Botucatu, 740 • Heme oxygenase 1 04023-900 São Paulo, SP 30 min of oxygen deprivation by gas exchange. Acridine orange/ Brasil ethidium bromide dyes and lactate dehydrogenase activity were used Fax: +55-11-5573-9652 to monitor cell viability. The mRNA of COX-1 and -2 was amplified E-mail: [email protected] and semi-quantified before and after hypoxia in cells treated or not with indomethacin, a non-selective COX inhibitor. Expression of Research supported by CAPES and RANTES (regulated upon activation, normal T cell expressed and FAPESP (Nos. 04/08311-6 and secreted) protein and the protective role of heme oxygenase-1 (HO-1) 04/13826-5). -

Significance of Heme and Heme Degradation in the Pathogenesis Of
International Journal of Molecular Sciences Review Significance of Heme and Heme Degradation in the Pathogenesis of Acute Lung and Inflammatory Disorders Stefan W. Ryter Proterris, Inc., Boston, MA 02118, USA; [email protected] Abstract: The heme molecule serves as an essential prosthetic group for oxygen transport and storage proteins, as well for cellular metabolic enzyme activities, including those involved in mitochondrial respiration, xenobiotic metabolism, and antioxidant responses. Dysfunction in both heme synthesis and degradation pathways can promote human disease. Heme is a pro-oxidant via iron catalysis that can induce cytotoxicity and injury to the vascular endothelium. Additionally, heme can modulate inflammatory and immune system functions. Thus, the synthesis, utilization and turnover of heme are by necessity tightly regulated. The microsomal heme oxygenase (HO) system degrades heme to carbon monoxide (CO), iron, and biliverdin-IXα, that latter which is converted to bilirubin-IXα by biliverdin reductase. Heme degradation by heme oxygenase-1 (HO-1) is linked to cytoprotection via heme removal, as well as by activity-dependent end-product generation (i.e., bile pigments and CO), and other potential mechanisms. Therapeutic strategies targeting the heme/HO-1 pathway, including therapeutic modulation of heme levels, elevation (or inhibition) of HO-1 protein and activity, and application of CO donor compounds or gas show potential in inflammatory conditions including sepsis and pulmonary diseases. Keywords: acute lung injury; carbon monoxide; heme; heme oxygenase; inflammation; lung dis- ease; sepsis Citation: Ryter, S.W. Significance of Heme and Heme Degradation in the Pathogenesis of Acute Lung and Inflammatory Disorders. Int. J. Mol. 1. Introduction Sci. -

Heme Oxygenase-1 Affects Cytochrome P450 Function Through the Formation of Heteromeric Complexes: Interactions Between CYP1A2 and Heme Oxygenase-1
bioRxiv preprint doi: https://doi.org/10.1101/2020.09.14.296467; this version posted September 14, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Heme oxygenase-1 affects cytochrome P450 function through the formation of heteromeric complexes: Interactions between CYP1A2 and heme oxygenase-1 J. Patrick Connick, James R. Reed, George F. Cawley, and Wayne L. Backes* Department of Pharmacology and Experimental Therapeutics, Louisiana State University Health Sciences Center – New Orleans, LA *Corresponding author: Wayne L. Backes E-mail: [email protected] Running title: Interactions between CYP1A2 and HO-1 Keywords: Cytochrome P450, heme oxygenase, protein-protein interaction, bioluminescence resonance energy transfer (BRET), membrane protein, structure-function, electron transfer, NADPH-cytochrome P450 reductase, CYP1A2 1 bioRxiv preprint doi: https://doi.org/10.1101/2020.09.14.296467; this version posted September 14, 2020. The copyright holder for this preprint (which was not certified by peer review) is the author/funder. All rights reserved. No reuse allowed without permission. Abstract can be “uncoupled,” leading to generation of superoxide and H2O2 (4). Heme oxygenase 1 (HO-1) and the cytochromes P450 (P450s) are endoplasmic reticulum-bound HO-1 is responsible for the first step of heme enzymes that rely on the same protein, NADPH- degradation, converting the substrate to biliverdin cytochrome P450 reductase (POR), to provide the (5,6). This enzyme protects cells from oxidative electrons necessary for substrate metabolism. damage, and is induced by agents that are related to Although the HO-1 and P450 systems are oxidative stress, including many xenobiotics, such interconnected due to their common electron donor, as aspirin, statins, niacin, etc. -

Lecture 2 Nitric Oxide Synthase and Heme Oxygenase Knockout Mice Ð What Have We Learned?
International Journal of Impotence Research (2000) 12, Suppl 3, S42±S44 ß 2000 Macmillan Publishers Ltd All rights reserved 0955-9930/00 $15.00 www.nature.com/ijir Lecture 2 Nitric oxide synthase and heme oxygenase knockout mice Ð what have we learned? AL Burnett1* 1Department of Urology, The Johns Hopkins University School of Medicine and The James Buchanan Brady Urological Institute, The Johns Hopkins Hospital, Baltimore, MD 21287, USA The seemingly unthinkable involvement of gaseous transgenic technology or as a basic means to ensure molecules in basic biochemical mechanisms of sexual reproducibility of species. Given these concerns, do physiology only a few years ago seems an almost these mutant animals possibly yield useful informa- indisputable concept within this research ®eld today. tion regarding sexual function? The answer is that Major advances have occurred in the scienti®c under- they do. Early and ongoing investigation of these standing of penile erection and ejaculation, with animals has been informative with results suggest- experimental evidence amply supporting the roles of ing standard and apparently compensatory mechan- nitric oxide (NO) and carbon monoxide (CO) in these isms in¯uencing sexual function. This brief article sexual functions. NO has now been well characterized summarizes research ®ndings associated with the as a major peripheral mediator of penile erection, sexual function of transgenic mice with deleted released by a neurological stimulus upon its synthesis NOS and HO-2 genes, which clari®es roles of these involving neuronal NO synthase (NOS) contained in genes and their chemical products in mammalian the autonomic innervation of the penis and endothe- sexual physiology.