Atrial Fibrillation
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Table 2. 2012 AGS Beers Criteria for Potentially
Table 2. 2012 AGS Beers Criteria for Potentially Inappropriate Medication Use in Older Adults Strength of Organ System/ Recommendat Quality of Recomm Therapeutic Category/Drug(s) Rationale ion Evidence endation References Anticholinergics (excludes TCAs) First-generation antihistamines Highly anticholinergic; Avoid Hydroxyzin Strong Agostini 2001 (as single agent or as part of clearance reduced with e and Boustani 2007 combination products) advanced age, and promethazi Guaiana 2010 Brompheniramine tolerance develops ne: high; Han 2001 Carbinoxamine when used as hypnotic; All others: Rudolph 2008 Chlorpheniramine increased risk of moderate Clemastine confusion, dry mouth, Cyproheptadine constipation, and other Dexbrompheniramine anticholinergic Dexchlorpheniramine effects/toxicity. Diphenhydramine (oral) Doxylamine Use of diphenhydramine in Hydroxyzine special situations such Promethazine as acute treatment of Triprolidine severe allergic reaction may be appropriate. Antiparkinson agents Not recommended for Avoid Moderate Strong Rudolph 2008 Benztropine (oral) prevention of Trihexyphenidyl extrapyramidal symptoms with antipsychotics; more effective agents available for treatment of Parkinson disease. Antispasmodics Highly anticholinergic, Avoid Moderate Strong Lechevallier- Belladonna alkaloids uncertain except in Michel 2005 Clidinium-chlordiazepoxide effectiveness. short-term Rudolph 2008 Dicyclomine palliative Hyoscyamine care to Propantheline decrease Scopolamine oral secretions. Antithrombotics Dipyridamole, oral short-acting* May -

Dofetilide (Tikosyn): a New Drug to Control Atrial Fibrillation
CURRENT DRUG THERAPY WALID I. SALIBA, MD Section of Cardiac Electrophysiology and Pacing, CREDIT Department of Cardiology, Cleveland Clinic Dofetilide (Tikosyn): A new drug to control atrial fibrillation ABSTRACT OFETILIDE (Tikosyn), a new antiarrhyth- mic drug, can convert atrial fibrillation Dofetilide, a new class III antiarrhythmic agent, selectively and atrial flutter to sinus rhythm in approxi- blocks a specific cardiac potassium channel, lKr, increasing mately 30% of cases and maintain sinus the effective refractory period of the myocyte and thereby rhythm after electrical or pharmacologic con- terminating reentrant arrhythmias. Given orally, it appears version for up to 1 year in 60% to 70% of to effectively convert atrial fibrillation and atrial flutter to cases, without increasing the risk of sudden sinus rhythm and maintain sinus rhythm after conversion in death in patients at high risk. appropriately selected patients. This paper reviews the Such new drugs are needed, as many of the pharmacology of dofetilide, the evidence of its antiarrhythmic drugs in use up to now have effectiveness, and the appropriate precautions in using it. actually produced higher mortality rates in clinical trials than did placebo, or cause unac- ceptable side effects. KEY POINTS This article reviews the mechanism of action, safety, effectiveness, and clinical use of Dofetilide is generally well tolerated but like other class III dofetilide. drugs can cause torsades de pointes. The risk is dose- dependent and can be minimized by adjusting the dosage • PROBLEMS WITH PREVIOUS DRUGS according to creatinine clearance and QT interval, by excluding patients with known risk factors for long QT A variety of drugs have been used to terminate syndrome and torsades de pointes, and by starting or prevent atrial and ventricular arrhythmias, treatment in an inpatient monitored setting for the first 3 but their safety, efficacy, and tolerability in days. -

Safety and Efficacy of Ibutilide in Cardioversion of Atrial Flutter And
J Am Board Fam Med: first published as 10.3122/jabfm.2011.01.080096 on 5 January 2011. Downloaded from CLINICAL REVIEW Safety and Efficacy of Ibutilide in Cardioversion of Atrial Flutter and Fibrillation Madhuri Nair, MD, Lekha K. George, MD, and Santhosh K. G. Koshy, MD This article reviews the safety and efficacy of ibutilide for use in patients with atrial fibrillation and flut- ter. Ibutilide, a class III antiarrhythmic agent, is primarily used for conversion of atrial flutter and fi- brillation and is a good alternative to electrical cardioversion. Ibutilide has a conversion rate of up to 75% to 80% in recent-onset atrial fibrillation and flutter; the conversion rate is higher for atrial flutter than for atrial fibrillation. It is also safe in the conversion of chronic atrial fibrillation/flutter among patients receiving oral amiodarone therapy. Ibutilide pretreatment facilitates transthoracic defibrilla- tion and decreases the energy requirement of electrical cardioversion by both monophasic and biphasic shocks. Pretreatment with ibutilide before electrical defibrillation has a conversion rate of 100% com- pared with 72% with no pretreatment. Ibutilide is also safe and efficient in the treatment of atrial fibril- lation in patients who have had cardiac surgery, and in accessory pathway–mediated atrial fibrillation where the conversion rate of ibutilide is as high as 95%. There is up to a 4% risk of torsade de pointes and a 4.9% risk of monomorphic ventricular tachycardia. Hence, close monitoring in an intensive care unit setting is warranted during and at least for 4 hours after drug infusion. The anticoagulation strat- egy is the same as for any other mode of cardioversion.(J Am Board Fam Med 2011;24:86–92.) Keywords: Antiarrhythmics, Arrhythmia, Atrial Fibrillation, Cardiovascular Disorders, Cardioversion, Drug Ther- copyright. -

Antiparasitic Properties of Cardiovascular Agents Against Human Intravascular Parasite Schistosoma Mansoni
pharmaceuticals Article Antiparasitic Properties of Cardiovascular Agents against Human Intravascular Parasite Schistosoma mansoni Raquel Porto 1, Ana C. Mengarda 1, Rayssa A. Cajas 1, Maria C. Salvadori 2 , Fernanda S. Teixeira 2 , Daniel D. R. Arcanjo 3 , Abolghasem Siyadatpanah 4, Maria de Lourdes Pereira 5 , Polrat Wilairatana 6,* and Josué de Moraes 1,* 1 Research Center for Neglected Diseases, Guarulhos University, Praça Tereza Cristina 229, São Paulo 07023-070, SP, Brazil; [email protected] (R.P.); [email protected] (A.C.M.); [email protected] (R.A.C.) 2 Institute of Physics, University of São Paulo, São Paulo 05508-060, SP, Brazil; [email protected] (M.C.S.); [email protected] (F.S.T.) 3 Department of Biophysics and Physiology, Federal University of Piaui, Teresina 64049-550, PI, Brazil; [email protected] 4 Ferdows School of Paramedical and Health, Birjand University of Medical Sciences, Birjand 9717853577, Iran; [email protected] 5 CICECO-Aveiro Institute of Materials & Department of Medical Sciences, University of Aveiro, 3810-193 Aveiro, Portugal; [email protected] 6 Department of Clinical Tropical Medicine, Faculty of Tropical Medicine, Mahidol University, Bangkok 10400, Thailand * Correspondence: [email protected] (P.W.); [email protected] (J.d.M.) Citation: Porto, R.; Mengarda, A.C.; Abstract: The intravascular parasitic worm Schistosoma mansoni is a causative agent of schistosomiasis, Cajas, R.A.; Salvadori, M.C.; Teixeira, a disease of great global public health significance. Praziquantel is the only drug available to F.S.; Arcanjo, D.D.R.; Siyadatpanah, treat schistosomiasis and there is an urgent demand for new anthelmintic agents. -
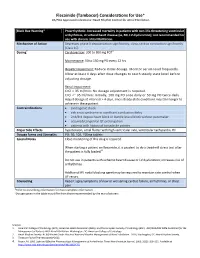
Flecainide Considerations For
Flecainide (Tambocor) Considerations for Use* US/FDA Approved Indications: Heart Rhythm Control for Atrial Fibrillation Black Box Warning* Proarrhythmic. Increased mortality in patients with non-life-threatening ventricular arrhythmias, structural heart disease (ie, MI, LV dysfunction); not recommended for use with chronic atrial fibrillation. Mechanism of Action Depresses phase 0 depolarization significantly, slows cardiac conduction significantly (Class 1C). Dosing† Cardioversion: 200 to 300 mg PO‡1 Maintenance: 50 to 150 mg PO every 12 hrs Hepatic Impairment: Reduce initial dosage. Monitor serum level frequently. Allow at least 4 days after dose changes to reach steady state level before adjusting dosage. Renal Impairment: CrCl > 35 ml/min: No dosage adjustment is required. CrCl <= 35 ml/min: Initially, 100 mg PO once daily or 50 mg PO twice daily. Adjust dosage at intervals > 4 days, since steady-state conditions may take longer to achieve in these patient Contraindications cardiogenic shock sick sinus syndrome or significant conduction delay 2nd/3rd degree heart block or bundle brand block without pacemaker acquired/congenital QT prolongation patients with history of torsade de pointes Major Side Effects hypotension, atrial flutter with high ventricular rate, ventricular tachycardia, HF Dosage forms and Strengths PO: 50, 100, 150mg tablets Special Notes Close monitoring of this drug is required. When starting a patient on flecainide, it is prudent to do a treadmill stress test after the patient is fully loaded.4 Do not use in patients with ischemic heart disease or LV dysfunction; increases risk of arrhythmias. Additional AV nodal blocking agent may be required to maintain rate control when AF recurs. -

Multaq, INN-Dronedarone
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT MULTAQ 400 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 400 mg of dronedarone (as hydrochloride). Excipient with known effect: Each tablet also contains 41.65 mg of lactose (as monohydrate). For the full list of excipients, see section 6.1. 3. PHARMACEUTICAL FORM Film-coated tablet (tablet). White, oblong shaped tablets, engraved with a double wave marking on one side and “4142”code on the other side. 4. CLINICAL PARTICULARS 4.1 Therapeutic indications MULTAQ is indicated for the maintenance of sinus rhythm after successful cardioversion in adult clinically stable patients with paroxysmal or persistent atrial fibrillation (AF). Due to its safety profile (see sections 4.3 and 4.4), MULTAQ should only be prescribed after alternative treatment options have been considered. MULTAQ must not be given to patients with left ventricular systolic dysfunction or to patients with current or previous episodes of heart failure. 4.2 Posology and method of administration Treatment should be initiated and monitored only under specialist supervision (see section 4.4). Treatment with dronedarone can be initiated in an outpatient setting. Treatment with Class I or III antiarrhythmics (such as flecainide, propafenone, quinidine, disopyramide, dofetilide, sotalol, amiodarone) must be stopped before starting dronedarone. There is limited information on the optimal timing to switch from amiodarone to dronedarone. It should be considered that amiodarone may have a long duration of action after discontinuation due to its long half-life. If a switch is envisaged, this should be done under the supervision of a specialist (see sections 4.3 and 5.1). -

A Proposal for the Clinical Use of Flecainide
A Proposal for the Clinical Use of Flecainide JEFFREY L. ANDERSON, MD, JAMES R. STEWART, MD, and BARRY J. CREVEY, MD Effective antiarrhythmic therapy requires a carefully sponse rate is observed in preventing induction of considered approach, including an understanding sustained ventricular tachycardia, and these pa- of the arrhythmia, the underlying cardiac disease tients should be carefully selected. Flecainide is and the drug’s pharmacokinetics. Flecainide is a promising in the treatment of supraventricular new antiarrhythmic drug that may soon be released tachycardias using atrioventricular nodal or extra- for general use. Flecainide demonstrates unsur- nodal reentrant pathways, although this use is still passed efficacy in chronic ventricular arrhythmias investigational in the United States. The drug’s use in stable patients and may become a first-choice for arrhythmias during acute myocardial infarction drug because of its ease of administration, efficacy requires further study. Flecainide possesses modest and favorable tolerance. Twice-daily dosing with negative inotropic potential. Proarrhythmic or other 100 to 200 mg usually provides effective therapy. adverse reactions have occurred primarily in set- Clinical experience suggests flecainide to be indi- tings of high drug level, poor ventricular function cated in the treatment of uniform and multiform or refractory, malignant arrhythmias, suggesting ventricular premature complexes, coupled ven- caution in these groups. tricular premature complexes, and episodes of nonsustained -

Abstract Materials and Methods Summary
A human ex-vivo contractility-based assay for the simultaneous prediction of drug-induced inotropic and pro-arrhythmia risk Najah Abi-Gerges, Ashley Alamillo, Phachareeya Ratchada, Guy Page, Yannick Miron, Nathalie Nguyen, Paul E Miller and Andre Ghetti AnaBios Corp., San Diego, CA 92109, USA Contact email: [email protected] Abstract Human ventricular trabeculae-based model predicts Human ventricular trabeculae-based model the torsadogenic potential of dofetilide and d,l-sotalol predicts safety of verapamil Cardiac safety remains the leading cause of drug development discontinuation and withdrawal of post marketing approvals. This has called into question the reliability of current preclinical safety- testing paradigms, which rely predominantly on animal models, and has led to demands for more predictive tools. To this aim, we sought to develop and validate a new ex-vivo human-based model that uses human ventricular trabeculae, paced under tension and Figure 3. Effects of verapamil on Fc parameters in human ex-vivo ventricular combined with continuous recordings of the exerted force of trabeculae. Verapamil decreased the force contraction (Fc). We were especially interested in establishing if of contraction with an IC50 value of 1.46µM, while it had no effect on TR70 and did not this strategy could provide a more predictive approach for induce any ACs or triggered contractions when tested up to 222-fold its fETPC. n=7 assessing both inotropic activity as well as pro-arrhythmia risk. trabeculae. Note that the negative inotropic Since the T-wave recorded in ECG in the clinics marks ventricular effect of verapamil is in line with its known 2+ repolarization and is therefore an electrical measure of the inhibition of Ca channel). -

Guideline for Preoperative Medication Management
Guideline: Preoperative Medication Management Guideline for Preoperative Medication Management Purpose of Guideline: To provide guidance to physicians, advanced practice providers (APPs), pharmacists, and nurses regarding medication management in the preoperative setting. Background: Appropriate perioperative medication management is essential to ensure positive surgical outcomes and prevent medication misadventures.1 Results from a prospective analysis of 1,025 patients admitted to a general surgical unit concluded that patients on at least one medication for a chronic disease are 2.7 times more likely to experience surgical complications compared with those not taking any medications. As the aging population requires more medication use and the availability of various nonprescription medications continues to increase, so does the risk of polypharmacy and the need for perioperative medication guidance.2 There are no well-designed trials to support evidence-based recommendations for perioperative medication management; however, general principles and best practice approaches are available. General considerations for perioperative medication management include a thorough medication history, understanding of the medication pharmacokinetics and potential for withdrawal symptoms, understanding the risks associated with the surgical procedure and the risks of medication discontinuation based on the intended indication. Clinical judgement must be exercised, especially if medication pharmacokinetics are not predictable or there are significant risks associated with inappropriate medication withdrawal (eg, tolerance) or continuation (eg, postsurgical infection).2 Clinical Assessment: Prior to instructing the patient on preoperative medication management, completion of a thorough medication history is recommended – including all information on prescription medications, over-the-counter medications, “as needed” medications, vitamins, supplements, and herbal medications. Allergies should also be verified and documented. -

Drug Interaction of Amiodarone, Flecainide, Metoprolol and Diltiazem Leading to Heart Block Vijay Kahajuria, Sanjeev Gupta, Roshi, Neelam Rani
JK SCIENCE CASE REPORT Drug Interaction of Amiodarone, Flecainide, Metoprolol and Diltiazem leading to Heart Block Vijay Kahajuria, Sanjeev Gupta, Roshi, Neelam Rani Abstract Amiodarone, flecainide, metoprolol and diltiazem individually are known to cause heart blocks due to their cardiac depressant property but the current case report is worth reporting because it resulted because of drug interaction of multiple drugs due to possible medication error. Key Words Amiodarone, Flecainide, Metoprolol, Diltiazem, Heart Blocks Introduction Adverse Drug Reactions (ADRs) in cardiology are thyroid dysfunction. His hemoglobin levels were 13g/dl, common. There are numerous reports of drug induced total leucocyte count 6000/UL, neutrophils 56.5%, bradycardia and heart block (1). Co-morbid conditions lymphocytes 30.5%, eosinophils 5.2%, monocytes often coexist with cardiac ailments and results in 7.6%,basophils 0.2%, platelet count 1.5 lacs, serum urea- polypharmacy which enhance the chances of drug 33mg/dl, serum creatinine 0.90mg/dl, prothrombin time interactions culminating to adverse events. In the current 10.9 sec ,INR 1.04,hepatitis serology was non reactive, study report patient was prescribed multiple drugs for blood sugar random 182 mg/dl, TSH 1.4uIU/ml, serum cardiac arrhythmia and he presented with heart block sodium 142 mmol/L,serum potassium 5 mmol/L. and bradycardia. Though amiodarone, flecainide, He was prescribed tab.atenolol 20mg and metoprolol and diltiazem individually are known to cause tab.procainamide 20mg which did not bring any relief. heart blocks due to their cardiac depressant property but So radiofrequency ablation was done which was the current case report is worth reporting because it uneventful. -

Flecainide in Ventricular Arrhythmias: from Old Myths to New Perspectives
Journal of Clinical Medicine Review Flecainide in Ventricular Arrhythmias: From Old Myths to New Perspectives Carlo Lavalle 1,*, Sara Trivigno 1 , Giampaolo Vetta 1 , Michele Magnocavallo 1 , Marco Valerio Mariani 1 , Luca Santini 2, Giovanni Battista Forleo 3 , Massimo Grimaldi 4, Roberto Badagliacca 1, Luigi Lanata 5 and Renato Pietro Ricci 6 1 Department of Cardiovascular, Respiratory, Nephrological, Anesthesiological and Geriatric Sciences, Sapienza University of Rome, Policlinico Umberto I, 00161 Rome, Italy; [email protected] (S.T.); [email protected] (G.V.); [email protected] (M.M.); [email protected] (M.V.M.); [email protected] (R.B.) 2 Department of Cardiology, Ospedale GB Grassi, 00121 Ostia, Italy; [email protected] 3 Department of Cardiology, Azienda Ospedaliera-Universitaria “Luigi Sacco”, 20057 Milan, Italy; [email protected] 4 Department of Cardiology, Ospedale Generale Regionale F. Miulli, Acquaviva delle Fonti, 70021 Bari, Italy; fi[email protected] 5 Medical Affairs Department, Dompé Farmaceutici SpA, 20057 Milan, Italy; [email protected] 6 Centro Cardio-Aritmologico, 00152 Rome, Italy; [email protected] * Correspondence: [email protected]; Tel.: +39-335376901 Abstract: Flecainide is an IC antiarrhythmic drug (AAD) that received in 1984 Food and Drug Administration approval for the treatment of sustained ventricular tachycardia (VT) and subsequently Citation: Lavalle, C.; Trivigno, S.; for rhythm control of atrial fibrillation (AF). Currently, flecainide is mainly employed for sinus Vetta, G.; Magnocavallo, M.; Mariani, rhythm maintenance in AF and the treatment of idiopathic ventricular arrhythmias (IVA) in absence M.V.; Santini, L.; Forleo, G.B.; Grimaldi, M.; Badagliacca, R.; Lanata, of ischaemic and structural heart disease on the basis of CAST data. -

EGSC Drug and Alcohol Policy
EGSC Student Handbook, “Institutional Policy” section Revisions adopted by the President’s Cabinet 9/22/15 Approved by President’s Cabinet 9-23-14 Drugs and Alcohol President Bush’s National Drug Control Strategy issued in September of 1989 proposed that the Congress pass legislation to require schools, colleges, and universities to implement and enforce firm drug prevention programs and policies. On December 12, 1989, the President signed the Drug-Free Schools and Communities Act Amendments (Public Law 101-226). This law establishes a process to accomplish the President’s goal. Public Law 101-226 requires that schools maintain standards of conduct that clearly prohibit, at a minimum, the unlawful possession, use, or distribution of drugs and alcohol on school property or as any part of school activities. In support of Public Law 101-226, East Georgia State College recognizes and supports local, state, and federal laws with respect to the sale, use, distribution, and possession of alcoholic beverages and illegal drugs. To this end, the possession, consumption, distribution, manufacture, or sale (without valid medical or dental prescription) of alcoholic beverages, illegal or dangerous drugs on East Georgia State College property or at institutionally-approved events off campus is prohibited. East Georgia State College also adheres to the following: Drug Free School Zone – it is unlawful for persons to manufacture, distribute, dispense, or possess with intent to distribute illegal drugs within 1,000 feet of any elementary or secondary school property. A first conviction is punishable by imprisonment for not more than 20 years and/or a fine of not more than $20,000, and a second or subsequent conviction is punishable by imprisonment for at least five years but not more than 40 years and/or a fine of not more than $40,000.