Hyperintense Punctiform Images in the White Matter: a Diagnostic Approachଝ
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Headache in Multiple Sclerosis from a Different Perspective: a Prospective Study
Volume 9 • Number 1 • March 2018 JOURNAL OF CLINICAL AND RESEARCHORIGINAL ARTICLEARTICLE EXPERIMENTAL INVESTIGATIONS Headache in Multiple Sclerosis from A Different Perspective: A Prospective Study Gökhan Özer¹, Ufuk Ergün², Levent Ertuğrul İnan³ 1 Sanko University Faculty of Medicine, Department of Neurology, Gaziantep, ABSTRACT Turkey Objective: It is known that patients with multiple sclerosis have a high incidence of headache. 2 Kırıkkale University School of Medicine, Kırıkkale, Turkey Although there is increasing evidence to suggest that periaqueductal gray matter (PAG) plays 3 Bozok University School of Medicine, a role in the pathophysiology of migraine headache, it is not known whether the type of Yozgat, Turkey headache may be a predictor of a MS relapse. Patients and Methods: The study enrolled 100 patients (68 females, 32 males) with clinically confirmed MS diagnosis established by McDonald diagnostic criteria. The type and duration of MS, MRI localization of lesions and cognitive status were recorded for all patients. Patients were questioned whether they experience headache during MS attacks. Results: Sixty-eight percent of the patients had headache and 32% of the patients were free of headache. Of the patients with headache, 16% had tension –type headache (TTH), 14% had migraine, 11% had primary stabbing headache (PSH), 8% had TTH+ migraine, 6% had PSH+ migraine, 6% had medication overuse headache , 2% had medication overuse headache + E-mail: [email protected] migraine, 2% had paroxysmal hemicrania, 1% had cervicogenic headache, 1% had chronic TTH, E-mail: [email protected] and 1% had unclassified headache. There was a statistically significant relationship between the E-mail: [email protected] presence of headache and MS relapse (p<0.001). -

Vaccination and Demyelination: Is There a Link? Examples with Anti
Vaccination and demyelination : Is there a link? Examples with anti-hepatitis B and papillomavirus vaccines Julie Mouchet Le Moal To cite this version: Julie Mouchet Le Moal. Vaccination and demyelination : Is there a link? Examples with anti-hepatitis B and papillomavirus vaccines. Human health and pathology. Université de Bordeaux, 2019. English. NNT : 2019BORD0015. tel-02134582 HAL Id: tel-02134582 https://tel.archives-ouvertes.fr/tel-02134582 Submitted on 20 May 2019 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. THÈSE PRÉSENTÉE POUR OBTENIR LE GRADE DE DOCTEUR DE L’UNIVERSITÉ DE BORDEAUX ÉCOLE DOCTORALE : Sociétés, Politique, Santé Publique (SP2) SPÉCIALITÉ : Pharmacologie option Pharmaco-épidémiologie, Pharmacovigilance Par Julie MOUCHET LE MOAL VACCINATION ET RISQUE DE DEMYELINISATION : EXISTE-T-IL UN LIEN ? EXEMPLES DES VACCINS ANTI-HEPATITE B ET ANTI-PAPILLOMAVIRUS Sous la direction de : Monsieur le Professeur Bernard Bégaud Soutenue publiquement le 29 Janvier 2019 Composition du jury Président : Christophe TZOURIO, Professeur des Universités -

Multiple Sclerosis Accepted: 15 May 2016 Receiving Tysabri
Iranian Journal Letter to Editor of Neurology Iran J Neurol 2016; 15(3): 175-176 Bacterial meningitis in a patient Received: 10 Mar 2016 with multiple sclerosis Accepted: 15 May 2016 receiving Tysabri Abdorreza Naser Moghadasi1, Soroor Advani2, Shiva Rahimi2 1 Multiple Sclerosis Research Center, Neuroscience Institute, Department of Neurology, Sina Hospital, Tehran University of Medical Sciences, Tehran, Iran 2 Department of Neurology, Sina Hospital, Tehran University of Medical Sciences, Tehran, Iran Keywords lesions (Figure 1-A) with no evidence of PML or Bacterial Meningitis; Multiple Sclerosis; Tysabri HSE encephalitis. Meningeal enhancement was seen after the injection of the contrast medium (Figure 1-B). The most important adverse effect of natalizumab is progressive multifocal leukoencephalopathy (PML).1 Apart from PML, there are reports of other cerebral infections including herpes simplex encephalitis (HSE)2,3 and cryptococcal meningitis4 in the literature. The patient was a 29-year-old woman, a known case of multiple sclerosis (MS) for at least 5 years. She was treated using natalizumab since 6 month before. She was under treatment with prednisolone 1 g daily for 5 days for optic neuritis, which was 2 weeks before the onset of symptoms of meningitis. Approximately three days before visiting the neurologist, a continuous headache in the left temporal lobe was developed. Figure 1. Periventricular lesions confirming the The patient was febrile at that time as well. diagnosis of multiple sclerosis (MS) in the FLAIR MRI Besides, two days before this, she had started view (A). Meningeal enhancement was seen after ciprofloxacin for treatment of a urinary gadolinium injection (B) tract infection. -

Clinical and MRI Clues and Pitfalls in the Diagnosis and Differential Diagnosis of Multiple Sclerosis
Clinical and MRI clues and pitfalls in the diagnosis and differential diagnosis of Multiple Sclerosis Aksel Siva, M.D. MS Clinic & Department Of Neurology Istanbul University Cerrahpaşa School of Medicine [email protected] MSParis2017 - 7th Joint ECTRIMS - ACTRIMS Meeting, 25 - 28 October 2017 Disclosure • Received research grants to my department from The Scientific and Technological Research Council Of Turkey - Health Sciences Research Grants numbers : 109S070 and 112S052.; and also unrestricted research grants from Merck-Serono and Novartis to our Clinical Neuroimmunology Unit • Honoraria or consultation fees and/or travel and registration coverage for attending several national or international congresses or symposia, from Merck Serono, Biogen Idec/Gen Pharma of Turkey, Novartis, Genzyme, Roche and Teva. • Educational presentations at programmes & symposia prepared by Excemed internationally and at national meetings and symposia sponsored by Bayer- Schering AG; Merck-Serono;. Novartis, Genzyme and Teva-Turkey; Biogen Idec/Gen Pharma of Turkey Introduction… • The incidence and prevalence rates of MS are increasing, so are the number of misdiagnosed cases as MS! • One major source of misdiagnosis is misinterpretation of nonspecific clinical and imaging findings and misapplication of MRI diagnostic criteria resulting in an overdiagnosis of MS! • The differential diagnosis of MS includes the MS spectrum and related disorders that covers subclinical & clinical MS phenotypes, MS variants and inflammatory astrocytopathies, as well as other Ab-associated atypical inflammatory-demyelinating syndromes • There are a number of systemic diseases in which either the clinical or MRI findings or both may mimic MS, which further cause confusion! Related publication *Siva A. Common Clinical and Imaging Conditions Misdiagnosed as Multiple Sclerosis. -

Serial Magnetic Resonance Imaging Changes of Pseudotumor Lesions In
Yan et al. BMC Neurol (2021) 21:219 https://doi.org/10.1186/s12883-021-02250-4 CASE REPORT Open Access Serial magnetic resonance imaging changes of pseudotumor lesions in retinal vasculopathy with cerebral leukoencephalopathy and systemic manifestations: a case report Yuying Yan1, Shuai Jiang1, Ruilin Wang2, Xiang Wang3, Peng Li3* and Bo Wu1* Abstract Background: Retinal vasculopathy with cerebral leukoencephalopathy and systemic manifestations (RVCL-S) is an adult-onset rare monogenic microvasculopathy. Its typical neuroimaging features are punctate white matter lesions or pseudotumor alterations. RVCL-S is often under-recognized and misdiagnosed because of its rarity and similar imaging manifestations to multiple sclerosis or brain malignant mass. Case presentation: Here we report a case of a 36-year-old Chinese man who developed multiple tumefactive brain lesions spanning over two years leading to motor aphasia, cognitive decline, and limb weakness. He also presented with slight vision loss, and fundus fuorescein angiography indicated retinal vasculopathy. He underwent brain biopsies twice and showed no evidence of malignancy. Given the family history that his father died of a brain mass of unclear etiology, RVCL-S was suspected, and genetic analysis confrmed the diagnosis with a heterozygous insertion mutation in the three-prime repair exonuclease 1 gene. He was given courses of corticosteroids and cyclophospha- mide but received little response. Conclusions: The present case is one of the few published reports of RVCL-S with two-year detailed imaging data. Serial magnetic resonance images showed the progression pattern of the lesions. Our experience emphasizes that a better understanding of RVCL-S and considering it as a diferential diagnosis in patients with tumefactive brain lesions may help avoid unnecessary invasive examinations and make an earlier diagnosis. -

MR Images, Brain Lesions, and Deep Learning
applied sciences Review MR Images, Brain Lesions, and Deep Learning Darwin Castillo 1,2,3,* , Vasudevan Lakshminarayanan 2,4 and María José Rodríguez-Álvarez 3 1 Departamento de Química y Ciencias Exactas, Sección Fisicoquímica y Matemáticas, Universidad Técnica Particular de Loja, San Cayetano Alto s/n, Loja 11-01-608, Ecuador 2 Theoretical and Experimental Epistemology Lab, School of Optometry and Vision Science, University of Waterloo, Waterloo, ON N2L3G1, Canada; [email protected] 3 Instituto de Instrumentación para Imagen Molecular (i3M) Universitat Politècnica de València—Consejo Superior de Investigaciones Científicas (CSIC), E-46022 Valencia, Spain; [email protected] 4 Departments of Physics, Electrical and Computer Engineering and Systems Design Engineering, University of Waterloo, Waterloo, ON N2L3G1, Canada * Correspondence: [email protected]; Tel.: +593-07-370-1444 (ext. 3204) Featured Application: This review provides a critical review of deep/machine learning algo- rithms used in the identification of ischemic stroke and demyelinating brain diseases. It eval- uates their strengths and weaknesses when applied to real world clinical data. Abstract: Medical brain image analysis is a necessary step in computer-assisted/computer-aided diagnosis (CAD) systems. Advancements in both hardware and software in the past few years have led to improved segmentation and classification of various diseases. In the present work, we review the published literature on systems and algorithms that allow for classification, identification, and detection of white matter hyperintensities (WMHs) of brain magnetic resonance (MR) images, specifically in cases of ischemic stroke and demyelinating diseases. For the selection criteria, we used bibliometric networks. Of a total of 140 documents, we selected 38 articles that deal with the main objectives of this study. -

Radiologically Isolated Syndrome: a Review for Neuroradiologists
REVIEW ARTICLE Radiologically Isolated Syndrome: A Review for Neuroradiologists M. Hosseiny, S.D. Newsome, and D.M. Yousem ABSTRACT SUMMARY: Radiologically isolated syndrome refers to an entity in which white matter lesions fulfilling the criteria for multiple sclerosis occur in individuals without a history of a clinical demyelinating attack or alternative etiology. Since its introduction in 2009, the diagnostic criteria of radiologically isolated syndrome and its clinical relevance have been widely debated by neu- rologists and radiologists. The aim of the present study was to review the following: 1) historical evolution of radiologically iso- lated syndrome criteria, 2) clinical and imaging findings in adults and children with radiologically isolated syndrome, 3) imaging features of patients with radiologically isolated syndrome at high risk for conversion to MS, and 4) challenges and controversies for work-up, management, and therapeutic interventions of patients with radiologically isolated syndrome. ABBREVIATIONS: CIS ¼ clinically isolated syndrome; DIS ¼ dissemination in space; DIT ¼ dissemination in time; MAGNIMS ¼ Magnetic Resonance Imaging in MS; RIS ¼ radiologically isolated syndrome; RRMS ¼ relapsing-remitting multiple sclerosis; CADASIL ¼ cerebral autosomal dominant arteriopathy with sub- cortical infarcts and leukoencephalopathy; ADEM ¼ acute disseminated encephalomyelitis adiologically isolated syndrome (RIS) refers to an entity in patients are being evaluated for headache, trauma, or nonspecific Rwhich brain or spine MR -

Demyelination: What Is It and Why Does It Happen?
Demyelination: What Is It and Why Does It Happen? • Causes • Symptoms • Types • MS and Demyelination • Treatment and Diagnosis • Vaccines • Takeaway What is demyelination? Nerves send and receive messages from every part of your body and process them in your brain. Nerves allow you to speak, see, feel, and think. Many nerves are coated in myelin. Myelin is an insulating material. When it’s worn away or damaged, nerves can deteriorate, causing problems in the brain and throughout the body. Damage to myelin around nerves is called demyelination. Nerves Nerves are made up of neurons. Neurons are composed of a cell body, dendrites, and an axon. The axon sends messages from one neuron to the next. The axon also connects neurons to other cells, such as muscle cells. Some axons are extremely short. Others are 3 feet long. Some axons are covered in myelin. Myelin protects the axons and helps carry axon messages as quickly as possible. Myelin Myelin is made of membrane layers that cover an axon. This is similar to the idea of an electrical wire with coating to protect the metal underneath. Myelin allows a nerve signal to travel faster. In unmyelinated neurons, a signal can travel along the nerves at about 1 meter per second. In a myelinated neuron, the signal can travel 100 meters per second. Certain diseases can damage myelin. Demyelination slows down messages sent along axons and causes the axon to deteriorate. Depending upon the location of the damage, axon loss can cause problems with feeling, moving, seeing, hearing, and thinking clearly. CAUSES Causes of demyelination Inflammation is the most common cause of myelin damage. -

Cerebrospinal Fluid Analysis in Multiple Sclerosis Diagnosis: an Update
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Archivio istituzionale della ricerca - Università di Palermo medicina Review Cerebrospinal Fluid Analysis in Multiple Sclerosis Diagnosis: An Update Bruna Lo Sasso 1, Luisa Agnello 1, Giulia Bivona 1 , Chiara Bellia 1 and Marcello Ciaccio 1,2,* 1 Institute of Clinical Biochemistry, Clinical Molecular Medicine and Laboratory Medicine, Department of Biomedicine, Neuroscience and Advanced Diagnostics, University of Palermo, 90100 Palermo, Italy; [email protected] (B.L.S.); [email protected] (L.A.); [email protected] (G.B.); [email protected] (C.B.) 2 Department Laboratory Medicine, University-Hospital, 90100 Palermo, Italy * Correspondence: [email protected]; Tel.: +39-091-23865701; Fax: +39-091-655-3275 Received: 25 March 2019; Accepted: 30 May 2019; Published: 4 June 2019 Abstract: Multiple sclerosis (MS) is an immune-mediated demyelinating disease of the central nervous system (CNS) with brain neurodegeneration. MS patients present heterogeneous clinical manifestations in which both genetic and environmental factors are involved. The diagnosis is very complex due to the high heterogeneity of the pathophysiology of the disease. The diagnostic criteria have been modified several times over the years. Basically, they include clinical symptoms, presence of typical lesions detected by magnetic resonance imaging (MRI), and laboratory findings. The analysis of cerebrospinal fluid (CSF) allows an evaluation of inflammatory processes circumscribed to the CNS and reflects changes in the immunological pattern due to the progression of the pathology, being fundamental in the diagnosis and monitoring of MS. The detection of the oligoclonal bands (OCBs) in both CSF and serum is recognized as the “gold standard” for laboratory diagnosis of MS, though presents analytical limitations. -

Acute Disseminated Encephalomyelitis Or Multiple Sclerosis: Can the Initial Presentation Help in Establishing a Correct Diagnosi
636 REVIEW Acute disseminated encephalomyelitis or multiple sclerosis: can the initial presentation help in establishing a correct Arch Dis Child: first published as on 20 May 2005. Downloaded from diagnosis? R C Dale, J A Branson ............................................................................................................................... Arch Dis Child 2005;90:636–639. doi: 10.1136/adc.2004.062935 The differential diagnosis of CNS white matter disease is and MS cases must manifest disseminated disease of the CNS (more than one clinical or broad, and can be divided into vascular, metabolic, radiological site). Diseases isolated to specific infective, or inflammatory aetiologies. Isolated areas of the CNS (isolated optic neuritis, trans- inflammatory disorders of the CNS are often associated verse myelitis, and brain stem dysfunction) are considered distinct from both ADEM and MS with demyelination, and the two terms (inflammatory and (clinically and prognostically), and will not be demyelinating) are often used in conjunction. When the discussed in this review.89 disease is monophasic, the term acute disseminated 1 DEMOGRAPHICS encephalomyelitis (ADEM) is used. ADEM typically occurs Monophasic ADEM is more common in children, as a post-infectious phenomenon, and by definition, must whereas MS is more common in adults. Between be an isolated (monophasic) episode. If a relapse occurs 2.7% and 4.4% of MS presentations occur in children less than 16 years of age.8 Mikaeloff et al shortly after the ADEM presentation in association with a showed a mean age of 7.1 years and 12.0 years in further infection or steroid withdrawal, the term MDEM paediatric ADEM and MS patients respectively.8 (multiphasic disseminated encephalomyelitis) is used. -

A Diffusion Tensor Magnetic Resonance Imaging Study of Brain Tissue from Patients with Migraine M a Rocca, B Colombo, M Inglese, M Codella, G Comi, M Filippi
501 J Neurol Neurosurg Psychiatry: first published as 10.1136/jnnp.74.4.501 on 1 April 2003. Downloaded from SHORT REPORT A diffusion tensor magnetic resonance imaging study of brain tissue from patients with migraine M A Rocca, B Colombo, M Inglese, M Codella, G Comi, M Filippi ............................................................................................................................. J Neurol Neurosurg Psychiatry 2003;74:501–503 PATIENTS AND METHODS Objective: To measure in vivo, using diffusion tensor mag- We studied 34 patients (31 women and three men; mean age netic resonance imaging (DT-MRI) the extent of pathologi- 35.2 years, range 18–55 years; mean duration of the disease cal damage of normal appearing brain tissue (NABT) from 13.6 years, range 1–40 years; mean number of episodes per patients with migraine. year 44.6, range 12–70) with migraine.10 All possible alterna- Methods: Dual echo and DT-MRI scans of the brain were tive diagnoses were carefully excluded.10 There were 28 acquired from 34 patients with migraine and 17 sex and patients affected by “migraine without aura” and six patients age matched healthy volunteers. Mean diffusivity (MD) affected by “migraine with aura”.10 Patients with hyper- and fractional anisotropy (FA) histograms of the NABT tension, hypercholesteraemia, diabetes mellitus, vascular/ were obtained from all subjects and the histograms’ peak heart diseases, and other major systemic and neurological heights and average NABT MD and FA measured. When conditions were excluded. At the time of the MRI assessment, present, average MD and FA values of T2 visible lesions 11 patients were treated with prophylactic drugs. Seventeen were also measured. -
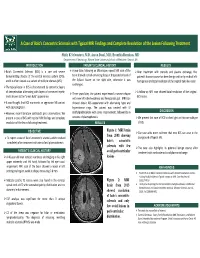
A Case of Balo's Concentric Sclerosis with Typical MRI Findings and Complete Resolution of the Lesion Followi
A Case of Balo’s Concentric Sclerosis with Typical MRI Findings and Complete Resolution of the Lesion Following Treatment Shitiz K Sriwastava, M.D, Aaron Desai, M.D, Evanthia Bernitsas, MD Department of Neurology, Wayne State University School of Medicine, Detroit, MI INTRODUCTION PATEINT’S CLINCIAL HISTORY RESULTS Balo’s Concentric Sclerosis (BCS) is a rare and severe A year later, following an MS relapse, repeat MRI scan of the After treatment with steroids and plasma exchange, the demyelinating disease of the central nervous system (CNS), brain showed a small enhancing focus at the posterior level of patient’s disease course has been benign with only residual left and it is often viewed as a variant of multiple sclerosis (MS). the Sylvian fissure on the right side, otherwise it was hemiparesis and total resolution of the original Balo‐like lesion. unchanged. The typical lesion in BCS is characterized by concentric layers of demyelination alternating with layers of preserved myelin A follow‐up MRI scan showed total resolution of the original Three years later, the patient experienced a severe relapse and is known as the “onion bulb” appearance. BCS lesion. with new left‐sided weakness and hemiparetic gait. MRI scan It was thought that BCS represents an aggressive MS variant showed classic BCS appearance with alternating hypo and with poor prognosis. hyperintense rings. The patient was treated with IV DISCUSSION However, recent literature contradicts prior observations. We methylprednisolone with some improvement, followed by 6 present a case of BCS with typical MRI findings and complete sessions of plasmapheresis. We present this case of BCS to shed light on this rare subtype resolution of the lesion following treatment.