Carbon Assimilation Strategies in Ultrabasic Groundwater: Clues from the Integrated
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
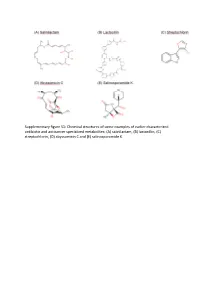
Chemical Structures of Some Examples of Earlier Characterized Antibiotic and Anticancer Specialized
Supplementary figure S1: Chemical structures of some examples of earlier characterized antibiotic and anticancer specialized metabolites: (A) salinilactam, (B) lactocillin, (C) streptochlorin, (D) abyssomicin C and (E) salinosporamide K. Figure S2. Heat map representing hierarchical classification of the SMGCs detected in all the metagenomes in the dataset. Table S1: The sampling locations of each of the sites in the dataset. Sample Sample Bio-project Site depth accession accession Samples Latitude Longitude Site description (m) number in SRA number in SRA AT0050m01B1-4C1 SRS598124 PRJNA193416 Atlantis II water column 50, 200, Water column AT0200m01C1-4D1 SRS598125 21°36'19.0" 38°12'09.0 700 and above the brine N "E (ATII 50, ATII 200, 1500 pool water layers AT0700m01C1-3D1 SRS598128 ATII 700, ATII 1500) AT1500m01B1-3C1 SRS598129 ATBRUCL SRS1029632 PRJNA193416 Atlantis II brine 21°36'19.0" 38°12'09.0 1996– Brine pool water ATBRLCL1-3 SRS1029579 (ATII UCL, ATII INF, N "E 2025 layers ATII LCL) ATBRINP SRS481323 PRJNA219363 ATIID-1a SRS1120041 PRJNA299097 ATIID-1b SRS1120130 ATIID-2 SRS1120133 2168 + Sea sediments Atlantis II - sediments 21°36'19.0" 38°12'09.0 ~3.5 core underlying ATII ATIID-3 SRS1120134 (ATII SDM) N "E length brine pool ATIID-4 SRS1120135 ATIID-5 SRS1120142 ATIID-6 SRS1120143 Discovery Deep brine DDBRINP SRS481325 PRJNA219363 21°17'11.0" 38°17'14.0 2026– Brine pool water N "E 2042 layers (DD INF, DD BR) DDBRINE DD-1 SRS1120158 PRJNA299097 DD-2 SRS1120203 DD-3 SRS1120205 Discovery Deep 2180 + Sea sediments sediments 21°17'11.0" -

Genomics 98 (2011) 370–375
Genomics 98 (2011) 370–375 Contents lists available at ScienceDirect Genomics journal homepage: www.elsevier.com/locate/ygeno Whole-genome comparison clarifies close phylogenetic relationships between the phyla Dictyoglomi and Thermotogae Hiromi Nishida a,⁎, Teruhiko Beppu b, Kenji Ueda b a Agricultural Bioinformatics Research Unit, Graduate School of Agricultural and Life Sciences, University of Tokyo, 1-1-1 Yayoi, Bunkyo-ku, Tokyo 113-8657, Japan b Life Science Research Center, College of Bioresource Sciences, Nihon University, Fujisawa, Japan article info abstract Article history: The anaerobic thermophilic bacterial genus Dictyoglomus is characterized by the ability to produce useful Received 2 June 2011 enzymes such as amylase, mannanase, and xylanase. Despite the significance, the phylogenetic position of Accepted 1 August 2011 Dictyoglomus has not yet been clarified, since it exhibits ambiguous phylogenetic positions in a single gene Available online 7 August 2011 sequence comparison-based analysis. The number of substitutions at the diverging point of Dictyoglomus is insufficient to show the relationships in a single gene comparison-based analysis. Hence, we studied its Keywords: evolutionary trait based on whole-genome comparison. Both gene content and orthologous protein sequence Whole-genome comparison Dictyoglomus comparisons indicated that Dictyoglomus is most closely related to the phylum Thermotogae and it forms a Bacterial systematics monophyletic group with Coprothermobacter proteolyticus (a constituent of the phylum Firmicutes) and Coprothermobacter proteolyticus Thermotogae. Our findings indicate that C. proteolyticus does not belong to the phylum Firmicutes and that the Thermotogae phylum Dictyoglomi is not closely related to either the phylum Firmicutes or Synergistetes but to the phylum Thermotogae. © 2011 Elsevier Inc. -

Diversity of Understudied Archaeal and Bacterial Populations of Yellowstone National Park: from Genes to Genomes Daniel Colman
University of New Mexico UNM Digital Repository Biology ETDs Electronic Theses and Dissertations 7-1-2015 Diversity of understudied archaeal and bacterial populations of Yellowstone National Park: from genes to genomes Daniel Colman Follow this and additional works at: https://digitalrepository.unm.edu/biol_etds Recommended Citation Colman, Daniel. "Diversity of understudied archaeal and bacterial populations of Yellowstone National Park: from genes to genomes." (2015). https://digitalrepository.unm.edu/biol_etds/18 This Dissertation is brought to you for free and open access by the Electronic Theses and Dissertations at UNM Digital Repository. It has been accepted for inclusion in Biology ETDs by an authorized administrator of UNM Digital Repository. For more information, please contact [email protected]. Daniel Robert Colman Candidate Biology Department This dissertation is approved, and it is acceptable in quality and form for publication: Approved by the Dissertation Committee: Cristina Takacs-Vesbach , Chairperson Robert Sinsabaugh Laura Crossey Diana Northup i Diversity of understudied archaeal and bacterial populations from Yellowstone National Park: from genes to genomes by Daniel Robert Colman B.S. Biology, University of New Mexico, 2009 DISSERTATION Submitted in Partial Fulfillment of the Requirements for the Degree of Doctor of Philosophy Biology The University of New Mexico Albuquerque, New Mexico July 2015 ii DEDICATION I would like to dedicate this dissertation to my late grandfather, Kenneth Leo Colman, associate professor of Animal Science in the Wool laboratory at Montana State University, who even very near the end of his earthly tenure, thought it pertinent to quiz my knowledge of oxidized nitrogen compounds. He was a man of great curiosity about the natural world, and to whom I owe an acknowledgement for his legacy of intellectual (and actual) wanderlust. -

Thermaerobacter Marianensis Type Strain (7P75a)
Lawrence Berkeley National Laboratory Recent Work Title Complete genome sequence of Thermaerobacter marianensis type strain (7p75a). Permalink https://escholarship.org/uc/item/2km2g1rj Journal Standards in genomic sciences, 3(3) ISSN 1944-3277 Authors Han, Cliff Gu, Wei Zhang, Xiaojing et al. Publication Date 2010-12-15 DOI 10.4056/sigs.1373474 Peer reviewed eScholarship.org Powered by the California Digital Library University of California Standards in Genomic Sciences (2010) 3:337-345 DOI:10.4056/sigs.1373474 Complete genome sequence of Thermaerobacter T marianensis type strain (7p75a ) Cliff Han1,2, Wei Gu1,2, Xiaojing Zhang1,2, Alla Lapidus1, Matt Nolan1, Alex Copeland1, Susan Lucas1, Tijana Glavina Del Rio1, Hope Tice1, Jan-Fang Cheng1, Roxane Tapia1,2, Lynne Goodwin1,2, Sam Pitluck1, Ioanna Pagani1, Natalia Ivanova1, Konstantinos Mavromatis1, Natalia Mikhailova1, Amrita Pati1, Amy Chen3, Krishna Palaniappan3, Miriam Land1,4, Loren Hauser1,4, Yun-Juan Chang1,4, Cynthia D. Jeffries1,4, Susanne Schneider5, Manfred Rohde6, Markus Göker5, Rüdiger Pukall5, Tanja Woyke1, James Bristow1, Jonathan A. Eisen1,7, Victor Markowitz3, Philip Hugenholtz1, Nikos C. Kyrpides1, Hans-Peter Klenk5*, and John C. Detter1,2 1 DOE Joint Genome Institute, Walnut Creek, California, USA 2 Los Alamos National Laboratory, Bioscience Division, Los Alamos, New Mexico, USA 3 Biological Data Management and Technology Center, Lawrence Berkeley National Laboratory, Berkeley, California, USA 4 Oak Ridge National Laboratory, Oak Ridge, Tennessee, USA 5 DSMZ - German Collection of Microorganisms and Cell Cultures GmbH, Braunschweig, Germany 6 HZI – Helmholtz Centre for Infection Research, Braunschweig, Germany 7 University of California Davis Genome Center, Davis, California, USA *Corresponding author: Hans-Peter Klenk Keywords: strictly aerobic, none-motile, Gram-variable, thermophilic, chemoheterotrophic, deep-sea, family Incertae Sedis XVII, Clostridiales, GEBA Thermaerobacter marianensis Takai et al. -

Sporulation Evolution and Specialization in Bacillus
bioRxiv preprint doi: https://doi.org/10.1101/473793; this version posted March 11, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC 4.0 International license. Research article From root to tips: sporulation evolution and specialization in Bacillus subtilis and the intestinal pathogen Clostridioides difficile Paula Ramos-Silva1*, Mónica Serrano2, Adriano O. Henriques2 1Instituto Gulbenkian de Ciência, Oeiras, Portugal 2Instituto de Tecnologia Química e Biológica, Universidade Nova de Lisboa, Oeiras, Portugal *Corresponding author: Present address: Naturalis Biodiversity Center, Marine Biodiversity, Leiden, The Netherlands Phone: 0031 717519283 Email: [email protected] (Paula Ramos-Silva) Running title: Sporulation from root to tips Keywords: sporulation, bacterial genome evolution, horizontal gene transfer, taxon- specific genes, Bacillus subtilis, Clostridioides difficile 1 bioRxiv preprint doi: https://doi.org/10.1101/473793; this version posted March 11, 2019. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted bioRxiv a license to display the preprint in perpetuity. It is made available under aCC-BY-NC 4.0 International license. Abstract Bacteria of the Firmicutes phylum are able to enter a developmental pathway that culminates with the formation of a highly resistant, dormant spore. Spores allow environmental persistence, dissemination and for pathogens, are infection vehicles. In both the model Bacillus subtilis, an aerobic species, and in the intestinal pathogen Clostridioides difficile, an obligate anaerobe, sporulation mobilizes hundreds of genes. -

Proquest Dissertations
Approaches to assessing microbial communities in soil, two examples: Biosurfactant production and phenanthrene degradation Item Type text; Dissertation-Reproduction (electronic) Authors Bodour, Adria Publisher The University of Arizona. Rights Copyright © is held by the author. Digital access to this material is made possible by the University Libraries, University of Arizona. Further transmission, reproduction or presentation (such as public display or performance) of protected items is prohibited except with permission of the author. Download date 01/10/2021 06:55:39 Link to Item http://hdl.handle.net/10150/280136 INFORMATION TO USERS This manuscript has been reproduced from the microfiinn master. UMI films the text directly from the original or copy submitted. Thus, some thesis and dissertation copies are in typewriter face, while others may be from any type of computer printer. The quality of this reproduction is dependent upon the quality of the copy submitted. Broken or indistinct print, colored or poor quality illustrations and photographs, print bleedthrough, substandard margins, and improper alignment can adversely affect reproduction. In the unlikely event that the author did not send UMI a complete manuscript and there are missing pages, these will be noted. Also, if unauthorized copyright material had to be removed, a note will indicate the deletion. Oversize materials (e.g., maps, drawings, charts) are reproduced by sectioning the original, beginning at the upper left-hand corner and continuing from left to right in equal -

Phenotypic and Microbial Influences on Dairy Heifer Fertility and Calf Gut Microbial Development
Phenotypic and microbial influences on dairy heifer fertility and calf gut microbial development Connor E. Owens Dissertation submitted to the faculty of the Virginia Polytechnic Institute and State University in partial fulfillment of the requirements for the degree of Doctor of Philosophy In Animal Science, Dairy Rebecca R. Cockrum Kristy M. Daniels Alan Ealy Katharine F. Knowlton September 17, 2020 Blacksburg, VA Keywords: microbiome, fertility, inoculation Phenotypic and microbial influences on dairy heifer fertility and calf gut microbial development Connor E. Owens ABSTRACT (Academic) Pregnancy loss and calf death can cost dairy producers more than $230 million annually. While methods involving nutrition, climate, and health management to mitigate pregnancy loss and calf death have been developed, one potential influence that has not been well examined is the reproductive microbiome. I hypothesized that the microbiome of the reproductive tract would influence heifer fertility and calf gut microbial development. The objectives of this dissertation were: 1) to examine differences in phenotypes related to reproductive physiology in virgin Holstein heifers based on outcome of first insemination, 2) to characterize the uterine microbiome of virgin Holstein heifers before insemination and examine associations between uterine microbial composition and fertility related phenotypes, insemination outcome, and season of breeding, and 3) to characterize the various maternal and calf fecal microbiomes and predicted metagenomes during peri-partum and post-partum periods and examine the influence of the maternal microbiome on calf gut development during the pre-weaning phase. In the first experiment, virgin Holstein heifers (n = 52) were enrolled over 12 periods, on period per month. On -3 d before insemination, heifers were weighed and the uterus was flushed. -

Crystalline Iron Oxides Stimulate Methanogenic Benzoate Degradation in Marine Sediment-Derived Enrichment Cultures
The ISME Journal https://doi.org/10.1038/s41396-020-00824-7 ARTICLE Crystalline iron oxides stimulate methanogenic benzoate degradation in marine sediment-derived enrichment cultures 1,2 1 3 1,2 1 David A. Aromokeye ● Oluwatobi E. Oni ● Jan Tebben ● Xiuran Yin ● Tim Richter-Heitmann ● 2,4 1 5 5 1 2,3 Jenny Wendt ● Rolf Nimzyk ● Sten Littmann ● Daniela Tienken ● Ajinkya C. Kulkarni ● Susann Henkel ● 2,4 2,4 1,3 2,3,4 1,2 Kai-Uwe Hinrichs ● Marcus Elvert ● Tilmann Harder ● Sabine Kasten ● Michael W. Friedrich Received: 19 May 2020 / Revised: 9 October 2020 / Accepted: 22 October 2020 © The Author(s) 2020. This article is published with open access Abstract Elevated dissolved iron concentrations in the methanic zone are typical geochemical signatures of rapidly accumulating marine sediments. These sediments are often characterized by co-burial of iron oxides with recalcitrant aromatic organic matter of terrigenous origin. Thus far, iron oxides are predicted to either impede organic matter degradation, aiding its preservation, or identified to enhance organic carbon oxidation via direct electron transfer. Here, we investigated the effect of various iron oxide phases with differing crystallinity (magnetite, hematite, and lepidocrocite) during microbial 1234567890();,: 1234567890();,: degradation of the aromatic model compound benzoate in methanic sediments. In slurry incubations with magnetite or hematite, concurrent iron reduction, and methanogenesis were stimulated during accelerated benzoate degradation with methanogenesis as the dominant electron sink. In contrast, with lepidocrocite, benzoate degradation, and methanogenesis were inhibited. These observations were reproducible in sediment-free enrichments, even after five successive transfers. Genes involved in the complete degradation of benzoate were identified in multiple metagenome assembled genomes. -

Which Organisms Are Used for Anti-Biofouling Studies
Table S1. Semi-systematic review raw data answering: Which organisms are used for anti-biofouling studies? Antifoulant Method Organism(s) Model Bacteria Type of Biofilm Source (Y if mentioned) Detection Method composite membranes E. coli ATCC25922 Y LIVE/DEAD baclight [1] stain S. aureus ATCC255923 composite membranes E. coli ATCC25922 Y colony counting [2] S. aureus RSKK 1009 graphene oxide Saccharomycetes colony counting [3] methyl p-hydroxybenzoate L. monocytogenes [4] potassium sorbate P. putida Y. enterocolitica A. hydrophila composite membranes E. coli Y FESEM [5] (unspecified/unique sample type) S. aureus (unspecified/unique sample type) K. pneumonia ATCC13883 P. aeruginosa BAA-1744 composite membranes E. coli Y SEM [6] (unspecified/unique sample type) S. aureus (unspecified/unique sample type) graphene oxide E. coli ATCC25922 Y colony counting [7] S. aureus ATCC9144 P. aeruginosa ATCCPAO1 composite membranes E. coli Y measuring flux [8] (unspecified/unique sample type) graphene oxide E. coli Y colony counting [9] (unspecified/unique SEM sample type) LIVE/DEAD baclight S. aureus stain (unspecified/unique sample type) modified membrane P. aeruginosa P60 Y DAPI [10] Bacillus sp. G-84 LIVE/DEAD baclight stain bacteriophages E. coli (K12) Y measuring flux [11] ATCC11303-B4 quorum quenching P. aeruginosa KCTC LIVE/DEAD baclight [12] 2513 stain modified membrane E. coli colony counting [13] (unspecified/unique colony counting sample type) measuring flux S. aureus (unspecified/unique sample type) modified membrane E. coli BW26437 Y measuring flux [14] graphene oxide Klebsiella colony counting [15] (unspecified/unique sample type) P. aeruginosa (unspecified/unique sample type) graphene oxide P. aeruginosa measuring flux [16] (unspecified/unique sample type) composite membranes E. -

The Keystone Species of Precambrian Deep Bedrock Biosphere the Supplement Related to This Article Is Available Online at Doi:10.5194/Bgd-12-18103-2015-Supplement
Discussion Paper | Discussion Paper | Discussion Paper | Discussion Paper | Biogeosciences Discuss., 12, 18103–18150, 2015 www.biogeosciences-discuss.net/12/18103/2015/ doi:10.5194/bgd-12-18103-2015 BGD © Author(s) 2015. CC Attribution 3.0 License. 12, 18103–18150, 2015 This discussion paper is/has been under review for the journal Biogeosciences (BG). The keystone species Please refer to the corresponding final paper in BG if available. of Precambrian deep bedrock biosphere The keystone species of Precambrian L. Purkamo et al. deep bedrock biosphere belong to Burkholderiales and Clostridiales Title Page Abstract Introduction L. Purkamo1, M. Bomberg1, R. Kietäväinen2, H. Salavirta1, M. Nyyssönen1, M. Nuppunen-Puputti1, L. Ahonen2, I. Kukkonen2,a, and M. Itävaara1 Conclusions References Tables Figures 1VTT Technical Research Centre of Finland Ltd., Espoo, Finland 2Geological Survey of Finland (GTK), Espoo, Finland anow at: University of Helsinki, Helsinki, Finland J I Received: 13 September 2015 – Accepted: 23 October 2015 – Published: 11 November 2015 J I Correspondence to: L. Purkamo (lotta.purkamo@vtt.fi) Back Close Published by Copernicus Publications on behalf of the European Geosciences Union. Full Screen / Esc Printer-friendly Version Interactive Discussion 18103 Discussion Paper | Discussion Paper | Discussion Paper | Discussion Paper | Abstract BGD The bacterial and archaeal community composition and the possible carbon assimi- lation processes and energy sources of microbial communities in oligotrophic, deep, 12, 18103–18150, 2015 crystalline bedrock fractures is yet to be resolved. In this study, intrinsic microbial com- 5 munities from six fracture zones from 180–2300 m depths in Outokumpu bedrock were The keystone species characterized using high-throughput amplicon sequencing and metagenomic predic- of Precambrian deep tion. -

Afipia Clevelandensis Sp
JOURNAL OF CLINICAL MICROBIOLOGY, Nov. 1991, p. 2450-2460 Vol. 29, No. 11 0095-1137/91/112450-11$02.00/0 Copyright © 1991, American Society for Microbiology Proposal of Afipia gen. nov., with Afipia felis sp. nov. (Formerly the Cat Scratch Disease Bacillus), Afipia clevelandensis sp. nov. (Formerly the Cleveland Clinic Foundation Strain), Afipia broomeae sp. nov., and Three Unnamed Genospecies DON J. BRENNER,'* DANNIE G. HOLLIS,' C. WAYNE MOSS,' CHARLES K. ENGLISH,2 GERALDINE S. HALL,3 JUDY VINCENT,4 JON RADOSEVIC,5 KRISTIN A. BIRKNESS,1 WILLIAM F. BIBB,' FREDERICK D. QUINN,' B. SWAMINATHAN,1 ROBERT E. WEAVER,' MICHAEL W. REEVES,' STEVEN P. O'CONNOR,6 PEGGY S. HAYES,' FRED C. TENOVER,7 ARNOLD G. STEIGERWALT,' BRADLEY A. PERKINS,' MARYAM I. DANESHVAR,l BERTHA C. HILL,7 JOHN A. WASHINGTON,3 TONI C. WOODS,' SUSAN B. HUNTER,' TED L. HADFIELD,2 GLORIA W. AJELLO,1 ARNOLD F. KAUFMANN,8 DOUGLAS J. WEAR,2 AND JAY D. WENGER' Meningitis and Special Pathogens Branch,' Respiratory Diseases Branch,6 and Bacterial Zoonoses Activity,8 Division of Bacterial and Mycotic Diseases, and Hospital Infections Program,7 Center for Infectious Diseases, Centers for Disease Control, Atlanta, Georgia 30333; Department ofInfectious and Parasitic Diseases Pathology, Armed Forces Institute ofPathology, Washington, DC 20306-60002; Department of Microbiology, Cleveland Clinic Foundation, Cleveland, Ohio 441953; Department ofPediatrics, Tripler Army Medical Center, Tripler AMC, Hawaii 968594; and Indiana State Board of Health, Disease Control Laboratory Division, Indianapolis, Indiana 46206-19645 Received 3 June 1991/Accepted 5 August 1991 On the basis of phenotypic characterization and DNA relatedness determinations, the genus Afipia gen. -

Draft Genome Sequence of Dethiobacter Alkaliphilus Strain AHT1T, a Gram-Positive Sulfidogenic Polyextremophile Emily Denise Melton1, Dimitry Y
Melton et al. Standards in Genomic Sciences (2017) 12:57 DOI 10.1186/s40793-017-0268-9 EXTENDED GENOME REPORT Open Access Draft genome sequence of Dethiobacter alkaliphilus strain AHT1T, a gram-positive sulfidogenic polyextremophile Emily Denise Melton1, Dimitry Y. Sorokin2,3, Lex Overmars1, Alla L. Lapidus4, Manoj Pillay6, Natalia Ivanova5, Tijana Glavina del Rio5, Nikos C. Kyrpides5,6,7, Tanja Woyke5 and Gerard Muyzer1* Abstract Dethiobacter alkaliphilus strain AHT1T is an anaerobic, sulfidogenic, moderately salt-tolerant alkaliphilic chemolithotroph isolated from hypersaline soda lake sediments in northeastern Mongolia. It is a Gram-positive bacterium with low GC content, within the phylum Firmicutes. Here we report its draft genome sequence, which consists of 34 contigs with a total sequence length of 3.12 Mbp. D. alkaliphilus strain AHT1T was sequenced by the Joint Genome Institute (JGI) as part of the Community Science Program due to its relevance to bioremediation and biotechnological applications. Keywords: Extreme environment, Soda lake, Sediment, Haloalkaliphilic, Gram-positive, Firmicutes Introduction genome of a haloalkaliphilic Gram-positive sulfur dispro- Soda lakes are formed in environments where high rates portionator within the phylum Firmicutes: Dethiobacter of evaporation lead to the accumulation of soluble carbon- alkaliphilus AHT1T. ate salts due to the lack of dissolved divalent cations. Con- sequently, soda lakes are defined by their high salinity and Organism information stable highly alkaline pH conditions, making them dually Classification and features extreme environments. Soda lakes occur throughout the The haloalkaliphilic anaerobe D. alkaliphilus AHT1T American, European, African, Asian and Australian conti- was isolated from hypersaline soda lake sediments in nents and host a wide variety of Archaea and Bacteria, northeastern Mongolia [10].