Oxidative Stability, Characterization and Food Applications of a Rice Bran Oil-Based Structured Lipid
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

AN-Iodine-Value.Pdf
MANTECH INC. 160 Southgate Drive, Unit 2 Guelph, Ontario, Canada N1G 4P5 P: (519) 763-4245 www.mantech-inc.com Iodine Value Introduction: Iodine value refers to the percentage of iodine absorbed by a substance such as a fat or oil 1. It is an indication of a fat’s potential to be oxidized as it measures the reaction of iodine with the double bonds of unsaturated fatty acids 2. Since fats with a greater number of double bonds provide more sites for oxidation, the iodine value of a fat helps predict fat stability 2. Normally, iodine is absorbed quite slowly but this method makes use of Wijs, which is a stable solution consisting of iodine monochloride (ICl) in acetic acid that reduces absorption time to approximately half an hour. The iodine monochloride halogenates the double bonds in the fat, and the residual ICl is reduced to free iodine in the presence of potassium iodide (KI) 3. The free iodine can then be titrated with sodium thiosulfate. The reaction is as follows: ICl + H2O → HIO + HCl residual iodine water hypoiodus hydrochloric monochloride acid acid - + HIO + I + H → H 2O + I 2 hypoiodus iodide water iodine acid 2- - 2- I2 + 2S 2O3 → 2I + S 4O6 iodine thiosulfate iodide tetrathionate This document will show that the PC-Titrate automated titration system is capable of accurately and precisely measuring the % Iodine in various fatty samples. Please contact your local Man-Tech sales representative to find out how this method can work for you. Conforms To: AOAC Official Method 993.2, AOCS Tg 1a-64, ASTM method D1959-97 Sample: Fats and Oils Concentration Range: Tested between 0.02 and 15% Apparatus: 1. -
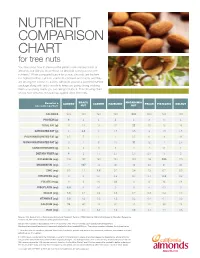
Nutrient Comparison Chart
NUTRIENT COMPARISON CHART for tree nuts You may know how to measure the perfect one-ounce portion of almonds, but did you know those 23 almonds come packed with nutrients? When compared ounce for ounce, almonds are the tree nut highest in fiber, calcium, vitamin E, riboflavin and niacin, and they are among the lowest in calories. Almonds provide a powerful nutrient package along with tasty crunch to keep you going strong, making them a satisfying snack you can feel good about. The following chart shows how almonds measure up against other tree nuts. BRAZIL MACADAMIA Based on a ALMOND CASHEW HAZELNUT PECAN PISTACHIO WALNUT one-ounce portion1 NUT NUT CALORIES 1602 190 160 180 200 200 160 190 PROTEIN (g) 6 4 4 4 2 3 6 4 TOTAL FAT (g) 14 19 13 17 22 20 13 19 SATURATED FAT (g) 1 4.5 3 1.5 3.5 2 1.5 1.5 POLYUNSATURATED FAT (g) 3.5 7 2 2 0.5 6 4 13 MONOUNSATURATED FAT (g) 9 7 8 13 17 12 7 2.5 CARBOHYDRATES (g) 6 3 9 5 4 4 8 4 DIETARY FIBER (g) 4 2 1.5 2.5 2.5 2.5 3 2 POTASSIUM (mg) 208 187 160 193 103 116 285 125 MAGNESIUM (mg) 77 107 74 46 33 34 31 45 ZINC (mg) 0.9 1.2 1.6 0.7 0.4 1.3 0.7 0.9 VITAMIN B6 (mg) 0 0 0.1 0.2 0.1 0.1 0.3 0.2 FOLATE (mcg) 12 6 20 32 3 6 14 28 RIBOFLAVIN (mg) 0.3 0 0.1 0 0 0 0.1 0 NIACIN (mg) 1.0 0.1 0.4 0.5 0.7 0.3 0.4 0.3 VITAMIN E (mg) 7.3 1.6 0.3 4.3 0.2 0.4 0.7 0.2 CALCIUM (mg) 76 45 13 32 20 20 30 28 IRON (mg) 1.1 0.7 1.7 1.3 0.8 0.7 1.1 0.8 Source: U.S. -

International Journal for Scientific Research & Development| Vol. 7, Issue 03, 2019 | ISSN (Online): 2321-0613
IJSRD - International Journal for Scientific Research & Development| Vol. 7, Issue 03, 2019 | ISSN (online): 2321-0613 Significance of Ethanol Blended Biodiesel Powered Diesel Engine Aditya Deshmukh1 Shrikrushna Dhakne2 Tohid Pathan3 Rahul Dimbar4 C. Srinidhi5 1,2,3,4UG Students 5Assistant Professor 1,2,3,4,5Department of Mechanical Engineering 1,2,3,4,5Suman Ramesh Tulsiani Tehnical Campus Faculty of Engineering Khamshet Pune, Maharashtra, India Abstract— In recent years, energy consumption is increasing engines. This chemical treatment is known as due to increase in the demand for the energy due to transesterification. Transesterification involves breaking of industrialization and increase in number of automobiles. This large triglycerides into a smaller Monoalkyl ester. The High leads to depletion of fossil fuels and hence researchers Viscosity of triglycerides can be reduced by the concentrate on alternative fuels which reduce the engine transesterification process in which triglyceride decompose emission and are environmentally friendly. This leads to into glycerol and glycerin, which is the biodiesel. The biodiesel produced from non-edible as an immediate biodiesel can be produced from a variety of edible and substitute to the fossil diesel. In this work, we used Kenaf oil nonedible oils, animal fats etc. The chosen oil will be for the production of the biodiesel as it has considerable subjected to transesterification in the existence of catalyst to potential for the production of the biodiesel. In this work, produce biodiesel. biodiesel was produced from the Kenaf oil using Indian consumption of petroleum fuels for the transesterification and we found that the properties of the period of 2008-09 was 30.50 million tons. -

Palm Oil and Rice Bran Oil: Current Status and Future Prospects
International Journal of Plant Physiology and Biochemistry Vol. 3(8), pp. 125-132, August 2011 Available online at http://www.academicjournals.org/ijppb ISSN-2141-2162 ©2011 Academic Journals Review Palm oil and rice bran oil: Current status and future prospects Kusum R., Bommayya H., Fayaz Pasha P. and Ramachandran H. D.* Department of Biochemistry, Dr. Ambedkar Veedhi Bangalore University, Bangalore - 560001, India. Accepted 17 June, 2011 The continued demand for edible oils by the ever increasing population makes it pertinent to explore new sources. In this direction, two new edible oils namely palm oil and rice bran oil have been subjected to nutritional and toxicological evaluations of their chemicals constituents. An attempt has been made in this article to assess the acceptability of the two oils based on the various investigations that have been carried out so far. Key words: Palm oil, rice bran oil, anti-oxidants, cholesterol fatty acids, phospholipids, tocopherols, oryzanol, cardiovascular diseases. INTRODUCTION Vegetable oils are the main source of dietary fat for Among the oils under consideration, palm oil and rice almost all sections of the Indian population and there is a bran oil offer great scope in India, as they are widely continued growing demand from both caterers and con- preferred by the vanaspathi industries and also by the sumers. Although the Indian population has a penchant Indian consumer. The oil palm gives higher yields in for a variety of deep fried products, there is also a greater comparison with other oil yielding species. Rice bran oil awareness of the problems such as atherosclerosis also offers high potential, as India has high rice caused by saturated fats. -

Olive Oil Jars Left Behind By
live oil jars left behind by the ancient Greeks are testament to our centuries- old use of cooking oil. Along with salt and pepper, oil Oremains one of the most important and versatile tools in your kitchen. It keeps food from sticking to pans, adds flavor and moisture, and conducts the heat that turns a humble stick of potato into a glorious french fry. Like butter and other fats, cooking oil also acts as a powerful solvent, unleashing fat-soluble nutrients and flavor compounds in everything from tomatoes and onions to spices and herbs. It’s why so many strike recipes begin with heating garlic in oil rather than, say, simmering it in water. The ancient Greeks didn’t tap many cooking oils. (Let’s see: olive oil, olive oil, or—ooh, this is exciting!—how about olive oil?) But you certainly can. From canola to safflower to grapeseed to walnut, each oil has its own unique flavor (or lack thereof), aroma, and optimal cooking temperature. Choosing the right kind for the task at hand can save you money, boost your health, and improve your cooking. OK, so you probably don’t stop to consider your cooking oil very often. But there’s a surprising amount to learn about What’s this? this liquid gold. BY VIRGINIAWILLIS Pumpkin seed oil suspended in corn oil—it looks like a homemade Lava Lamp! 84 allrecipes.com PHOTOS BY KATE SEARS WHERE TO store CANOLA OIL GRAPESEED OIL are more likely to exhibit the characteristic YOUR OIL flavor and aroma of their base nut or seed. -
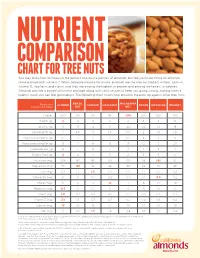
Chart for Tree Nuts
NUTRIENT COMPARISON CHART FOR TREE NUTS You may know how to measure the perfect one-ounce portion of almonds, but did you know those 23 almonds come packed with nutrients? When compared ounce for ounce, almonds are the tree nut highest in fiber, calcium, vitamin E, riboflavin and niacin, and they are among the highest in protein and among the lowest in calories. Almonds provide a powerful nutrient package along with tasty crunch to keep you going strong, making them a healthy snack you can feel good about. The following chart shows how almonds measure up against other tree nuts. BRAZIL MACADAMIA Based on a ALMOND CASHEW HAZELNUT PECAN PISTACHIO WALNUT one-ounce portion1 NUT NUT Calories 1602 190 160 180 200 200 160 190 Protein (g) 6 4 4 4 2 3 6 4 Total Fat (g) 14 19 13 17 22 20 13 18 Saturated Fat (g) 1 4.5 3 1.5 3.5 2 1.5 1.5 Polyunsaturated Fat (g) 3.5 7 2 2 0.5 6 4 13 Monounsaturated Fat (g) 9 7 8 13 17 12 7 2.5 Carbohydrates (g) 6 3 9 5 4 4 8 4 Dietary Fiber (g) 4 2 1 3 2 3 3 2 Potassium (mg) 208 187 160 193 103 116 285 125 Magnesium (mg) 77 107 74 46 33 34 31 45 Zinc (mg) 0.9 1.2 1.6 0.7 0.4 1.3 0.7 0.9 Vitamin B6 (mg) 0 0 0.1 0.2 0.1 0.1 0.3 0.2 Folate (mcg) 12 6 20 32 3 6 14 28 Riboflavin (mg) 0.3 0 0.1 0 0 0 0.1 0 Niacin (mg) 1.0 0.1 0.4 0.5 0.7 0.3 0.4 0.3 Vitamin E (mg) 7.3 1.6 0.3 4.3 0.2 0.4 0.6 0.2 Calcium (mg) 76 45 13 32 20 20 30 28 Iron (mg) 1.1 0.7 1.7 1.3 0.8 0.7 1.1 0.8 Source: U.S. -

Cooking Oil Facts
Cooking Oil Facts As you enter a department store, you behold an array of cooking oils sporting all types of jargon on the packaging -- saturated fats, unsaturated fats, refined, filtered, ricebran oil, vanaspati, etc. Confused already? With so much variety and so many brands flooding the market today, buying the right cooking oil can prove a tough task. Different oils fill different needs - for health, taste and cooking. For good health, our bodies need a variety of healthy fats that are found naturally in different oils. When cooking, it's essential to know which oils are best for baking, sautéing and frying and which are healthiest used raw. Why have Oil (fats)? Contrary to popular belief, fat is actually a valuable part of one's diet, allowing people to absorb nutrients that require fat in order to metabolize in the body. Natural fats contain varying ratios of three types of fats: saturated, monounsaturated and polyunsaturated. • Saturated fats are hard at room temperature. They're stable, resist oxidation, and are found primarily in meat, dairy, palm and coconut oil. • Polyunsaturated fats are liquid at room temperature and the least stable. They oxidize easily and are found in seafood corn, safflower, soybean, and sunflower oils. • Monounsaturated fats are more stable than polyunsaturated fats. They're found in canola, nut and olive oils. It is recommended to limit saturated fats in the diet due to their association with cardiovascular disease. Also, you should try to rely more on monounsaturated than polyunsaturated fats. What are the varieties of Oil available in the market? Choosing which oil should be used in cooking is a big issue and concern for many people because of the fat and cholesterol contents of cooking oil. -

Nutrition Handout N09: What Are the Types of Fat?
N09 What Are the Types of Fat? Most foods contain several different kinds of fat. Some are better for your health than others. It is wise to choose healthier types of fat, and enjoy them in moderation. Keep in mind that even healthier fats contain calories and should be used sparingly for weight management. Here is some information about healthy and harmful dietary fats. The four major types of fats are: • Monounsaturated fats • Polyunsaturated fats • Saturated fats • Trans fats Monounsaturated and polyunsaturated fats are known as “healthy fats” because they are good for your heart, cholesterol levels, and overall health. These fats tend to be “liquid” at room temperature. Consider beneficial polyunsaturated fats containing Omega-3 fatty acids found in fatty fish, flaxseed, and walnuts. www.move.va.gov Nutrition Handouts • N09 Version 5.0 Page 1 of 3 Healthy Dietary Fats Monounsaturated Fat Polyunsaturated Fat Olive oil Soybean oil Canola oil Corn oil Sunflower oil Safflower oil Peanut oil Walnuts Olives Sunflower, sesame, and pumpkin seeds; flaxseed Nuts (almonds, peanuts, hazelnuts, Fatty fish (salmon, tuna, mackerel, herring, macadamia nuts, pecans, cashews) trout, anchovies, sardines, and eel) Avocados Soymilk Peanut butter Tofu Tips for increasing healthy fats in your diet: • Cook with olive oil. • Plan snacks of nuts or olives. • Eat more avocados. • Dress your own salads instead of using commercial dressings. Saturated fats and trans fats are known as the “harmful fats.” They increase your risk of disease and elevate cholesterol. Saturated fats tend to be solid at room temperature, but they are also found in liquid tropical oils (palm and coconut). -

Edible Oils As Practical Phase Change Materials for Thermal Energy Storage
applied sciences Article Edible Oils as Practical Phase Change Materials for Thermal Energy Storage Samer Kahwaji 1 and Mary Anne White 1,2,* 1 Department of Chemistry, Dalhousie University, Halifax, B3H 4R2, Canada; [email protected] 2 Clean Technologies Research Institute, Dalhousie University, Halifax, B3H 4R2, Canada * Correspondence: [email protected] Received: 21 February 2019; Accepted: 17 April 2019; Published: 19 April 2019 Featured Application: Passive thermal management of residential greenhouses. Abstract: Edible oils could provide more accessible alternatives to other phase change materials (PCMs) for consumers who wish to build a thermal energy storage (TES) system with sustainable materials. Edible oils have good shelf life, can be acquired easily from local stores and can be less expensive than other PCMs. In this work, we explore whether margarine, vegetable shortening, and coconut oil are feasible PCMs, by investigations of their thermal properties and thermal stability. We found that margarine and vegetable shortening are not useful for TES due to their low latent heat of fusion, DfusH, and poor thermal stability. In contrast, coconut oil remained thermally stable after 200 melt-freeze cycles, and has a large D H of 105 11 J g 1, a low degree of supercooling fus ± − and a transition temperature, Tmpt = 24.5 1.5 C, that makes it very useful for TES in buildings. ± ◦ We also determined coconut oil’s heat capacity and thermal conductivity as functions of temperature and used the measured properties to evaluate the feasibility of coconut oil for thermal buffering and passive heating of a residential-scale greenhouse. Keywords: phase change material (PCM); passive thermal management; thermal properties; coconut oil; margarine 1. -

Fatty Acids: Essential…Therapeutic
Volume 3, No.2 May/June 2000 A CONCISE UPDATE OF IMPORTANT ISSUES CONCERNING NATURAL HEALTH INGREDIENTS Written and Edited By: Thomas G. Guilliams Ph.D. FATTY ACIDS: Essential...Therapeutic Few things have been as confusing to both patient and health care provider as the issue of fats and oils. Of all the essential nutrients required for optimal health, fatty acids have not only been forgotten they have been considered hazardous. Health has somehow been equated with “low-fat” or “fat-free” for so long, to suggest that fats could be essential or even therapeutic is to risk credibility. We hope to give a view of fats that is both balanced and scientific. This review will cover the basics of most fats that will be encountered in dietary or supplemental protocols. Recommendations to view essential fatty acids in a similar fashion as essential vitamins and minerals will be combined with therapeutic protocols for conditions ranging from cardiovascular disease, skin conditions, diabetes, nerve related disorders, retinal disorders and more. A complete restoration of health cannot be accomplished until there is a restoration of fatty acid nutritional information among health care professionals and their patients. Fats- What are they? Dietary fats come to us from a variety of sources, but primarily in the form of triglycerides. That is, three fatty acid molecules connected by a glycerol backbone (see fatty acid primer page 3 for diagram). These fatty acids are then used as energy by our cells or modified into phospholipids to be used as cell or organelle membranes. Some fatty acids are used in lipoprotein molecules to shuttle cholesterol and fats to and from cells, and fats may also be stored for later use. -

Six Features of RICE BRAN OIL 4
Six Features of RICE BRAN OIL 4. Excellent 25 Oxidation Stability 20 1. Excellent Taste Palatability In AOM test, time-peroxide value Nutritionally, it has been recognized that RICE BRAN OIL easily drops itself from of 100 of oil at 98°C was the lowest foodstuff when cooked; hence it is most suitable edible oil for frying and deep-frying to 15 oxide safety level where the flavor (hours) provide crispy texture of fried foods with better palatability. of oil started to turn bad. Higher time-peroxide value means longer 10 The fatty acid composition including linoleic acid and oleic acid, especially 40~50% of maintenance of good flavor. As Time AOM oleic acid may be felt in good taste when it is use as salad oil. Meanwhile, soybean oil shown below, RICE BRAN OIL and which contains high content of unsaturated fatty acids may increase oxidation and Corn oil both exhibited a relatively 5 rancidity of oil. high stability against oxidation upon comparison with other edible 0 oils. RICE soybean canola oil corn oil rapeseed safflower BRAN OIL oil oil oil 2. Deep-fry without 30 5. Light Oil Viscosity, 30 Oily and Lingering Less Oil Absorbed 25 25 Upon deep-frying, moisture when Cooking oil) ) evaporates forming bubbles m 20 causing uneven food surface. RICE BRAN OIL is excellent for 20 After heating the oil for 21hour at frying due to its light viscosity and (mg/500g 15 180°C, 1cm3 of potato was less oil being absorbed during 15 thrown into the oil and extent of cooking. -

Relationship Between Dietary Intake of Fatty Acids and Disease Activity in Pediatric Inflammatory Bowel Disease Patients
Relationship between Dietary Intake of Fatty Acids and Disease Activity in Pediatric Inflammatory Bowel Disease Patients A thesis submitted to the Graduate School of the University of Cincinnati in partial fulfillment of the requirements for the degree of Master of Science in the Department of Nutrition of the College of Allied Health Sciences by Michael R. Ciresi B.S. The Ohio State University June 2008 Committee Chair: Grace Falciglia, Ph.D. Abstract Background. Crohn’s disease (CD) and ulcerative colitis (UC), collectively known as inflammatory bowel disease (IBD), are chronic illnesses that affect predominately the gastrointestinal tract. The pathogenesis and etiology remain unclear but the importance of environmental factors, in particular diet, is evidenced by the increased incidence rates of the recent decades that genetic inheritance cannot account for. In particular, the quantity of fatty acid consumption has been consistently linked with IBD risk. While several studies have investigated the connections between diet, etiology, signs and symptoms associated with IBD, very few have explored the relationship between disease state and specific fatty acid intake in the pediatric IBD population. Methods. In this cross-sectional study, 100 pediatric patients from Cincinnati Children’s Hospital and the Hospital for Sick Children in Toronto with diagnosed IBD (73 with Crohn’s disease (CD) and 27 with ulcerative colitis (UC)) were included. Three-day diet records were collected from the patients for the assessment of their dietary intake. The abbreviated Pediatric Crohn’s Disease Activity Index (PCDAI), the abbreviated Ulcerative Colitis Activity Index (PUCAI), and markers of inflammation (lipopolysaccharide binding protein (LBP) and S100A12) were used to assess disease severity.