Global Analysis of LARP1 Translation Targets Reveals Tunable and Dynamic Features of 5′ TOP Motifs
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-
Multiplexed Single-Cell Transcriptional Response Profiling to Define Cancer
ARTICLE https://doi.org/10.1038/s41467-020-17440-w OPEN Multiplexed single-cell transcriptional response profiling to define cancer vulnerabilities and therapeutic mechanism of action James M. McFarland 1,11, Brenton R. Paolella 1,11, Allison Warren1, Kathryn Geiger-Schuller 1,2, Tsukasa Shibue1, Michael Rothberg1, Olena Kuksenko1,2, William N. Colgan 1, Andrew Jones1, Emily Chambers1, Danielle Dionne1,2, Samantha Bender1, Brian M. Wolpin3,4,5, Mahmoud Ghandi 1, Itay Tirosh2,6, Orit Rozenblatt-Rosen1,2, Jennifer A. Roth1, Todd R. Golub 1,3,7,8, Aviv Regev 1,2,8,9,10, ✉ ✉ ✉ Andrew J. Aguirre 1,3,4,5,12 , Francisca Vazquez 1,12 & Aviad Tsherniak 1,12 1234567890():,; Assays to study cancer cell responses to pharmacologic or genetic perturbations are typically restricted to using simple phenotypic readouts such as proliferation rate. Information-rich assays, such as gene-expression profiling, have generally not permitted efficient profiling of a given perturbation across multiple cellular contexts. Here, we develop MIX-Seq, a method for multiplexed transcriptional profiling of post-perturbation responses across a mixture of samples with single-cell resolution, using SNP-based computational demultiplexing of single- cell RNA-sequencing data. We show that MIX-Seq can be used to profile responses to chemical or genetic perturbations across pools of 100 or more cancer cell lines. We combine it with Cell Hashing to further multiplex additional experimental conditions, such as post- treatment time points or drug doses. Analyzing the high-content readout of scRNA-seq reveals both shared and context-specific transcriptional response components that can identify drug mechanism of action and enable prediction of long-term cell viability from short- term transcriptional responses to treatment. -

La-Related Protein 1 (LARP1) Binds the Mrna Cap, Blocking Eif4f Assembly
1 La-related protein 1 (LARP1) binds the mRNA cap, blocking eIF4F assembly 2 on TOP mRNAs 3 Roni M. Lahr1, Bruno D. Fonseca2, Gabrielle E. Ciotti1, Hiba A. Al-Ashtal1, Jian-Jun Jia2, 4 Marius R. Niklaus2, Sarah P. Blagden3, Tommy Alain2, Andrea J. Berman1* 5 1University of Pittsburgh, Department of Biological Sciences, Pittsburgh, PA, USA. 6 2Children’s Hospital of Eastern Ontario Research Institute, Ottawa, ON, Canada. 7 3University of Oxford, Department of Oncology, Oxford UK. 8 *Correspondence to: [email protected]. 9 10 Abstract: 11 The 5’terminal oligopyrimidine (5’TOP) motif is a cis-regulatory RNA element located 12 immediately downstream of the 7-methyl-guanosine [m7G] cap of TOP mRNAs, which encode 13 ribosomal proteins and translation factors. In eukaryotes, this motif coordinates the synchronous 14 and stoichiometric expression of the protein components of the translation machinery. La-related 15 protein 1 (LARP1) binds TOP mRNAs, regulating their stability and translation. We present 16 crystal structures of the human LARP1 DM15 region in complex with a 5’TOP motif, a cap 17 analog (m7GTP), and a capped cytosine (m7GpppC) resolved to 2.6, 1.8 and 1.7 Å, respectively. 18 Our binding, competition, and immunoprecipitation data corroborate and elaborate on the 19 mechanism of 5’TOP motif binding by LARP1. We show that LARP1 directly binds the cap and 20 adjacent 5’TOP motif of TOP mRNAs, effectively impeding access of eIF4E to the cap and 21 preventing eIF4F assembly. Thus, LARP1 is a specialized TOP mRNA cap-binding protein that 22 controls ribosome biogenesis. -

L-Leucine Increases Translation of RPS14 and LARP1 in Erythroblasts
LETTERS TO THE EDITOR Table 1. Top 20 differentially translated known 5’TOP mRNAs in L-leucine increases translation of RPS14 and LARP1 L-leucine treated erythroblasts from del(5q) myelodysplastic syn- in erythroblasts from del(5q) myelodysplastic drome patients. syndrome patients Genes LogFC of TE in patients z score patients Deletion of the long arm of chromosome 5 [del(5q)] is RPS15 3.55 2.46 the most common cytogenetic abnormality found in the RPS27A 3.48 2.40 1 myelodysplastic syndromes (MDS). Patients with the 5q- RPS25 3.47 2.39 syndrome have macrocytic anemia and the del(5q) as the RPS20 3.43 2.35 sole karyotypic abnormality.1 Haploinsufficiency of the ribosomal protein gene RPS14, mapping to the common- RPL12 3.35 2.29 ly deleted region (CDR) on chromosome 5q,2 underlies PABPC4 3.01 2.01 3 the erythroid defect found in the 5q- syndrome, and is RPS24 2.97 1.98 associated with p53 activation,4-6 a block in the process- ing of pre-ribosomal RNA,3 and deregulation of riboso- RPS3 2.95 1.96 mal- and translation-related genes.7 Defective mRNA EEF2 2.83 1.86 translation represents a potential therapeutic target in the RPS18 2.76 1.80 5q- syndrome and other ribosomopathies, such as RPS26 2.75 1.79 Diamond-Blackfan anemia (DBA).8 Evidence suggests that the translation enhancer L- RPS5 2.69 1.74 leucine may have some efficacy in the treatment of the RPS21 2.64 1.70 8 5q- syndrome and DBA. A DBA patient treated with L- RPS9 2.54 1.62 leucine showed a marked improvement in anemia and 8 EIF3E 2.53 1.61 achieved transfusion independence. -
![EEF1D Mouse Monoclonal Antibody [Clone ID: OTI4B9] Product Data](https://docslib.b-cdn.net/cover/2076/eef1d-mouse-monoclonal-antibody-clone-id-oti4b9-product-data-82076.webp)
EEF1D Mouse Monoclonal Antibody [Clone ID: OTI4B9] Product Data
OriGene Technologies, Inc. 9620 Medical Center Drive, Ste 200 Rockville, MD 20850, US Phone: +1-888-267-4436 [email protected] EU: [email protected] CN: [email protected] Product datasheet for CF811676 EEF1D Mouse Monoclonal Antibody [Clone ID: OTI4B9] Product data: Product Type: Primary Antibodies Clone Name: OTI4B9 Applications: IHC, WB Recommended Dilution: WB 1:500~2000, IHC 1:2000 Reactivity: Human, Mouse, Rat Host: Mouse Isotype: IgG1 Clonality: Monoclonal Immunogen: Full length human recombinant protein of human EEF1D (NP_115754) produced in E.coli. Formulation: Lyophilized powder (original buffer 1X PBS, pH 7.3, 8% trehalose) Reconstitution Method: For reconstitution, we recommend adding 100uL distilled water to a final antibody concentration of about 1 mg/mL. To use this carrier-free antibody for conjugation experiment, we strongly recommend performing another round of desalting process. (OriGene recommends Zeba Spin Desalting Columns, 7KMWCO from Thermo Scientific) Purification: Purified from mouse ascites fluids or tissue culture supernatant by affinity chromatography (protein A/G) Conjugation: Unconjugated Storage: Store at -20°C as received. Stability: Stable for 12 months from date of receipt. Gene Name: Homo sapiens eukaryotic translation elongation factor 1 delta (EEF1D), transcript variant 1, mRNA. Database Link: NP_115754 Entrez Gene 1936 Human P29692 This product is to be used for laboratory only. Not for diagnostic or therapeutic use. View online » ©2021 OriGene Technologies, Inc., 9620 Medical Center Drive, Ste 200, Rockville, MD 20850, US 1 / 3 EEF1D Mouse Monoclonal Antibody [Clone ID: OTI4B9] – CF811676 Background: This gene encodes a subunit of the elongation factor-1 complex, which is responsible for the enzymatic delivery of aminoacyl tRNAs to the ribosome. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

A Chemical-Genetic Screen for Identifying Substrates of the Er Kinase Perk
University of Pennsylvania ScholarlyCommons Publicly Accessible Penn Dissertations 2014 A Chemical-Genetic Screen for Identifying Substrates of the Er Kinase Perk Nancy L. Maas University of Pennsylvania, [email protected] Follow this and additional works at: https://repository.upenn.edu/edissertations Part of the Biology Commons, Cell Biology Commons, and the Molecular Biology Commons Recommended Citation Maas, Nancy L., "A Chemical-Genetic Screen for Identifying Substrates of the Er Kinase Perk" (2014). Publicly Accessible Penn Dissertations. 1354. https://repository.upenn.edu/edissertations/1354 This paper is posted at ScholarlyCommons. https://repository.upenn.edu/edissertations/1354 For more information, please contact [email protected]. A Chemical-Genetic Screen for Identifying Substrates of the Er Kinase Perk Abstract Cells constantly encounter changing environments that challenge the ability to adapt and survive. Signal transduction networks enable cells to appropriately sense and respond to these changes, and are often mediated through the activity of protein kinases. Protein kinases are a class of enzyme responsible for regulating a broad spectrum of cellular functions by transferring phosphate groups from ATP to substrate proteins, thereby altering substrate activity and function. PERK is a resident kinase of the endoplasmic reticulum, and is responsible for sensing perturbations in the protein folding capacity of the ER. When the influx of unfolded, nascent proteins exceeds the folding capacity of the ER, PERK initiates a cascade of signaling events that enable cell adaptation and ER stress resolution. These signaling pathways are not only essential for the survival of normal cells undergoing ER stress, but are also co-opted by tumor cells in order to survive the oxygen and nutrient-restricted conditions of the tumor microenvironment. -

Genes with 5' Terminal Oligopyrimidine Tracts Preferentially Escape Global Suppression of Translation by the SARS-Cov-2 NSP1 Protein
Downloaded from rnajournal.cshlp.org on September 28, 2021 - Published by Cold Spring Harbor Laboratory Press Genes with 5′ terminal oligopyrimidine tracts preferentially escape global suppression of translation by the SARS-CoV-2 Nsp1 protein Shilpa Raoa, Ian Hoskinsa, Tori Tonna, P. Daniela Garciaa, Hakan Ozadama, Elif Sarinay Cenika, Can Cenika,1 a Department of Molecular Biosciences, University of Texas at Austin, Austin, TX 78712, USA 1Corresponding author: [email protected] Key words: SARS-CoV-2, Nsp1, MeTAFlow, translation, ribosome profiling, RNA-Seq, 5′ TOP, Ribo-Seq, gene expression 1 Downloaded from rnajournal.cshlp.org on September 28, 2021 - Published by Cold Spring Harbor Laboratory Press Abstract Viruses rely on the host translation machinery to synthesize their own proteins. Consequently, they have evolved varied mechanisms to co-opt host translation for their survival. SARS-CoV-2 relies on a non-structural protein, Nsp1, for shutting down host translation. However, it is currently unknown how viral proteins and host factors critical for viral replication can escape a global shutdown of host translation. Here, using a novel FACS-based assay called MeTAFlow, we report a dose-dependent reduction in both nascent protein synthesis and mRNA abundance in cells expressing Nsp1. We perform RNA-Seq and matched ribosome profiling experiments to identify gene-specific changes both at the mRNA expression and translation level. We discover that a functionally-coherent subset of human genes are preferentially translated in the context of Nsp1 expression. These genes include the translation machinery components, RNA binding proteins, and others important for viral pathogenicity. Importantly, we uncovered a remarkable enrichment of 5′ terminal oligo-pyrimidine (TOP) tracts among preferentially translated genes. -

Apoptotic Genes As Potential Markers of Metastatic Phenotype in Human Osteosarcoma Cell Lines
17-31 10/12/07 14:53 Page 17 INTERNATIONAL JOURNAL OF ONCOLOGY 32: 17-31, 2008 17 Apoptotic genes as potential markers of metastatic phenotype in human osteosarcoma cell lines CINZIA ZUCCHINI1, ANNA ROCCHI2, MARIA CRISTINA MANARA2, PAOLA DE SANCTIS1, CRISTINA CAPANNI3, MICHELE BIANCHINI1, PAOLO CARINCI1, KATIA SCOTLANDI2 and LUISA VALVASSORI1 1Dipartimento di Istologia, Embriologia e Biologia Applicata, Università di Bologna, Via Belmeloro 8, 40126 Bologna; 2Laboratorio di Ricerca Oncologica, Istituti Ortopedici Rizzoli; 3IGM-CNR, Unit of Bologna, c/o Istituti Ortopedici Rizzoli, Via di Barbiano 1/10, 40136 Bologna, Italy Received May 29, 2007; Accepted July 19, 2007 Abstract. Metastasis is the most frequent cause of death among malignant primitive bone tumor, usually developing in children patients with osteosarcoma. We have previously demonstrated and adolescents, with a high tendency to metastasize (2). in independent experiments that the forced expression of Metastases in osteosarcoma patients spread through peripheral L/B/K ALP and CD99 in U-2 OS osteosarcoma cell lines blood very early and colonize primarily the lung, and later markedly reduces the metastatic ability of these cancer cells. other skeleton districts (3). Since disseminated hidden micro- This behavior makes these cell lines a useful model to assess metastases are present in 80-90% of OS patients at the time the intersection of multiple and independent gene expression of diagnosis, the identification of markers of invasiveness signatures concerning the biological problem of dissemination. and metastasis forms a target of paramount importance in With the aim to characterize a common transcriptional profile planning the treatment of osteosarcoma lesions and enhancing reflecting the essential features of metastatic behavior, we the prognosis. -

Role of Cyclin-Dependent Kinase 1 in Translational Regulation in the M-Phase
cells Review Role of Cyclin-Dependent Kinase 1 in Translational Regulation in the M-Phase Jaroslav Kalous *, Denisa Jansová and Andrej Šušor Institute of Animal Physiology and Genetics, Academy of Sciences of the Czech Republic, Rumburska 89, 27721 Libechov, Czech Republic; [email protected] (D.J.); [email protected] (A.Š.) * Correspondence: [email protected] Received: 28 April 2020; Accepted: 24 June 2020; Published: 27 June 2020 Abstract: Cyclin dependent kinase 1 (CDK1) has been primarily identified as a key cell cycle regulator in both mitosis and meiosis. Recently, an extramitotic function of CDK1 emerged when evidence was found that CDK1 is involved in many cellular events that are essential for cell proliferation and survival. In this review we summarize the involvement of CDK1 in the initiation and elongation steps of protein synthesis in the cell. During its activation, CDK1 influences the initiation of protein synthesis, promotes the activity of specific translational initiation factors and affects the functioning of a subset of elongation factors. Our review provides insights into gene expression regulation during the transcriptionally silent M-phase and describes quantitative and qualitative translational changes based on the extramitotic role of the cell cycle master regulator CDK1 to optimize temporal synthesis of proteins to sustain the division-related processes: mitosis and cytokinesis. Keywords: CDK1; 4E-BP1; mTOR; mRNA; translation; M-phase 1. Introduction 1.1. Cyclin Dependent Kinase 1 (CDK1) Is a Subunit of the M Phase-Promoting Factor (MPF) CDK1, a serine/threonine kinase, is a catalytic subunit of the M phase-promoting factor (MPF) complex which is essential for cell cycle control during the G1-S and G2-M phase transitions of eukaryotic cells. -
Type of the Paper (Article
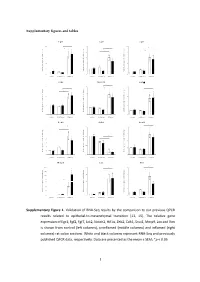
Supplementary figures and tables E g r 1 F g f2 F g f7 1 0 * 5 1 0 * * e e e * g g g * n n n * a a a 8 4 * 8 h h h * c c c d d d * l l l o o o * f f f * n n n o o o 6 3 6 i i i s s s s s s e e e r r r p p p x x x e e e 4 2 4 e e e n n n e e e g g g e e e v v v i i i t t t 2 1 2 a a a l l l e e e R R R 0 0 0 c o n tro l u n in fla m e d in fla m e d c o n tro l u n in fla m e d in fla m e d c o n tro l u n in fla m e d in fla m e d J a k 2 N o tc h 2 H if1 * 3 4 6 * * * e e e g g g n n n a a * * a * h h * h c c c 3 * d d * d l l l * o o o f f 2 f 4 n n n o o o i i i s s s s s s e e e r r 2 r p p p x x x e e e e e e n n n e e 1 e 2 g g g e e 1 e v v v i i i t t t a a a l l l e e e R R R 0 0 0 c o n tro l u n in fla m e d in fla m e d c o n tro l u n in fla m e d in fla m e d c o n tro l u n in fla m e d in fla m e d Z e b 2 C d h 1 S n a i1 * * 7 1 .5 4 * * e e e g g g 6 n n n * a a a * h h h c c c 3 * d d d l l l 5 o o o f f f 1 .0 * n n n * o o o i i i 4 * s s s s s s e e e r r r 2 p p p x x x 3 e e e e e e n n n e e e 0 .5 g g g 2 e e e 1 v v v i i i t t t a a a * l l l e e e 1 * R R R 0 0 .0 0 c o n tro l u n in fla m e d in fla m e d c o n tro l u n in fla m e d in fla m e d c o n tro l u n in fla m e d in fla m e d M m p 9 L o x V im 2 0 0 2 0 8 * * * e e e * g g g 1 5 0 * n n n * a a a * h h h * c c c 1 5 * 6 d d d l l l 1 0 0 o o o f f f n n n o o o i i i 5 0 s s s s s s * e e e r r r 1 0 4 3 0 p p p * x x x e e e * e e e n n n e e e 2 0 g g g e e e 5 2 v v v i i i t t t a a a l l l 1 0 e e e R R R 0 0 0 c o n tro l u n in fla m e d in fla m e d c o n tro l u n in fla m e d in fla m e d c o n tro l u n in fla m e d in fla m e d Supplementary Figure 1. -

Investigation of RNA Binding Proteins Regulated by Mtor
Investigation of RNA binding proteins regulated by mTOR Thesis submitted to the University of Leicester for the degree of Doctor of Philosophy Katherine Morris BSc (University of Leicester) March 2017 1 Investigation of RNA binding proteins regulated by mTOR Katherine Morris, MRC Toxicology Unit, University of Leicester, Leicester, LE1 9HN The mammalian target of rapamycin (mTOR) is a serine/threonine protein kinase which plays a key role in the transduction of cellular energy signals, in order to coordinate and regulate a wide number of processes including cell growth and proliferation via control of protein synthesis and protein degradation. For a number of human diseases where mTOR signalling is dysregulated, including cancer, the clinical relevance of mTOR inhibitors is clear. However, understanding of the mechanisms by which mTOR controls gene expression is incomplete, with implications for adverse toxicological effects of mTOR inhibitors on clinical outcomes. mTOR has been shown to regulate 5’ TOP mRNA expression, though the exact mechanism remains unclear. It has been postulated that this may involve an intermediary factor such as an RNA binding protein, which acts downstream of mTOR signalling to bind and regulate translation or stability of specific messages. This thesis aimed to address this question through the use of whole cell RNA binding protein capture using oligo‐d(T) affinity isolation and subsequent proteomic analysis, and identify RNA binding proteins with differential binding activity following mTOR inhibition. Following validation of 4 identified mTOR‐dependent RNA binding proteins, characterisation of their specific functions with respect to growth and survival was conducted through depletion studies, identifying a promising candidate for further work; LARP1. -

Systematically Profiling the Expression of Eif3 Subunits in Glioma Reveals
Chai et al. Cancer Cell Int (2019) 19:155 https://doi.org/10.1186/s12935-019-0867-1 Cancer Cell International PRIMARY RESEARCH Open Access Systematically profling the expression of eIF3 subunits in glioma reveals the expression of eIF3i has prognostic value in IDH-mutant lower grade glioma Rui‑Chao Chai1,4,6†, Ning Wang2†, Yu‑Zhou Chang3, Ke‑Nan Zhang1,6, Jing‑Jun Li1,6, Jun‑Jie Niu5, Fan Wu1,6*, Yu‑Qing Liu1,6* and Yong‑Zhi Wang1,3,4,6* Abstract Background: Abnormal expression of the eukaryotic initiation factor 3 (eIF3) subunits plays critical roles in tumo‑ rigenesis and progression, and also has potential prognostic value in cancers. However, the expression and clinical implications of eIF3 subunits in glioma remain unknown. Methods: Expression data of eIF3 for patients with gliomas were obtained from the Chinese Glioma Genome Atlas (CGGA) (n 272) and The Cancer Genome Atlas (TCGA) (n 595). Cox regression, the receiver operating characteristic (ROC) curves= and Kaplan–Meier analysis were used to study= the prognostic value. Gene oncology (GO) and gene set enrichment analysis (GSEA) were utilized for functional prediction. Results: In both the CGGA and TCGA datasets, the expression levels of eIF3d, eIF3e, eIF3f, eIF3h and eIF3l highly were associated with the IDH mutant status of gliomas. The expression of eIF3b, eIF3i, eIF3k and eIF3m was increased with the tumor grade, and was associated with poorer overall survival [All Hazard ratio (HR) > 1 and P < 0.05]. By contrast, the expression of eIF3a and eIF3l was decreased in higher grade gliomas and was associated with better overall sur‑ vival (Both HR < 1 and P < 0.05).