Advanced Blood Cell Identification
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

B- and T-Cell Neoplasm Features and Fine Points
B- and T-cell neoplasm features and fine points Karen Lusky June 2021—A case of monoclonal B-cell lymphocytosis and a tour of B- and T-cell morphologies were at the heart of a CAP20 virtual presentation on neoplastic lymphocytosis. Kyle Bradley, MD, associate professor of hematopathology and director of surgical pathology at Emory University, spoke last fall on reactive (CAP TODAY, May 2021) and neoplastic lymphocytosis, with Olga Pozdnyakova, MD, PhD, who addressed neutrophilia and monocytosis (CAP TODAY, February and March 2021). Together they took attendees through a morphology-based approach to hematopoietic neoplasms presenting with an abnormal WBC differential. “Some malignant lymphocytes can closely resemble normal cells,” Dr. Bradley said in a recent interview. “The classic example would be chronic lymphocytic leukemia because individual lymphocytes in that neoplasm can look very similar to normal lymphocytes.” Dr. Bradley shared the case of a 59-year-old man with a clonal B-cell population identified by flow cytometry. It was positive for CD19, CD20, CD5, CD23, CD200, and Kappa, and negative for CD10, FMC7, and Lambda. “This is a typical phenotype for chronic lymphocytic leukemia/small lymphocytic lymphoma,” he said. However, “the blood smear is not what you typically think of for CLL.” (Fig. 1). The differential diagnosis is CLL versus monoclonal B-cell lymphocytosis, he said. In the current and previous WHO classification, CLL requires greater than 5.0 × 109/L clonal B cells. Lower counts are acceptable if there are signs or symptoms of lymphoma. Monoclonal B-cell lymphocytosis is defined as ≤ 5.0 × 109/L clonal B cells in otherwise healthy subjects. -

Morphological Study of Human Blood for Different Diseases
Research Article ISSN: 2574 -1241 DOI: 10.26717/BJSTR.2020.30.004893 Morphological Study of Human Blood for Different Diseases Muzafar Shah1*, Haseena1, Kainat1, Noor Shaba1, Sania1, Sadia1, Akhtar Rasool2, Fazal Akbar2 and Muhammad Israr3 1Centre for Animal Sciences & Fisheries, University of Swat, Pakistan 2Centre for Biotechnology and Microbiology, University of Swat, Pakistan 3Department of Forensic Sciences, University of Swat, Pakistan *Corresponding author: Muzafar Shah, Centre for Animal Sciences & Fisheries, University of Swat, Pakistan ARTICLE INFO ABSTRACT Received: August 25, 2020 The aim of our study was the screening of blood cells on the basis of morphology for different diseased with Morphogenetic characters I e. ear lobe attachment, clinodactyly Published: September 07, 2020 and tongue rolling. For this purpose, 318 blood samples were collected randomly. Samples were examined under the compound microscopic by using 100x with standard Citation: Muzafar Shah, Haseena, method. The results show 63 samples were found normal while in 255 samples, different Kainat, Noor Shaba, Sania, Sadia, et al. types of morphological changes were observed which was 68.5%, in which Bite cell 36%, Morphological Study of Human Blood for Elliptocyte 34%, Tear drop cell 30%, Schistocyte 26%, Hypochromic cell 22.5%, Irregular Different Diseases. Biomed J Sci & Tech Res contracted cell 16%, Echinocytes 15.5%, Roleaux 8%, Boat shape 6.5%, Sickle cell 5%, Keratocyte 4% and Acanthocytes 1.5%. During the screening of slides, bite cell, elliptocyte, tear drop cell, schistocytes, hypochromic cell, irregular contracted cells were found 30(1)-2020.Keywords: BJSTR.Human MS.ID.004893. blood; Diseases; frequently while echinocytes, boat shape cell, acanthocytes, sickle cells and keratocytes Morphological; Acanthocytes; Keratocyte were found rarely. -

Burkitt Lymphoma
Board Review- Part 2B: Malignant HemePath 4/25/2018 Small Lymphocytic Lymphoma SLL: epidemiology SLL: 6.7% of non-Hodgkin lymphoma. Majority of patients >50 y/o (median 65). M:F ratio 2:1. Morphology • Lymph nodes – Effacement of architecture, pseudofollicular pattern of pale areas of large cells in a dark background of small cells. Occasionally is interfollicular. – The predominant cell is a small lymphocyte with clumped chromatin, round nucleus, ocassionally a nucleolus. – Mitotic activity usually very low. Morphology - Pseudofollicles or proliferation centers contain small, medium and large cells. - Prolymphocytes are medium-sized with dispersed chromatin and small nucleoli. - Paraimmunoblasts are medium to large cells with round to oval nuclei, dispersed chromatin, central eosinophilic nucleoli and slightly basophilic cytoplasm. Small Lymphocytic Lymphoma Pseudo-follicle Small Lymphocytic Lymphoma Prolymphocyte Paraimmunoblast Immunophenotype Express weak or dim surface IgM or IgM and IgD, CD5, CD19, CD20 (weak), CD22 (weak), CD79a, CD23, CD43, CD11c (weak). CD10-, cyclin D1-. FMC7 and CD79b negative or weak. Immunophenotype Cases with unmutated Ig variable region genes are reported to be CD38+ and ZAP70+. Immunophenotype Cytoplasmic Ig is detectable in about 5% of the cases. CD5 and CD23 are useful in distinguishing from MCL. Rarely CLL is CD23-. Rarely MCL is CD23+. Perform Cyclin D1 in CD5+/CD23- cases. Some cases with typical CLL morphology may have a different profile (CD5- or CD23-, FMC7+ or CD11c+, or strong sIg, or CD79b+). Genetics Antigen receptor genes: Ig heavy and light chain genes are rearranged. Suggestion of 2 distinct types of SLL defined by the mutational status of the IgVH genes: 40-50% show no somatic mutations of their variable region genes (naïve cells, unmutated). -

Drinking Water Health Advisory for the Cyanobacterial Toxin Cylindrospermopsin
United States Office of Water EPA- 820R15101 Environmental Mail Code 4304T June 2015 Protection Agency Drinking Water Health Advisory for the Cyanobacterial Toxin Cylindrospermopsin Drinking Water Health Advisory for the Cyanobacterial Toxin Cylindrospermopsin Prepared by: U.S. Environmental Protection Agency Office of Water (4304T) Health and Ecological Criteria Division Washington, DC 20460 EPA Document Number: 820R15101 Date: June 15, 2015 ACKNOWLEDGMENTS This document was prepared by U.S. EPA Scientists Lesley V. D’Anglada, Dr.P.H. (lead) and Jamie Strong, Ph.D. Health and Ecological Criteria Division, Office of Science and Technology, Office of Water. EPA gratefully acknowledges the valuable contributions from Health Canada’s Water and Air Quality Bureau, in developing the Analytical Methods and Treatment Technologies information included in this document. This Health Advisory was provided for review and comments were received from staff in the following U.S. EPA Program Offices: U.S. EPA Office of Ground Water and Drinking Water U.S. EPA Office of Science and Technology U.S. EPA Office of Research and Development U.S. EPA Office of Children’s Health Protection U.S. EPA Office of General Counsel This Health Advisory was provided for review and comments were received from the following other federal and health agencies: Health Canada U.S. Department of Health and Human Services, Centers for Disease Control and Prevention Drinking Water Health Advisory for Cylindrospermopsin - June 2015 i TABLE OF CONTENTS ACKNOWLEDGMENTS.....................................................................................................................I -
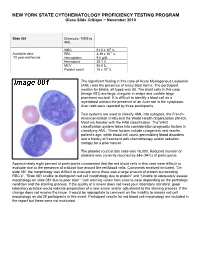
10 11 Cyto Slides 81-85
NEW YORK STATE CYTOHEMATOLOGY PROFICIENCY TESTING PROGRAM Glass Slide Critique ~ November 2010 Slide 081 Diagnosis: MDS to AML 9 WBC 51.0 x 10 /L 12 Available data: RBC 3.39 x 10 /L 72 year-old female Hemoglobin 9.6 g/dL Hematocrit 29.1 % MCV 86.0 fL Platelet count 16 x 109 /L The significant finding in this case of Acute Myelogenous Leukemia (AML) was the presence of many blast forms. The participant median for blasts, all types was 88. The blast cells in this case (Image 081) are large, irregular in shape and contain large prominent nucleoli. It is difficult to identify a blast cell as a myeloblast without the presence of an Auer rod in the cytoplasm. Auer rods were reported by three participants. Two systems are used to classify AML into subtypes, the French- American-British (FAB) and the World Health Organization (WHO). Most are familiar with the FAB classification. The WHO classification system takes into consideration prognostic factors in classifying AML. These factors include cytogenetic test results, patient’s age, white blood cell count, pre-existing blood disorders and a history of treatment with chemotherapy and/or radiation therapy for a prior cancer. The platelet count in this case was 16,000. Reduced number of platelets was correctly reported by 346 (94%) of participants. Approximately eight percent of participants commented that the red blood cells in this case were difficult to evaluate due to the presence of a bluish hue around the red blood cells. Comments received included, “On slide 081 the morphology was difficult to evaluate since there was a large amount of protein surrounding RBC’s”, “Slide 081 unable to distinguish red cell morphology due to protein” and “Unable to adequately assess morphology on slide 081 due to poor stain”. -

TOPIC 5 Lab – B: Diagnostic Tools & Therapies – Blood & Lymphatic
TOPIC 5 Lab – B: Diagnostic Tools & Therapies – Blood & Lymphatic Disorders Refer to chapter 17 and selected online sources. Refer to the front cover of Gould & Dyer for normal blood test values. Complete and internet search for videos from reliable sources on blood donations and blood tests. Topic 5 Lab - A: Blood and Lymphatic Disorders You’ll need to refer to an anatomy & physiology textbook or lab manual to complete many of these objectives. Blood Lab Materials Prepared slides of normal blood Prepared slides of specific blood pathologies Models of formed elements Plaque models of formed elements Blood typing model kits Blood Lab Objectives – by the end of this lab, students should be able to: 1. Describe the physical characteristics of blood. 2. Differentiate between the plasma and serum. 3. Identify the formed elements on prepared slides, diagrams and models and state their main functions. You may wish to draw what you see in the space provided. Formed Element Description / Function Drawing Erythrocyte Neutrophil s e t y c Eosinophils o l u n a r Basophils Leukocytes G e Monocytes t y c o l u n Lymphocytes a r g A Thrombocytes 4. Define differential white blood cell count. State the major function and expected range (percentage) of each type of white blood cell in normal blood. WBC Type Function Expected % Neutrophils Eosinophils Basophils Monocytes Lymphocytes 5. Calculation of the differential count? 6. Define and use in proper context: 1. achlorhydria 5. amyloidosis 2. acute leukemia 6. anemia 3. agnogenic myeloid metaplasia 7. autosplenectomy 4. aleukemic leukemia 8. basophilic stippling 9. -

Review Article Lymphocyte Differentiation, Repertoire Development and Migration: the Need for Mathematical Models
Comput. Math. Applic. Vol. 14, No. 9-12, pp. 657-697, 1987 0097-4943/87 $3.00+0.00 Printed in Great Britain Pergamon Journals Ltd REVIEW ARTICLE LYMPHOCYTE DIFFERENTIATION, REPERTOIRE DEVELOPMENT AND MIGRATION: THE NEED FOR MATHEMATICAL MODELS J. R. LUMB Department of Biology, Atlanta University, Atlanta, GA 30314, U.S.A. INTRODUCTION The immune system is a tremendously complicated and important system, whose central players, the lymphocytes, are able to recognize specific foreign invaders (antigens) with their specific cell surface receptors (immunoglobulins on B-lymphocytes and T cell receptors on T-lymphocytes). A number of different subpopulations of lymphocytes assume the various functions of the immune response: B cell/plasma cell production of specific immunoglobulin, helper/inducer T cells, suppressor T cells and specific cytotoxic T cells. Each of these lymphocyte subpopulations is composed of a large number of clones which each respond to specific antigens via their cell surface receptors. The study of lymphocyte kinetics involves cellular proliferation and differentiation under two different conditions: (1) the initial generation of the repertoire of lymphocytes, each of which displays a specific cell surface receptor, occurs in the absence of triggering by antigen; and (2) the specific clonal lymphocytic proliferation and differentiation occurs in response to antigen recog- nition by these receptors. Both processes involve considerations of cell cycle kinetics and population dynamics, and neither is completely understood. One approach to the study of complex systems, along with the experimental approach, is to construct mathematical models of the processes. Most of the mathematical models of the immune system have focused on the second phase, the antigen-induced activities (for reviews, see Refs [1-6]). -

Lymphopoiesis Cells and Suppresses the Earliest Events in Estrogen
Soluble Frizzled-Related Protein 1 Is Estrogen Inducible in Bone Marrow Stromal Cells and Suppresses the Earliest Events in Lymphopoiesis This information is current as of October 1, 2021. Takafumi Yokota, Kenji Oritani, Karla P. Garrett, Taku Kouro, Makoto Nishida, Isao Takahashi, Michiko Ichii, Yusuke Satoh, Paul W. Kincade and Yuzuru Kanakura J Immunol 2008; 181:6061-6072; ; doi: 10.4049/jimmunol.181.9.6061 Downloaded from http://www.jimmunol.org/content/181/9/6061 References This article cites 67 articles, 27 of which you can access for free at: http://www.jimmunol.org/content/181/9/6061.full#ref-list-1 http://www.jimmunol.org/ Why The JI? Submit online. • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists by guest on October 1, 2021 • Fast Publication! 4 weeks from acceptance to publication *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2008 by The American Association of Immunologists All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. The Journal of Immunology Soluble Frizzled-Related Protein 1 Is Estrogen Inducible in Bone Marrow Stromal Cells and Suppresses the Earliest Events in Lymphopoiesis1 Takafumi Yokota,2* Kenji Oritani,* Karla P. -

Fetal Lymphoid Progenitors Become Restricted to B-1 Fates Coincident with IL- 7Rα Expression
RESEARCH ARTICLE Fetal Lymphoid Progenitors Become Restricted to B-1 Fates Coincident with IL- 7Rα Expression Ryuji Iida1,2*, Kaori Shinoda1, Yuki Hayano1, Yoshinori Nagai3,4, Kiyoshi Takatsu3,5, Taku Kouro1 1 Laboratory of Immune Modulation, National Institutes of Biomedical Innovation, Health and Nutrition, Ibaraki, Osaka, Japan, 2 Department of Cell Cultures, National Institutes of Biomedical Innovation, Health and Nutrition, Ibaraki, Osaka, Japan, 3 Department of Immunobiology and Pharmacological Genetics, a11111 University of Toyama, Toyama-shi, Toyama, Japan, 4 JST, PRESTO, Kawaguchi, Saitama, Japan, 5 Toyama Prefectural Institute for Pharmaceutical Research, Imizu City, Toyama, Japan * [email protected] Abstract OPEN ACCESS B-1 cells represent a sub-fraction of B lymphocytes that participate in T cell-independent Citation: Iida R, Shinoda K, Hayano Y, Nagai Y, Takatsu K, Kouro T (2016) Fetal Lymphoid antibody production and contribute to innate immunity. While the production of B-1 cells is Progenitors Become Restricted to B-1 Fates favored during the fetal waves of lymphopoiesis, it has been unclear when and how that dif- Coincident with IL-7Rα Expression. PLoS ONE 11 ferentiation option is specified. To clarify this, lymphoid and hematopoietic progenitors of (10): e0165676. doi:10.1371/journal. pone.0165676 fetal liver (FL) and adult bone marrow (ABM) were examined for the B cell differentiation potential. Mouse common lymphoid progenitors (CLPs) and more primitive KSL fraction of Editor: Connie J Eaves, B.C. Cancer Agency, CANADA FL and ABM were transferred to SCID mice and donor-derived B cell subsets were ana- lyzed 4 weeks later. CLPs were also cultured on ST2 stromal cells for 6 days prior to trans- Received: July 30, 2016 plantation. -

Red-Cell and Plasma Lipids in Acanthocytosis
RED-CELL AND PLASMA LIPIDS IN ACANTHOCYTOSIS Peter Ways, … , Claude F. Reed, Donald J. Hanahan J Clin Invest. 1963;42(8):1248-1260. https://doi.org/10.1172/JCI104810. Research Article Find the latest version: https://jci.me/104810/pdf Journal of Clinical Investigation Vol. 42, No. 8, 1963 RED-CELL AND PLASMA LIPIDS IN ACANTHOCYTOSIS * By PETER WAYS,t CLAUDE F. REED, AND DONALD J. HANAHAN WITH THE TECHNICAL ASSISTANCE OF DOLORES DONG, SUSAN PALMER, MARION MURPHY, AND GERALDINE ROBERTS (From the Departments of Biochemistry and Medicine, University of Washington, Seattle, Wash., and the Department of Medicine, University of Rochester School of Medicine and Dentistry, Rochester, N. Y.) (Submitted for publication January 18, 1963; accepted April 11, 1963) The rare syndrome of acanthocytosis was first tological, and gastrointestinal) are all secondary described in 1950 by Bassen and Kornzweig (1) to the absence of low-density lipoprotein (6, 7). and two years later by Singer, Fisher, and Perl- This is still conjectural, however, and the bio- stein (2). The principal manifestations reported chemical defect responsible for the disease has not were steatorrhea, "atypical" retinitis pigmentosa, been elucidated. progressive neurological deficits, and "thorny" The clinical and chemical abnormalities of acan- red cells. The most complete case reports to date thocytosis suggested that it might be an ideal syn- are those of Meir, Schwartz, and Boshes (3) and drome in which to seek abnormalities in red-cell Rey (4), who well review the clinical aspects. lipids. Values previously reported for total red- The erythrocytes of affected individuals have char- cell lipids in one case (3) and for red-cell choles- acteristic, spikelike excrescences, a normal or de- terol and total phospholipid (6) in another were creased life-span, probably normal osmotic fragil- probably normal (normal values were not given), ity, normal acid fragility, increased susceptibility but the possibilities of qualitative changes were to mechanical trauma, and an increased rate of not investigated. -

Notch Receptor Ligation Progress to DN2/3 Stage Thymocytes with Propensity of Adult Lymphoid Progenitors To
Propensity of Adult Lymphoid Progenitors to Progress to DN2/3 Stage Thymocytes with Notch Receptor Ligation This information is current as Jiaxue Huang, Karla P. Garrett, Rosana Pelayo, Juan Carlos of September 29, 2021. Zúñiga-Pflücker, Howard T. Petrie and Paul W. Kincade J Immunol 2005; 175:4858-4865; ; doi: 10.4049/jimmunol.175.8.4858 http://www.jimmunol.org/content/175/8/4858 Downloaded from References This article cites 45 articles, 20 of which you can access for free at: http://www.jimmunol.org/content/175/8/4858.full#ref-list-1 http://www.jimmunol.org/ Why The JI? Submit online. • Rapid Reviews! 30 days* from submission to initial decision • No Triage! Every submission reviewed by practicing scientists • Fast Publication! 4 weeks from acceptance to publication by guest on September 29, 2021 *average Subscription Information about subscribing to The Journal of Immunology is online at: http://jimmunol.org/subscription Permissions Submit copyright permission requests at: http://www.aai.org/About/Publications/JI/copyright.html Email Alerts Receive free email-alerts when new articles cite this article. Sign up at: http://jimmunol.org/alerts The Journal of Immunology is published twice each month by The American Association of Immunologists, Inc., 1451 Rockville Pike, Suite 650, Rockville, MD 20852 Copyright © 2005 by The American Association of Immunologists All rights reserved. Print ISSN: 0022-1767 Online ISSN: 1550-6606. The Journal of Immunology Propensity of Adult Lymphoid Progenitors to Progress to DN2/ 3 Stage Thymocytes with Notch Receptor Ligation1 Jiaxue Huang,*† Karla P. Garrett,* Rosana Pelayo,* Juan Carlos Zu´n˜iga-Pflu¨cker,‡ Howard T. -

Prof. Salma Afrose
Drop of Blood – Unravels Mysteries Prof. Salma Afrose Department of Hematology Dhaka Medical College Peripheral Blood Film (PBF) PBF is a laboratory workup that involves cytology of Peripheral blood cell smear on a slide Clinical history Physical examination Lab investigation Diagnosis Importance of PBF Basic & highly informative hematological tool for • Screening • Diagnosis • Monitoring disease progression & • Therapeutic response So for successful clinical practice understanding interpretation of PBF is essential Indication • Clinical request from attending clinician (based on clinical suspicion) • Sometimes from laboratory due to abnormal finding on an automated counter • Unexplained cytopenia • Unexplained leukocytosis, lymphocytosis, monocytosis • Unexplained hemolysis or jaundice • Sepsis • Liver failure • Hematological malignancies • Severe bacterial sepsis • Parasitic infection • Anemia evaluation Clinical indications for examination of PBF • Features suggestive of anemia, unexplained jaundice, or both • Features suggestive of sickle cell disease – dactylitis or sudden splenic enlargement and pallor in a young child or, in an older child or adult, limb, abdominal, or chest pain • Features suggestive of thrombocytopenia (e.g. petechiae or abnormal bruising) or neutropenia (e.g. unexpected or sever infection) • Features suggestive of a lymphoma or other lymphoproliferative disorder – lymphadenopathy, splenomegaly, enlargement of the thymus (a mediastinal mass on radiology) or other lymphoid organs, skin lesions suggestive of infiltration,