Comparative Proteomic Analysis of Human Placenta Derived from Assisted Reproductive Technology
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Propranolol-Mediated Attenuation of MMP-9 Excretion in Infants with Hemangiomas
Supplementary Online Content Thaivalappil S, Bauman N, Saieg A, Movius E, Brown KJ, Preciado D. Propranolol-mediated attenuation of MMP-9 excretion in infants with hemangiomas. JAMA Otolaryngol Head Neck Surg. doi:10.1001/jamaoto.2013.4773 eTable. List of All of the Proteins Identified by Proteomics This supplementary material has been provided by the authors to give readers additional information about their work. © 2013 American Medical Association. All rights reserved. Downloaded From: https://jamanetwork.com/ on 10/01/2021 eTable. List of All of the Proteins Identified by Proteomics Protein Name Prop 12 mo/4 Pred 12 mo/4 Δ Prop to Pred mo mo Myeloperoxidase OS=Homo sapiens GN=MPO 26.00 143.00 ‐117.00 Lactotransferrin OS=Homo sapiens GN=LTF 114.00 205.50 ‐91.50 Matrix metalloproteinase‐9 OS=Homo sapiens GN=MMP9 5.00 36.00 ‐31.00 Neutrophil elastase OS=Homo sapiens GN=ELANE 24.00 48.00 ‐24.00 Bleomycin hydrolase OS=Homo sapiens GN=BLMH 3.00 25.00 ‐22.00 CAP7_HUMAN Azurocidin OS=Homo sapiens GN=AZU1 PE=1 SV=3 4.00 26.00 ‐22.00 S10A8_HUMAN Protein S100‐A8 OS=Homo sapiens GN=S100A8 PE=1 14.67 30.50 ‐15.83 SV=1 IL1F9_HUMAN Interleukin‐1 family member 9 OS=Homo sapiens 1.00 15.00 ‐14.00 GN=IL1F9 PE=1 SV=1 MUC5B_HUMAN Mucin‐5B OS=Homo sapiens GN=MUC5B PE=1 SV=3 2.00 14.00 ‐12.00 MUC4_HUMAN Mucin‐4 OS=Homo sapiens GN=MUC4 PE=1 SV=3 1.00 12.00 ‐11.00 HRG_HUMAN Histidine‐rich glycoprotein OS=Homo sapiens GN=HRG 1.00 12.00 ‐11.00 PE=1 SV=1 TKT_HUMAN Transketolase OS=Homo sapiens GN=TKT PE=1 SV=3 17.00 28.00 ‐11.00 CATG_HUMAN Cathepsin G OS=Homo -

Apoptosis and Differentiation Commitment: Novel Insights Revealed by Gene Profiling Studies in Mouse Embryonic Stem Cells
Cell Death and Differentiation (2006) 13, 564–575 & 2006 Nature Publishing Group All rights reserved 1350-9047/06 $30.00 www.nature.com/cdd Apoptosis and differentiation commitment: novel insights revealed by gene profiling studies in mouse embryonic stem cells D Duval1,2,4, M Trouillas3,4, C Thibault2, D Dembele´ 2, Introduction F Diemunsch2, B Reinhardt2, AL Mertz2, A Dierich2 Mouse embryonic stem (ES) cells, which are maintained and H Bœuf*,3 pluripotent in vitro with leukemia inhibitory factor (LIF) cytokine, are instrumental to study LIF-dependent cell 1 UMR5096-CNRS/UP/IRD, Perpignan, France 2 IGBMC/CNRS/INSERM, Strasbourg, France pluripotency as well as the first steps of differentiation 3 UMR-5164-CNRS-CIRID/Universite´ Bordeaux2, Bordeaux, France commitment triggered upon LIF starvation. As we recently 4 These authors contributed equally to this work reported, these cells could also be used to unravel the early * Corresponding author: H Bœuf, UMR-5164-CNRS-CIRID, Universite´ steps of apoptosis, a physiological cell death process Bordeaux2, Bat.1B, BP14, 146 rue Le´o Saignat, 33076 Bordeaux, France. occurring during the first embryogenesis stages. Indeed, the Tel: þ 05 57 57 46 33; Fax: þ 05 57 57 14 72; formation of the cavities, which starts at the blastocyst stage, E-mail:helene.bœ[email protected] is dependent on a specific cell death program, which includes caspase 3 cleavage and induction of the apoptosis-inducing Received 10.3.05; revised 01.9.05; accepted 01.9.05; published online 25.11.05 1 Edited by R De Maria factor (AIF)-complex proteins. -

Supplemental Table1a.Xlsx
Electronic Supplementary Material (ESI) for Analyst This journal is © The Royal Society of Chemistry 2013 Supplemental Table 1A This table includes the taxonomically significant peptides from HeLa cultured VACV samples purified by filter purification. The peptide, uniprot ID and protein name from one of the homologous proteins is provided. The species specificity is provided along with the frequency of observation (n = 10). Peptides observed in more than replicate data set are included. Peptide Uniprot ID Protein Description Specificity Freq. LLNENSYVPR P02786 protein 1 Primates, racoon 0.9 LATQLTGPVMPVR A8K4C8 60S ribosomal protein L13 Homo sapiens 0.8 HVGK B0JYN6 Alpha-2-HS-glycoprotein Bos taurus0.8 LAVDEEENADNNTK F8WBE5 protein 1 Great Apes 0.8 VTLTSEEEAR B4DJI1 L-lactate dehydrogenase Primates, Hamster 0.7 LGEYGFQNELIVR B0JYQ0 ALB protein Bos taurus 0.6 GTVTDFPGFDER D6RCN3 Annexin A5 Elephant 0.6 YTPSGQAGAAASESLF Fructose-bisphosphate Great apes, cucumber VSNHAY H3BR68 aldolase A (Fragment) and some rodents 0.6 VGGHAAEYGAEALER P01966 Hemoglobin subunit alpha Bos taurus 0.6 ALTGHLEEVVLALLK P04083 Annexin A1 some rodents 0.5 AQGPAASAEEPKPVEA Brain acid soluble protein PAANSDQTVTVKE P80723 1 Homo sapiens 0.5 FYALSASFEPFSNK P27797 Calreticulin Primates not gorilla 0.5 PK P02081 beta Bos taurus0.5 NLK F5H6B1 protein 1Gorilla 0.5 K P02786 protein 1Primates 0.5 YNSQLLSFVR P02786 protein 1 Great Apes 0.5 TPIVGQPSIPGGPVR B0JYN6 Alpha-2-HS-glycoprotein Bos taurus 0.4 AGTDLLNFLSSFIDPK P81644 Apolipoprotein A-II Bos taurus 0.4 SELPLDPLPVPTEEGNP -

The Importance of Eukaryotic Ferritins in Iron Handling and Cytoprotection
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by Archivio istituzionale della ricerca - Università di Brescia Biochem. J. (2015) 472, 1–15 doi:10.1042/BJ20150787 1 REVIEW ARTICLE The importance of eukaryotic ferritins in iron handling and cytoprotection Paolo Arosio*1, Fernando Carmona*, Raffaella Gozzelino†, Federica Maccarinelli* and Maura Poli* *Laboratory of Molecular Biology, Department of Molecular and Translational Medicine (DMMT), University of Brescia, Brescia, Italy †Inflammation and Neurodegeneration Laboratory, Chronic Disease Research Centre (CEDOC)/FCM, NOVA Medical School, Lisbon, Portugal Ferritins, the main intracellular iron storage proteins, have been structure is highly conserved among different phyla. It exerts an studied for over 60 years, mainly focusing on the mammalian important cytoprotective function against oxidative damage and ones. This allowed the elucidation of the structure of these proteins plays a role in innate immunity, where it also contributes to prevent and the mechanisms regulating their iron incorporation and parenchymal tissue from the cytotoxicity of pro-inflammatory mineralization. However, ferritin is present in most, although not agonists released by the activation of the immune response all, eukaryotic cells, comprising monocellular and multicellular activation. Less clear are the properties of the secretory ferritins invertebrates and vertebrates. The aim of this review is to provide expressed by insects and molluscs, which may be important for an update on the general properties of ferritins that are common to understanding the role played by serum ferritin in mammals. various eukaryotic phyla (except plants), and to give an overview on the structure, function and regulation of ferritins. -

Differential Proteomic Analysis of Hepatocellular Carcinomas From
CANCER GENOMICS & PROTEOMICS 17 : 669-685 (2020) doi:10.21873/cgp.20222 Differential Proteomic Analysis of Hepatocellular Carcinomas from Ppp2r5d Knockout Mice and Normal (Knockout) Livers CAROLINE LAMBRECHT 1, GABRIELA BOMFIM FERREIRA 2, JUDIT DOMÈNECH OMELLA 1, LOUIS LIBBRECHT 3, RITA DE VOS 4, RITA DERUA 1, CHANTAL MATHIEU 2, LUT OVERBERGH 2, ETIENNE WAELKENS 1 and VEERLE JANSSENS 1,5 1Laboratory of Protein Phosphorylation and Proteomics, Department Cellular and Molecular Medicine, University of Leuven (KU Leuven), Leuven, Belgium; 2Clinical and Experimental Endocrinology, Department Clinical and Experimental Medicine, University of Leuven (KU Leuven), Leuven, Belgium; 3Department of Pathology, Université Catholique de Louvain (UCL), Brussels, Belgium; 4Translational Cell and Tissue Research, Department Imaging and Pathology, University of Leuven (KU Leuven), Leuven, Belgium; 5LKI, KU Leuven Cancer Institute, Leuven, Belgium Abstract. Background: Hepatocellular carcinoma (HCC) ‘gastrointestinal disease’ as top hits. Conclusion: We is the major type of primary liver cancer. Mice lacking the identified several proteins for further exploration as novel tumor-suppressive protein phosphatase 2A subunit B56 δ potential HCC biomarkers, and independently underscored (Ppp2r5d ) spontaneously develop HCC, correlating with the relevance of Ppp2r5d knockout mice as a valuable increased c-MYC oncogenicity. Materials and Methods: We hepatocarcinogenesis model. used two-dimensional difference gel electrophoresis-coupled matrix-assisted laser desorption/ionization time-of-flight Hepatocellular carcinoma (HCC) is the most common primary mass spectrometry to identify differential proteomes of livers liver cancer, and the second leading cause of cancer-related death from wild-type, non-cancerous and HCC-affected B56 δ worldwide (1). Most patients with HCC are diagnosed at an knockout mice. -

Repression of Ferritin Light Chain Translation by Human Eif3
RESEARCH ARTICLE Repression of ferritin light chain translation by human eIF3 Mia C Pulos-Holmes1, Daniel N Srole1†, Maria G Juarez1, Amy S-Y Lee2, David T McSwiggen1, Nicholas T Ingolia1,3, Jamie H Cate1,3,4,5* 1Department of Molecular & Cell Biology, University of California, Berkeley, Berkeley, United States; 2Biology Department, Rosenstiel Basic Medical Science Research Center, Brandeis University, Waltham, United States; 3California Institute for Quantitative Biosciences, University of California, Berkeley, Berkeley, United States; 4Department of Chemistry, University of California, Berkeley, Berkeley, United States; 5Molecular Biophysics & Integrated Bioimaging Division, Lawrence Berkeley National Laboratory, Berkeley, United States Abstract A central problem in human biology remains the discovery of causal molecular links between mutations identified in genome-wide association studies (GWAS) and their corresponding disease traits. This challenge is magnified for variants residing in non-coding regions of the genome. Single-nucleotide polymorphisms (SNPs) in the 5’ untranslated region (5’-UTR) of the ferritin light chain (FTL) gene that cause hyperferritinemia are reported to disrupt translation *For correspondence: FTL [email protected] repression by altering iron regulatory protein (IRP) interactions with the mRNA 5’-UTR. Here, we show that human eukaryotic translation initiation factor 3 (eIF3) acts as a distinct repressor of † Present address: Department FTL mRNA translation, and eIF3-mediated FTL repression is disrupted by a subset of SNPs in FTL of Molecular & Medical that cause hyperferritinemia. These results identify a direct role for eIF3-mediated translational Pharmacology, David Geffen control in a specific human disease. School of Medicine at UCLA, DOI: https://doi.org/10.7554/eLife.48193.001 University of California, Los Angeles, Los Angeles, United States Competing interests: The Introduction authors declare that no Iron is essential for a spectrum of metabolic pathways and cellular growth. -

Ferritin Heavy Chain (FTH1) Rabbit Polyclonal Antibody Product Data
OriGene Technologies, Inc. 9620 Medical Center Drive, Ste 200 Rockville, MD 20850, US Phone: +1-888-267-4436 [email protected] EU: [email protected] CN: [email protected] Product datasheet for TA326948 Ferritin Heavy Chain (FTH1) Rabbit Polyclonal Antibody Product data: Product Type: Primary Antibodies Applications: IF, IHC, WB Recommended Dilution: WB 1:500 - 1:2000, IHC 1:50- 1:200, IF 1:50- 1:200 Reactivity: Human, Mouse, Rat Host: Rabbit Isotype: IgG Clonality: Polyclonal Immunogen: Recombinant protein of human FTH1 Formulation: Store at -20C or -80C. Avoid freeze / thaw cycles. Buffer: PBS with 0.02% sodium azide, 50% glycerol, pH7.3 Concentration: lot specific Purification: Affinity purification Conjugation: Unconjugated Storage: Store at -20°C as received. Stability: Stable for 12 months from date of receipt. Gene Name: ferritin heavy chain 1 Database Link: NP_002023 Entrez Gene 14319 MouseEntrez Gene 25319 RatEntrez Gene 2495 Human P02794 This product is to be used for laboratory only. Not for diagnostic or therapeutic use. View online » ©2021 OriGene Technologies, Inc., 9620 Medical Center Drive, Ste 200, Rockville, MD 20850, US 1 / 3 Ferritin Heavy Chain (FTH1) Rabbit Polyclonal Antibody – TA326948 Background: Ferritin (FTH) is a ubiquitous and highly conserved protein which plays a major role in iron homeostasis by sequestering and storing iron in a non-toxic and bioavailable form. The assembled ferritin molecule, often referred to as a nanocage, can store up to 4,500 atoms of iron. It forms a holoenzyme of ~450 kDa, consisting of 24 subunits made up of two types of polypeptide chains: ferritin heavy chain and ferritin light chain, each having unique functions. -

Ferritin Heavy Chain (F23): Sc-51887
SANTA CRUZ BIOTECHNOLOGY, INC. ferritin heavy chain (F23): sc-51887 The Power to Question BACKGROUND APPLICATIONS Mammalian ferritins consist of 24 subunits made up of two types of polypep- ferritin heavy chain (F23) is recommended for detection of ferritin of human tide chains, ferritin heavy chain and ferritin light chain, which each have origin by ELISA. unique functions. Ferritin heavy chains catalyze the first step in iron storage, Suitable for use as control antibody for ferritin heavy chain siRNA (h): the oxidation of Fe (II), whereas ferritin light chains promote the nucleation sc-40575. of ferrihydrite, enabling storage of Fe (III). The most prominent role of mam- malian ferritins is to provide iron-buffering capacity to cells. In addition to iron Molecular Weight of ferritin heavy chain: 21 kDa. buffering, heavy chain ferritin is also involved in the regulation of thymidine biosynthesis via increased expression of cytoplasmic serine hydroxymethyl- STORAGE transferase, which is a limiting factor in thymidylate synthesis in MCF-7 cells. Store at 4° C, **DO NOT FREEZE**. Stable for one year from the date of Light chain ferritin is involved in cataracts by at least two mechanisms, shipment. Non-hazardous. No MSDS required. hereditary hyperferritinemia cataract syndrome, in which light chain ferritin is overexpressed, and oxidative stress, an important factor in the develop- PROTOCOLS ment of ageing-related cataracts. The gene encoding human ferritin heavy See our web site at www.scbt.com or our catalog for detailed protocols chain maps to chromosome 11q13 and the human ferritin light chain gene and support products. maps to chromosome 19q13.3-q13.4. -

Ferritin L Antibody
Efficient Professional Protein and Antibody Platforms Ferritin L Antibody Basic information: Catalog No.: UPA63379 Source: Rabbit Size: 50ul/100ul Clonality: Monoclonal Concentration: 1mg/ml Isotype: Rabbit IgG Purification: Protein A affinity purified Useful Information: WB:1:1000-1:5000 Applications: IHC:1:100-1:500 ICC:1:100-1:200 Reactivity: Human, Mouse, Rat Specificity: This antibody recognizes Ferritin L protein. Immunogen: Recombinant protein Mammalian ferritins consist of 24 subunits made up of two types of poly- peptide chains, ferritin heavy chain and ferritin light chain, which each have unique functions. Ferritin heavy chains catalyze the first step in iron storage, the oxidation of Fe (II), whereas ferritin light chains promote the nucleation of ferrihydrite, enabling storage of Fe (III). The most prominent role of mammalian ferritins is to provide iron-buffering capacity to cells. In addition to iron buffering, heavy chain ferritin is also involved in the regulation of Description: thymidine biosynthesis via increased expression of cytoplasmic serine hy- droxymethyltransferase, which is a limiting factor in thymidylate synthesis in MCF-7 cells. Light chain ferritin is involved in cataracts by at least two mechanisms, hereditary hyperferritinemia cataract syndrome, in which light chain ferritin is overexpressed, and oxidative stress, an important factor in the development of ageing-related cataracts. The gene encoding human ferritin heavy chain maps to chromosome 11q13 and the human ferritin light chain gene maps to chromosome 19q13.3-q13.4. Uniprot: P02792(Human) P29391(Mouse) P02793(Rat) BiowMW: 54 kDa Buffer: 1*TBS (pH7.4), 1%BSA, 40%Glycerol. Preservative: 0.05% Sodium Azide. Storage: Store at 4°C short term and -20°C long term. -

ANXA3 Is Upregulated by Hypoxia-Inducible Factor 1-Alpha and Promotes Colon Cancer Growth
7449 Original Article ANXA3 is upregulated by hypoxia-inducible factor 1-alpha and promotes colon cancer growth Kunli Du1, Jiahui Ren2, Zhongxue Fu3, Xingye Wu3, Jianyong Zheng1, Xing Li4 1Department of Gastrointestinal Surgery, The First Affiliated Hospital of Air Force Medical University, Xi’an, China; 2Department of Anus and Intestine Surgery, Xi’an Mayinglong Anorectal Hospital, Xi’an, China; 3Department of Gastrointestinal Surgery, The First Affiliated Hospital of Chongqing Medical University, Chongqing, China; 4Nanjing Yuheming Medical Nutrition Research Institute, Nanjing, China Contributions: (I) Conception and design: J Zheng; (II) Administrative support: X Li; (III) Provision of study materials or patients: K Du, J Ren, Z Fu; (IV) Collection and assembly of data: K Du, J Ren, Z Fu; (V) Data analysis and interpretation: X Wu; (VI) Manuscript writing: All authors; (VII) Final approval of manuscript: All authors. Correspondence to: Xingye Wu. Department of Gastrointestinal Surgery, The First Affiliated Hospital of Chongqing Medical University, Youyi Road 1st, Yuzhong District, Chongqing 400016, China. Email: [email protected]; Jianyong Zheng. Department of Gastrointestinal Surgery, The First Affiliated Hospital of Air Force Medical University, 127 Changle West Road, Xincheng District, Xi’an 710000, China. Email: [email protected]. Background: Annexin A3 (ANXA3) is overexpressed in various cancers and is a potential target for cancer treatment. However, clinical implication and biological function of ANXA3 in colon cancer remain unknown. This study aimed to investigate the relationship between hypoxia-inducible factor 1-alpha (HIF-1α) and ANXA3, and explore the function of ANXA3 in colon carcinoma. Methods: Expression levels of HIF-1α and ANXA3 in human colon carcinoma specimens and colon cancer cell lines were detected by immunohistochemistry, real-time PCR and Western blot analysis. -
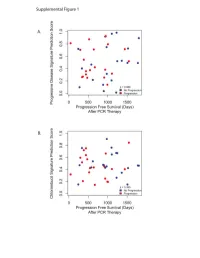
Supplementary Data
Progressive Disease Signature Upregulated probes with progressive disease U133Plus2 ID Gene Symbol Gene Name 239673_at NR3C2 nuclear receptor subfamily 3, group C, member 2 228994_at CCDC24 coiled-coil domain containing 24 1562245_a_at ZNF578 zinc finger protein 578 234224_at PTPRG protein tyrosine phosphatase, receptor type, G 219173_at NA NA 218613_at PSD3 pleckstrin and Sec7 domain containing 3 236167_at TNS3 tensin 3 1562244_at ZNF578 zinc finger protein 578 221909_at RNFT2 ring finger protein, transmembrane 2 1552732_at ABRA actin-binding Rho activating protein 59375_at MYO15B myosin XVB pseudogene 203633_at CPT1A carnitine palmitoyltransferase 1A (liver) 1563120_at NA NA 1560098_at AKR1C2 aldo-keto reductase family 1, member C2 (dihydrodiol dehydrogenase 2; bile acid binding pro 238576_at NA NA 202283_at SERPINF1 serpin peptidase inhibitor, clade F (alpha-2 antiplasmin, pigment epithelium derived factor), m 214248_s_at TRIM2 tripartite motif-containing 2 204766_s_at NUDT1 nudix (nucleoside diphosphate linked moiety X)-type motif 1 242308_at MCOLN3 mucolipin 3 1569154_a_at NA NA 228171_s_at PLEKHG4 pleckstrin homology domain containing, family G (with RhoGef domain) member 4 1552587_at CNBD1 cyclic nucleotide binding domain containing 1 220705_s_at ADAMTS7 ADAM metallopeptidase with thrombospondin type 1 motif, 7 232332_at RP13-347D8.3 KIAA1210 protein 1553618_at TRIM43 tripartite motif-containing 43 209369_at ANXA3 annexin A3 243143_at FAM24A family with sequence similarity 24, member A 234742_at SIRPG signal-regulatory protein gamma -

(12) United States Patent (10) Patent No.: US 8,084,017 B2 Ahrens (45) Date of Patent: Dec
US008O84017B2 (12) United States Patent (10) Patent No.: US 8,084,017 B2 Ahrens (45) Date of Patent: Dec. 27, 2011 (54) CONTRASTAGENTS FOR MAGNETIC Cohen, B., et al., “Ferritin as an Endogenous MRI Reporter for RESONANCE MAGING AND METHODS Noninvasive Imaging of Gene Expression in C6 Glioma Tumors.” RELATED THERETO Neoplasia, 7(2):109-117 (2005). Genove, G., “A new transgene reporter for in vivo magnetic reso (75) Inventor: Eric Ahrens, Pittsburgh, PA (US) nance imaging.” Nature Medicine, 11(4):450-454 (2005). Ichikawa, T., et al., “MRI of Transgene Expression: Correlation of (73) Assignee: Carnegie Mellon University, Pittsburgh, Therapeutic Gene Expression.” Neoplasia, 4(6):523-530 (2002). PA (US) Moore, A., et al., “Measuring transferrin receptor gene expression by NMR imaging.” Biochimica et Biophysica Acta, 14 02:239-249 (*) Notice: Subject to any disclaimer, the term of this (1998). patent is extended or adjusted under 35 Walter, G., et al., “Noninvasive measurement of gene expression in U.S.C. 154(b) by 738 days. skeletal muscle.” PNAS,97(10):5151-5155 (2000). Weissleder, R., et al., “MR Imaging and Scintigraphy of Gene (21) Appl. No.: 10/384,496 Expression through Melanin Induction.” Radiology, 204:425-429 (1997). (22) Filed: Mar. 7, 2003 Ahrens, et al., “A model for MRI contrast enhancement using T. agents”. Proc. Acad. Sci. USA. vol. 95, pp. 8443-8448, Applied (65) Prior Publication Data Biological Sciences (1998). Ahrens, et al., “Peripheral somatosensory fMRI in mouse at 11.7T". US 2003/0219385 A1 Nov. 27, 2003 NMR in Biomedicine, NMR Biomed. 14:318-324 (2001).