Chromosome 8P As a Potential Hub for Developmental Neuropsychiatric
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Peking University-Juntendo University Joint Symposium on Cancer Research and Treatment ADAM28 (A Disintegrin and Metalloproteinase 28) in Cancer Cell Proliferation and Progression
Whatʼs New from Juntendo University, Tokyo Juntendo Medical Journal 2017. 63(5), 322-325 Peking University - Juntendo University Joint Symposium on Cancer Research and Treatment ADAM28 (a Disintegrin and Metalloproteinase 28) in Cancer Cell Proliferation and Progression YASUNORI OKADA* *Department of Pathophysiology for Locomotive and Neoplastic Diseases, Juntendo University Graduate School of Medicine, Tokyo, Japan A disintegrinandmetalloproteinase 28 (ADAM28) is overexpressedpredominantlyby carcinoma cells in more than 70% of the non-small cell lung carcinomas, showing positive correlations with carcinoma cell proliferation and metastasis. ADAM28 cleaves insulin-like growth factor binding protein-3 (IGFBP-3) in the IGF-I/IGFBP-3 complex, leading to stimulation of cell proliferation by intact IGF-I released from the complex. ADAM28 also degrades von Willebrand factor (VWF), which induces apoptosis in human carcinoma cell lines with negligible ADAM28 expression, andthe VWF digestionby ADAM28-expressing carcinoma cells facilitates them to escape from VWF-induced apoptosis, resulting in promotion of metastasis. We have developed human antibodies against ADAM28 andshown that one of them significantly inhibits tumor growth andmetastasis using lung adenocarcinoma cells. Our data suggest that ADAM28 may be a new molecular target for therapy of the patients with ADAM28-expressing non-small cell lung carcinoma. Key words: a disintegrin and metalloproteinase 28 (ADAM28), cell proliferation, invasion, metastasis, human antibody inhibitor Introduction human cancers 2). However, development of the synthetic inhibitors of MMPs andtheir application Cancer cell proliferation andprogression are for treatment of the cancer patients failed 3). modulated by proteolytic cleavage of tissue micro- On the other hand, members of the ADAM (a environmental factors such as extracellular matrix disintegrin and metalloproteinase) gene family, (ECM), growth factors andcytokines, receptors another family belonging to the metzincin gene andcell adhesionmolecules. -

Systems and Chemical Biology Approaches to Study Cell Function and Response to Toxins
Dissertation submitted to the Combined Faculties for the Natural Sciences and for Mathematics of the Ruperto-Carola University of Heidelberg, Germany for the degree of Doctor of Natural Sciences Presented by MSc. Yingying Jiang born in Shandong, China Oral-examination: Systems and chemical biology approaches to study cell function and response to toxins Referees: Prof. Dr. Rob Russell Prof. Dr. Stefan Wölfl CONTRIBUTIONS The chapter III of this thesis was submitted for publishing under the title “Drug mechanism predominates over toxicity mechanisms in drug induced gene expression” by Yingying Jiang, Tobias C. Fuchs, Kristina Erdeljan, Bojana Lazerevic, Philip Hewitt, Gordana Apic & Robert B. Russell. For chapter III, text phrases, selected tables, figures are based on this submitted manuscript that has been originally written by myself. i ABSTRACT Toxicity is one of the main causes of failure during drug discovery, and of withdrawal once drugs reached the market. Prediction of potential toxicities in the early stage of drug development has thus become of great interest to reduce such costly failures. Since toxicity results from chemical perturbation of biological systems, we combined biological and chemical strategies to help understand and ultimately predict drug toxicities. First, we proposed a systematic strategy to predict and understand the mechanistic interpretation of drug toxicities based on chemical fragments. Fragments frequently found in chemicals with certain toxicities were defined as structural alerts for use in prediction. Some of the predictions were supported with mechanistic interpretation by integrating fragment- chemical, chemical-protein, protein-protein interactions and gene expression data. Next, we systematically deciphered the mechanisms of drug actions and toxicities by analyzing the associations of drugs’ chemical features, biological features and their gene expression profiles from the TG-GATEs database. -

Genome Sequence, Population History, and Pelage Genetics of the Endangered African Wild Dog (Lycaon Pictus) Michael G
Campana et al. BMC Genomics (2016) 17:1013 DOI 10.1186/s12864-016-3368-9 RESEARCH ARTICLE Open Access Genome sequence, population history, and pelage genetics of the endangered African wild dog (Lycaon pictus) Michael G. Campana1,2*, Lillian D. Parker1,2,3, Melissa T. R. Hawkins1,2,3, Hillary S. Young4, Kristofer M. Helgen2,3, Micaela Szykman Gunther5, Rosie Woodroffe6, Jesús E. Maldonado1,2 and Robert C. Fleischer1 Abstract Background: The African wild dog (Lycaon pictus) is an endangered African canid threatened by severe habitat fragmentation, human-wildlife conflict, and infectious disease. A highly specialized carnivore, it is distinguished by its social structure, dental morphology, absence of dewclaws, and colorful pelage. Results: We sequenced the genomes of two individuals from populations representing two distinct ecological histories (Laikipia County, Kenya and KwaZulu-Natal Province, South Africa). We reconstructed population demographic histories for the two individuals and scanned the genomes for evidence of selection. Conclusions: We show that the African wild dog has undergone at least two effective population size reductions in the last 1,000,000 years. We found evidence of Lycaon individual-specific regions of low diversity, suggestive of inbreeding or population-specific selection. Further research is needed to clarify whether these population reductions and low diversity regions are characteristic of the species as a whole. We documented positive selection on the Lycaon mitochondrial genome. Finally, we identified several candidate genes (ASIP, MITF, MLPH, PMEL) that may play a role in the characteristic Lycaon pelage. Keywords: Lycaon pictus, Genome, Population history, Selection, Pelage Background Primarily a hunter of antelopes, the African wild dog is a The African wild dog (Lycaon pictus) is an endangered highly distinct canine. -

Broad Poster Vivek
A novel computational method for finding regions with copy number abnormalities in cancer cells Vivek, Manuel Garber, and Mike Zody Broad Institute of MIT and Harvard, Cambridge, MA, USA Introduction Results Cancer can result from the over expression of oncogenes, genes which control and regulate cell growth. Sometimes oncogenes increase in 1 2 3 activity due to a specific genetic mutation called a translocation (Fig 1). SMAD4 – a gene known to be deleted in pancreatic COX10 – a gene deleted in cytochrome c oxidase AK001392 – a hereditary prostate cancer protein This translocation allows the oncogene to remain as active as its paired carcinoma deficiency, known to be related to cell proliferation gene. Amplification of this mutation can occur, thereby creating the proper conditions for uncontrolled cell growth; consequently, each Results from Analysis Program Results from Analysis Program Results from Analysis Program component of the translocation will amplify in similar quantities. In this mutation, the chromosomal region containing the oncogene displaces to Region 1 Region 2 R2 Region 1 Region 2 R2 Region 1 Region 2 R2 a region on another chromosome containing a gene that is expressed Chr18:47044749-47311978 Chr17:13930739-14654741 0.499070821478475 Chr17:13930739-14654741 Chr18:26861790-27072166 0.47355172850856 Chr17:12542326-13930738 Chr8:1789292-1801984 0.406208680312004 frequently. Actual region containing gene Actual region containing gene Actual region containing gene chr18: 45,842,214 - 48,514,513 chr17: 13,966,862 - 14,068,461 chr17: 12,542,326 - 13,930,738 Fig 1. Two chromosomal regions (abcdef and ghijk) are translocating to create two new regions (abckl and ghijedf). -

The Role of RNA Editing in Cancer Development and Metabolic Disorders
Washington University School of Medicine Digital Commons@Becker Open Access Publications 2018 The oler of RNA editing in cancer development and metabolic disorders Che-Pei Kung Washington University School of Medicine in St. Louis Leonard B. Maggi Jr. Washington University School of Medicine in St. Louis Jason D. Weber Washington University School of Medicine in St. Louis Follow this and additional works at: https://digitalcommons.wustl.edu/open_access_pubs Recommended Citation Kung, Che-Pei; Maggi, Leonard B. Jr.; and Weber, Jason D., ,"The or le of RNA editing in cancer development and metabolic disorders." Frontiers in endocrinology.9,. 762. (2018). https://digitalcommons.wustl.edu/open_access_pubs/7400 This Open Access Publication is brought to you for free and open access by Digital Commons@Becker. It has been accepted for inclusion in Open Access Publications by an authorized administrator of Digital Commons@Becker. For more information, please contact [email protected]. REVIEW published: 18 December 2018 doi: 10.3389/fendo.2018.00762 The Role of RNA Editing in Cancer Development and Metabolic Disorders Che-Pei Kung 1,2*, Leonard B. Maggi Jr. 1,2 and Jason D. Weber 1,2,3* 1 ICCE Institute, Washington University School of Medicine, Saint Louis, MO, United States, 2 Division of Molecular Oncology, Department of Medicine, Washington University School of Medicine, Saint Louis, MO, United States, 3 Siteman Cancer Center, Department of Cell Biology and Physiology, Washington University School of Medicine, Saint Louis, MO, United States Numerous human diseases arise from alterations of genetic information, most notably DNA mutations. Thought to be merely the intermediate between DNA and protein, changes in RNA sequence were an afterthought until the discovery of RNA editing 30 years ago. -

A Computational Approach for Defining a Signature of Β-Cell Golgi Stress in Diabetes Mellitus
Page 1 of 781 Diabetes A Computational Approach for Defining a Signature of β-Cell Golgi Stress in Diabetes Mellitus Robert N. Bone1,6,7, Olufunmilola Oyebamiji2, Sayali Talware2, Sharmila Selvaraj2, Preethi Krishnan3,6, Farooq Syed1,6,7, Huanmei Wu2, Carmella Evans-Molina 1,3,4,5,6,7,8* Departments of 1Pediatrics, 3Medicine, 4Anatomy, Cell Biology & Physiology, 5Biochemistry & Molecular Biology, the 6Center for Diabetes & Metabolic Diseases, and the 7Herman B. Wells Center for Pediatric Research, Indiana University School of Medicine, Indianapolis, IN 46202; 2Department of BioHealth Informatics, Indiana University-Purdue University Indianapolis, Indianapolis, IN, 46202; 8Roudebush VA Medical Center, Indianapolis, IN 46202. *Corresponding Author(s): Carmella Evans-Molina, MD, PhD ([email protected]) Indiana University School of Medicine, 635 Barnhill Drive, MS 2031A, Indianapolis, IN 46202, Telephone: (317) 274-4145, Fax (317) 274-4107 Running Title: Golgi Stress Response in Diabetes Word Count: 4358 Number of Figures: 6 Keywords: Golgi apparatus stress, Islets, β cell, Type 1 diabetes, Type 2 diabetes 1 Diabetes Publish Ahead of Print, published online August 20, 2020 Diabetes Page 2 of 781 ABSTRACT The Golgi apparatus (GA) is an important site of insulin processing and granule maturation, but whether GA organelle dysfunction and GA stress are present in the diabetic β-cell has not been tested. We utilized an informatics-based approach to develop a transcriptional signature of β-cell GA stress using existing RNA sequencing and microarray datasets generated using human islets from donors with diabetes and islets where type 1(T1D) and type 2 diabetes (T2D) had been modeled ex vivo. To narrow our results to GA-specific genes, we applied a filter set of 1,030 genes accepted as GA associated. -

To Study Mutant P53 Gain of Function, Various Tumor-Derived P53 Mutants
Differential effects of mutant TAp63γ on transactivation of p53 and/or p63 responsive genes and their effects on global gene expression. A thesis submitted in partial fulfillment of the requirements for the degree of Master of Science By Shama K Khokhar M.Sc., Bilaspur University, 2004 B.Sc., Bhopal University, 2002 2007 1 COPYRIGHT SHAMA K KHOKHAR 2007 2 WRIGHT STATE UNIVERSITY SCHOOL OF GRADUATE STUDIES Date of Defense: 12-03-07 I HEREBY RECOMMEND THAT THE THESIS PREPARED UNDER MY SUPERVISION BY SHAMA KHAN KHOKHAR ENTITLED Differential effects of mutant TAp63γ on transactivation of p53 and/or p63 responsive genes and their effects on global gene expression BE ACCEPTED IN PARTIAL FULFILLMENT OF THE REQUIREMENTS FOR THE DEGREE OF Master of Science Madhavi P. Kadakia, Ph.D. Thesis Director Daniel Organisciak , Ph.D. Department Chair Committee on Final Examination Madhavi P. Kadakia, Ph.D. Steven J. Berberich, Ph.D. Michael Leffak, Ph.D. Joseph F. Thomas, Jr., Ph.D. Dean, School of Graduate Studies 3 Abstract Khokhar, Shama K. M.S., Department of Biochemistry and Molecular Biology, Wright State University, 2007 Differential effect of TAp63γ mutants on transactivation of p53 and/or p63 responsive genes and their effects on global gene expression. p63, a member of the p53 gene family, known to play a role in development, has more recently also been implicated in cancer progression. Mice lacking p63 exhibit severe developmental defects such as limb truncations, abnormal skin, and absence of hair follicles, teeth, and mammary glands. Germline missense mutations of p63 have been shown to be responsible for several human developmental syndromes including SHFM, EEC and ADULT syndromes and are associated with anomalies in the development of organs of epithelial origin. -

32-12012: Human Beta Defensin-3 Description Product
9853 Pacific Heights Blvd. Suite D. San Diego, CA 92121, USA Tel: 858-263-4982 Email: [email protected] 32-12012: Human Beta Defensin-3 Gene : DEFB103A Gene ID : 414325 Uniprot ID : P81534 Alternative Name : DEFB-3, Beta-defensin 3, Defensin, beta 103, Defensin-like protein Description Source: Genetically modified E.coli. Predicted MW: Monomer, 5.2 kDa (45 aa) Beta-Defensin 3 (BD-3), also known as DEFB-3, is a member of the defensin class of antimicrobial peptides. Beta defensins exert host defense responses against viruses, bacteria, and fungi through the binding and permeabilizing of microbial membranes. BD-3 expression is stimulated by interferon-gamma and is an important molecule during adaptive immunity. BD-3 functions to activate monocytes and mast cells, and has antibacterial functions towards Gram-negative and Gram-positive bacteria. Further, BD-3 blocks human immunodeficiency virus type 1 (HIV-1) replication through the downregulation of the HIV-1 co-receptor, CXCR4. Product Info Amount : 20 µg / 100 µg Purification : Reducing and Non-Reducing SDS PAGE at >= 95% Lyophilized from a sterile (0.2 micron) filtered aqueous solution containing 0.1% Trifluoroacetic Content : Acid (TFA) Sterile water at 0.1 mg/mL Storage condition : Store at -20°C Amino Acid : GIINTLQKYY CRVRGGRCAV LSCLPKEEQI GKCSTRGRKC CRRKK Application Note Endotoxin: Less than 0.1 ng/µg (1 IEU/µg) as determined by LAL test. Centrifuge vial before opening, Suspend the product by gently pipetting the above recommended solution down the sides of the vial. DO NOT VORTEX. Allow several minutes for complete reconstitution. For prolonged storage, dilute to working aliquots in a 0.1% BSA solution, store at -80°C and avoid repeat freeze thaws. -

Mohammad Karbaschi Thesis
STRUCTURAL, PHYSIOLOGICAL AND MOLECULAR CHARACTERISATION OF THE AUSTRALIAN NATIVE RESURRECTION GRASS TRIPOGON LOLIIFORMIS (F.MUELL.) C.E.HUBB. DURING DEHYDRATION AND REHYDRATION Mohammad Reza Karbaschi Submitted in fulfilment of the requirements for the degree of Doctor of Philosophy Centre for Tropical Crops and Biocommodities Science and Engineering Faculty Queensland University of Technology November 2015 Keywords Arabidopsis thaliana; Agrobacterium-mediated transformation; Anatomy; Anti-apoptotic proteins; BAG4; Escherichia coli; Bulliform cells; C4 photosynthesis; Cell wall folding; Cell membrane integrity; Chaperone-mediated autophagy; Chlorophyll fluorescence; Hsc70/Hsp70; Desiccation tolerance, Dehydration; Drought; Electrolyte leakage; Freehand sectioning; Homoiochlorophyllous; Leaf structure; Leaf folding; Reactive oxygen species (ROS); Resurrection plant; Morphology; Monocotyledon; Nicotiana benthamiana; Photosynthesis; Physiology; Plant tissue; Programed cell death (PCD); Propidium iodide staining; Protein microarray chip; Sclerenchymatous tissue; Stress; Structure; Tripogon loliiformis; Ubiquitin; Vacuole fragmentation; Kranz anatomy; XyMS+; Structural, physiological and molecular characterisation of the Australian native resurrection grass Tripogon loliiformis (F.Muell.) C.E.Hubb. during dehydration and rehydration i Abstract Plants, as sessile organisms must continually adapt to environmental changes. Water deficit is one of the major environmental stresses that affects plants. While most plants can tolerate moderate dehydration -
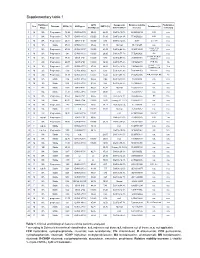
Supplementary Table 2 Supplementary Table 1
Supplementary table 1 Rai/ Binet IGHV Cytogenetic Relative viability Fludarabine- Sex Outcome CD38 (%) IGHV gene ZAP70 (%) Treatment (s) Stage identity (%) abnormalities* increase refractory 1 M 0/A Progressive 14,90 IGHV3-64*05 99,65 28,20 Del17p 18.0% 62,58322819 FCR n.a. 2 F 0/A Progressive 78,77 IGHV3-48*03 100,00 51,90 Del17p 24.8% 77,88052021 FCR n.a. 3 M 0/A Progressive 29,81 IGHV4-b*01 100,00 9,10 Del17p 12.0% 36,48 Len, Chl n.a. 4 M 1/A Stable 97,04 IGHV3-21*01 97,22 18,11 Normal 85,4191657 n.a. n.a. Chl+O, PCR, 5 F 0/A Progressive 87,00 IGHV4-39*07 100,00 43,20 Del13q 68.3% 35,23314039 n.a. HDMP+R 6 M 0/A Progressive 1,81 IGHV3-43*01 100,00 20,90 Del13q 77.7% 57,52490626 Chl n.a. Chl, FR, R-CHOP, 7 M 0/A Progressive 97,80 IGHV1-3*01 100,00 9,80 Del17p 88.5% 48,57389901 n.a. HDMP+R 8 F 2/B Progressive 69,07 IGHV5-a*03 100,00 16,50 Del17p 77.2% 107,9656878 FCR, BA No R-CHOP, FCR, 9 M 1/A Progressive 2,13 IGHV3-23*01 97,22 29,80 Del11q 16.3% 134,5866919 Yes Flavopiridol, BA 10 M 2/A Progressive 0,36 IGHV3-30*02 92,01 0,38 Del13q 81.9% 78,91844953 Unknown n.a. 11 M 2/B Progressive 15,17 IGHV3-20*01 100,00 13,20 Del11q 95.3% 75,52880995 FCR, R-CHOP, BR No 12 M 0/A Stable 0,14 IGHV3-30*02 90,62 7,40 Del13q 13.0% 13,0939004 n.a. -

Supplementary Data
Supplementary Methods Mutation and microdeletion screening by high resolution melting High-throughput mutation screening of DIS3L2 exons 1-16 and HDAC4 was performed by Lightscanner high resolution melting analysis (Idaho Technology, Salt Lake City, UT). Exons 17-21 of DIS3L2 were not sequenced due to an apparent genomic duplication and consequent inability to uniquely amplify these exons. DNA samples were amplified using LightScanner mastermix under the manufacturer’s guidelines (Idaho Technology). After PCR, samples were heated at 0.1°C/s in the Lightscanner instrument and fluorescence was collected from 60 to 95°C. Melting curves were analyzed using LightScanner software (v2.0, Idaho Technology). Microdeletion screening across DIS3L2 was performed on paired normal- tumor samples using Lightscanner Lunaprobe SNP genotyping. Seven SNPs, ~60 Kb apart (rs2679184, rs12988522, rs4973500, rs3100586, rs3116179, rs923333 and rs2633254) were amplified in separate reactions and analyzed as above. Detailed conditions and primer/probe sequences are available on request. Variant amplicons were sequenced as described below. Direct sequencing was also performed for all exons where a common polymorphism might mask detection of a mutation by Lightscanner. Samples with known LOH were analyzed entirely by direct sequencing, since Lightscanner detects altered melting profiles of DNA heteroduplexes and these cannot exist in hemizygous samples. Sequencing of candidate genes Direct sequencing of exons and flanking consensus splice signals was performed for DIS3L2, GIGYF2, NPPC, HDAC4, TWIST2 and miR-562. PCR amplification was performed using HotStarTaq Mastermix and Q solution (Qiagen, Valencia, CA); all conditions and primers are available on request. PCR products were treated with shrimp alkaline phosphatase and exonuclease-I (New England Biolabs, Ipswich, MA) and sequenced using BigDye terminator chemistry on a 3730xl sequencer (Applied Biosystems, Foster City, CA). -

Α7 Nicotinic Receptor Up-Regulation in Cholinergic Basal Forebrain Neurons in Alzheimer Disease
ORIGINAL CONTRIBUTION ␣7 Nicotinic Receptor Up-regulation in Cholinergic Basal Forebrain Neurons in Alzheimer Disease Scott E. Counts, PhD; Bin He, MD; Shaoli Che, MD, PhD; Milos D. Ikonomovic, MD; Steven T. DeKosky, MD; Stephen D. Ginsberg, PhD; Elliott J. Mufson, PhD Background: Dysfunction of basocortical cholinergic pro- Participants: Participants were members of the Rush jection neurons of the nucleus basalis (NB) correlates with Religious Orders Study cohort. cognitive deficits in Alzheimer disease (AD). Nucleus ba- Main Outcome Measures: Real-time quantitative poly- salis neurons receive cholinergic inputs and express nico- merase chain reaction was performed to validate micro- tinic acetylcholine receptors (nAChRs) and muscarinic array findings. AChRs (mAChRs), which may regulate NB neuron activ- ity in AD. Although alterations in these AChRs occur in Results: Cholinergic NB neurons displayed a statisti- the AD cortex, there is little information detailing whether cally significant up-regulation of ␣7 nAChR messenger defects in nAChR and mAChR gene expression occur in RNA expression in subjects with mild to moderate AD cholinergic NB neurons during disease progression. compared with those with NCI and MCI (PϽ.001). No differences were found for other nAChR and mAChR sub- types across the cohort. Expression levels of ␣7 nAChRs Objective: To determine whether nAChR and mAChR were inversely associated with Global Cognitive Score and gene expression is altered in cholinergic NB neurons dur- with Mini-Mental State Examination performance. ing the progression of AD. Conclusions: Up-regulation of ␣7 nAChRs may signal Design: Individual NB neurons from subjects diag- a compensatory response to maintain basocortical cho- nosed ante mortem as having no cognitive impairment linergic activity during AD progression.