The Radiochemistry of Protactinium Which Might Be Included in a Revised Version of the Monograph
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Table 2.Iii.1. Fissionable Isotopes1
FISSIONABLE ISOTOPES Charles P. Blair Last revised: 2012 “While several isotopes are theoretically fissionable, RANNSAD defines fissionable isotopes as either uranium-233 or 235; plutonium 238, 239, 240, 241, or 242, or Americium-241. See, Ackerman, Asal, Bale, Blair and Rethemeyer, Anatomizing Radiological and Nuclear Non-State Adversaries: Identifying the Adversary, p. 99-101, footnote #10, TABLE 2.III.1. FISSIONABLE ISOTOPES1 Isotope Availability Possible Fission Bare Critical Weapon-types mass2 Uranium-233 MEDIUM: DOE reportedly stores Gun-type or implosion-type 15 kg more than one metric ton of U- 233.3 Uranium-235 HIGH: As of 2007, 1700 metric Gun-type or implosion-type 50 kg tons of HEU existed globally, in both civilian and military stocks.4 Plutonium- HIGH: A separated global stock of Implosion 10 kg 238 plutonium, both civilian and military, of over 500 tons.5 Implosion 10 kg Plutonium- Produced in military and civilian 239 reactor fuels. Typically, reactor Plutonium- grade plutonium (RGP) consists Implosion 40 kg 240 of roughly 60 percent plutonium- Plutonium- 239, 25 percent plutonium-240, Implosion 10-13 kg nine percent plutonium-241, five 241 percent plutonium-242 and one Plutonium- percent plutonium-2386 (these Implosion 89 -100 kg 242 percentages are influenced by how long the fuel is irradiated in the reactor).7 1 This table is drawn, in part, from Charles P. Blair, “Jihadists and Nuclear Weapons,” in Gary A. Ackerman and Jeremy Tamsett, ed., Jihadists and Weapons of Mass Destruction: A Growing Threat (New York: Taylor and Francis, 2009), pp. 196-197. See also, David Albright N 2 “Bare critical mass” refers to the absence of an initiator or a reflector. -

An Introduction to Isotopic Calculations John M
An Introduction to Isotopic Calculations John M. Hayes ([email protected]) Woods Hole Oceanographic Institution, Woods Hole, MA 02543, USA, 30 September 2004 Abstract. These notes provide an introduction to: termed isotope effects. As a result of such effects, the • Methods for the expression of isotopic abundances, natural abundances of the stable isotopes of practically • Isotopic mass balances, and all elements involved in low-temperature geochemical • Isotope effects and their consequences in open and (< 200°C) and biological processes are not precisely con- closed systems. stant. Taking carbon as an example, the range of interest is roughly 0.00998 ≤ 13F ≤ 0.01121. Within that range, Notation. Absolute abundances of isotopes are com- differences as small as 0.00001 can provide information monly reported in terms of atom percent. For example, about the source of the carbon and about processes in 13 13 12 13 atom percent C = [ C/( C + C)]100 (1) which the carbon has participated. A closely related term is the fractional abundance The delta notation. Because the interesting isotopic 13 13 fractional abundance of C ≡ F differences between natural samples usually occur at and 13F = 13C/(12C + 13C) (2) beyond the third significant figure of the isotope ratio, it has become conventional to express isotopic abundances These variables deserve attention because they provide using a differential notation. To provide a concrete the only basis for perfectly accurate mass balances. example, it is far easier to say – and to remember – that Isotope ratios are also measures of the absolute abun- the isotope ratios of samples A and B differ by one part dance of isotopes; they are usually arranged so that the per thousand than to say that sample A has 0.3663 %15N more abundant isotope appears in the denominator and sample B has 0.3659 %15N. -

The Development of the Periodic Table and Its Consequences Citation: J
Firenze University Press www.fupress.com/substantia The Development of the Periodic Table and its Consequences Citation: J. Emsley (2019) The Devel- opment of the Periodic Table and its Consequences. Substantia 3(2) Suppl. 5: 15-27. doi: 10.13128/Substantia-297 John Emsley Copyright: © 2019 J. Emsley. This is Alameda Lodge, 23a Alameda Road, Ampthill, MK45 2LA, UK an open access, peer-reviewed article E-mail: [email protected] published by Firenze University Press (http://www.fupress.com/substantia) and distributed under the terms of the Abstract. Chemistry is fortunate among the sciences in having an icon that is instant- Creative Commons Attribution License, ly recognisable around the world: the periodic table. The United Nations has deemed which permits unrestricted use, distri- 2019 to be the International Year of the Periodic Table, in commemoration of the 150th bution, and reproduction in any medi- anniversary of the first paper in which it appeared. That had been written by a Russian um, provided the original author and chemist, Dmitri Mendeleev, and was published in May 1869. Since then, there have source are credited. been many versions of the table, but one format has come to be the most widely used Data Availability Statement: All rel- and is to be seen everywhere. The route to this preferred form of the table makes an evant data are within the paper and its interesting story. Supporting Information files. Keywords. Periodic table, Mendeleev, Newlands, Deming, Seaborg. Competing Interests: The Author(s) declare(s) no conflict of interest. INTRODUCTION There are hundreds of periodic tables but the one that is widely repro- duced has the approval of the International Union of Pure and Applied Chemistry (IUPAC) and is shown in Fig.1. -

Toxicological Profile for Plutonium
PLUTONIUM 1 1. PUBLIC HEALTH STATEMENT This public health statement tells you about plutonium and the effects of exposure to it. The Environmental Protection Agency (EPA) identifies the most serious hazardous waste sites in the nation. These sites are then placed on the National Priorities List (NPL) and are targeted for long-term federal clean-up activities. Plutonium has been found in at least 16 of the 1,689 current or former NPL sites. Although the total number of NPL sites evaluated for this substance is not known, strict regulations make it unlikely that the number of sites at which plutonium is found would increase in the future as more sites are evaluated. This information is important because these sites may be sources of exposure and exposure to this substance may be harmful. When a substance is released from a large area, such as an industrial plant, or from a container, such as a drum or bottle, it enters the environment. This release does not always lead to exposure. You are normally exposed to a substance only when you come in contact with it. You may be exposed by breathing, eating, or drinking the substance, or by skin contact. However, since plutonium is radioactive, you can also be exposed to its radiation if you are near it. External exposure to radiation may occur from natural or man-made sources. Naturally occurring sources of radiation are cosmic radiation from space or radioactive materials in soil or building materials. Man- made sources of radioactive materials are found in consumer products, industrial equipment, atom bomb fallout, and to a smaller extent from hospital waste and nuclear reactors. -

Chromium-Nickel Steels Depleted of Nickel Stable Isotope Ni-58 As a Material for Fast Reactor Claddings
IAEA-CN-114/15p Chromium-Nickel steels depleted of nickel stable isotope Ni-58 as a material for fast reactor claddings G.L.Khorasanov, A.P.Ivanov, A.I.Blokhin, N.A.Demin Institute of Physics and Power Engineering named after A.I.Leypunsky 1, Bondarenko square, Obninsk, Kaluga region, 249033 Russia Tel.: +7-08439-98505, Fax: +7-095-2302326, E-mail: [email protected] INTRODUCTION Presently, the creation of new materials for fast reactor (FR) claddings is one of the main tasks of the nuclear engineering [1]. Now the Russian austenitic steel grade TchS68 with 15 % of nickel content is used as a material for the Russian FR BN-600 claddings [2]. With such a cladding the BN-600 fuel rod is stably operating during 10 years up to the fuel burn-up fraction of 10 % of heavy atoms (h.a.). The cladding material becomes swelled and unfit for further service after the fuel burn-up fraction of 10 % h.a. The task of increasing the fuel burn-up fraction up to 14 % h.a. is now the principal one for the BN-600 operation. For this purpose the new radiation–resistant austenitic steel grade EK164 with 19 % of nickel content is now developing in the Bochvar Institute, Russia [3]. It is generally assumed that steel swelling is caused by the combined action of radiation damage and helium accumulation in the irradiated material. In this case, nickel stable isotope, Ni-58 (its content in natural nickel is equal to 68%) is a source of helium creation. After 4 neutron capture via (n,g) reaction, Ni-58 is transmuted to radioisotope Ni-59 (T1/2=7.6⋅10 years) which has a high value of (n,α) reaction cross-section for thermal and epithermal neutrons. -

Lithium Isotope Fractionation During Uptake by Gibbsite
Available online at www.sciencedirect.com ScienceDirect Geochimica et Cosmochimica Acta 168 (2015) 133–150 www.elsevier.com/locate/gca Lithium isotope fractionation during uptake by gibbsite Josh Wimpenny a,⇑, Christopher A. Colla a, Ping Yu b, Qing-Zhu Yin a, James R. Rustad a, William H. Casey a,c a Department of Earth and Planetary Sciences, University of California, One Shields Avenue, Davis, CA 95616, United States b The Keck NMR Facility, University of California, One Shields Avenue, Davis, CA 95616, United States c Department of Chemistry, University of California, One Shields Avenue, Davis, CA 95616, United States Received 5 November 2014; accepted in revised form 8 July 2015; available online 15 July 2015 Abstract The intercalation of lithium from solution into the six-membered l2-oxo rings on the basal planes of gibbsite is well-constrained chemically. The product is a lithiated layered-double hydroxide solid that forms via in situ phase change. The reaction has well established kinetics and is associated with a distinct swelling of the gibbsite as counter ions enter the interlayer to balance the charge of lithiation. Lithium reacts to fill a fixed and well identifiable crystallographic site and has no solvation waters. Our lithium-isotope data shows that 6Li is favored during this intercalation and that the À À solid-solution fractionation depends on temperature, electrolyte concentration and counter ion identity (whether Cl ,NO3 À or ClO4 ). We find that the amount of isotopic fractionation between solid and solution (DLisolid-solution) varies with the amount of lithium taken up into the gibbsite structure, which itself depends upon the extent of conversion and also varies À À À with electrolyte concentration and in the counter ion in the order: ClO4 <NO3 <Cl . -

Isotopic Composition of Fission Gases in Lwr Fuel
XA0056233 ISOTOPIC COMPOSITION OF FISSION GASES IN LWR FUEL T. JONSSON Studsvik Nuclear AB, Hot Cell Laboratory, Nykoping, Sweden Abstract Many fuel rods from power reactors and test reactors have been punctured during past years for determination of fission gas release. In many cases the released gas was also analysed by mass spectrometry. The isotopic composition shows systematic variations between different rods, which are much larger than the uncertainties in the analysis. This paper discusses some possibilities and problems with use of the isotopic composition to decide from which part of the fuel the gas was released. In high burnup fuel from thermal reactors loaded with uranium fuel a significant part of the fissions occur in plutonium isotopes. The ratio Xe/Kr generated in the fuel is strongly dependent on the fissioning species. In addition, the isotopic composition of Kr and Xe shows a well detectable difference between fissions in different fissile nuclides. 1. INTRODUCTION Most LWRs use low enriched uranium oxide as fuel. Thermal fissions in U-235 dominate during the earlier part of the irradiation. Due to the build-up of heavier actinides during the irradiation fissions in Pu-239 and Pu-241 increase in importance as the burnup of the fuel increases. The composition of the fission products varies with the composition of the fuel and the irradiation conditions. The isotopic composition of fission gases is often determined in connection with measurement of gases in the plenum of punctured fuel rods. It can be of interest to discuss how more information on the fuel behaviour can be obtained by use of information available from already performed determinations of gas compositions. -

Rapid Separations of Protactinium and Uranium Radioisotopes From
754 LETTERS TO THE EDITOR may be as small as one micron in diameter. Such particles are solutions with ether were required to further eliminate non products of breakdown or are introduced mto the enclosed system volatile foreign material which would extract into the ether. at the time of assembly. If special precautions were not taken to Ignition of the ether sample gave a weightless plate of the uranium eliminate stray particles, the subnormal breakdown strengths isotopes. obtained would lay within ±1O percent of a mean value which Approximate times required for the steps were: solution, for a given method of electrode polishing would be repeatable. extraction, and separation 10-20 seconds; washing 10-15 seconds; These mean values, reported earlier,! are in agreement with the and making of sample plate ("-'1 cc ether) ",15 seconds. results published by other workers. In nuclear chemical work where the main interest is in charac Experiments are continuing to determine whether the maximum terizing particular isotopes, emphasis is on radiochemical purity values reported here are indeed the intrinsic electric strengths of of the isotope rather than on chemical yield. The above procedures the liquids concerned. give only a 10-20 percent chemical yield-fa of which can be The author is indebted to the National Research Council of rapidly placed on a plate for counting, These yields are quite Canada for the award of a Fellowship which enabled him to carry satisfactory for the purpose, however, since contaminating isotopes out this work. have been reduced by a factor of 104 to lOS. Experimental results have shown that about 1 part in 104 of actinium, 1 part in lOS of 1 W. -

Periodic Table 1 Periodic Table
Periodic table 1 Periodic table This article is about the table used in chemistry. For other uses, see Periodic table (disambiguation). The periodic table is a tabular arrangement of the chemical elements, organized on the basis of their atomic numbers (numbers of protons in the nucleus), electron configurations , and recurring chemical properties. Elements are presented in order of increasing atomic number, which is typically listed with the chemical symbol in each box. The standard form of the table consists of a grid of elements laid out in 18 columns and 7 Standard 18-column form of the periodic table. For the color legend, see section Layout, rows, with a double row of elements under the larger table. below that. The table can also be deconstructed into four rectangular blocks: the s-block to the left, the p-block to the right, the d-block in the middle, and the f-block below that. The rows of the table are called periods; the columns are called groups, with some of these having names such as halogens or noble gases. Since, by definition, a periodic table incorporates recurring trends, any such table can be used to derive relationships between the properties of the elements and predict the properties of new, yet to be discovered or synthesized, elements. As a result, a periodic table—whether in the standard form or some other variant—provides a useful framework for analyzing chemical behavior, and such tables are widely used in chemistry and other sciences. Although precursors exist, Dmitri Mendeleev is generally credited with the publication, in 1869, of the first widely recognized periodic table. -

Highly Enriched Uranium: Striking a Balance
OFFICIAL USE ONLY - DRAFT GLOSSARY OF TERMS APPENDIX F GLOSSARY OF TERMS Accountability: That part of the safeguards and security program that encompasses the measurement and inventory verification systems, records, and reports to account for nuclear materials. Assay: Measurement that establishes the total quantity of the isotope of an element and the total quantity of that element. Atom: The basic component of all matter. Atoms are the smallest part of an element that have all of the chemical properties of that element. Atoms consist of a nucleus of protons and neutrons surrounded by electrons. Atomic energy: All forms of energy released in the course of nuclear fission or nuclear transformation. Atomic weapon: Any device utilizing atomic energy, exclusive of the means for transportation or propelling the device (where such means is a separable and divisible part of the device), the principal purpose of which is for use as, or for development of, a weapon, a weapon prototype, or a weapon test device. Blending: The intentional mixing of two different assays of the same material in order to achieve a desired third assay. Book inventory: The quantity of nuclear material present at a given time as reflected by accounting records. Burnup: A measure of consumption of fissionable material in reactor fuel. Burnup can be expressed as (a) the percentage of fissionable atoms that have undergone fission or capture, or (b) the amount of energy produced per unit weight of fuel in the reactor. Chain reaction: A self-sustaining series of nuclear fission reactions. Neutrons produced by fission cause more fission. Chain reactions are essential to the functioning of nuclear reactors and weapons. -

Mass Fraction and the Isotopic Anomalies of Xenon and Krypton in Ordinary Chondrites
Scholars' Mine Masters Theses Student Theses and Dissertations 1971 Mass fraction and the isotopic anomalies of xenon and krypton in ordinary chondrites Edward W. Hennecke Follow this and additional works at: https://scholarsmine.mst.edu/masters_theses Part of the Chemistry Commons Department: Recommended Citation Hennecke, Edward W., "Mass fraction and the isotopic anomalies of xenon and krypton in ordinary chondrites" (1971). Masters Theses. 5453. https://scholarsmine.mst.edu/masters_theses/5453 This thesis is brought to you by Scholars' Mine, a service of the Missouri S&T Library and Learning Resources. This work is protected by U. S. Copyright Law. Unauthorized use including reproduction for redistribution requires the permission of the copyright holder. For more information, please contact [email protected]. MASS FRACTIONATION AND THE ISOTOPIC ANOMALIES OF XENON AND KRYPTON IN ORDINARY CHONDRITES BY EDWARD WILLIAM HENNECKE, 1945- A THESIS Presented to the Faculty of the Graduate School of the UNIVERSITY OF MISSOURI-ROLLA In Partial Fulfillment of the Requirements for the Degree MASTER OF SCIENCE IN CHEMISTRY 1971 T2572 51 pages by Approved ~ (!.{ 1.94250 ii ABSTRACT The abundance and isotopic composition of all noble gases are reported in the Wellman chondrite, and the abundance and isotopic composition of xenon and krypton are reported in the gases released by stepwise heating of the Tell and Scurry chondrites. Major changes in the isotopic composition of xenon result from the presence of radio genic Xel29 and from isotopic mass fractionation. The isotopic com position of trapped krypton in the different temperature fractions of the Tell and Scurry chondrites also shows the effect of isotopic fractiona tion, and there is a covariance in the isotopic composition of xenon with krypton in the manner expected from mass dependent fractiona tion. -
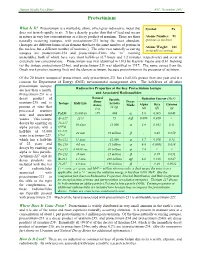
Protactinium
Human Health Fact Sheet ANL, November 2001 Protactinium What Is It? Protactinium is a malleable, shiny, silver-gray radioactive metal that Symbol: Pa does not tarnish rapidly in air. It has a density greater than that of lead and occurs in nature in very low concentrations as a decay product of uranium. There are three Atomic Number: 91 naturally occurring isotopes, with protactinium-231 being the most abundant. (protons in nucleus) (Isotopes are different forms of an element that have the same number of protons in Atomic Weight: 231 the nucleus but a different number of neutrons.) The other two naturally occurring (naturally occurring) isotopes are protactinium-234 and protactinium-234m (the “m” meaning metastable), both of which have very short half-lives (6.7 hours and 1.2 minutes, respectively) and occur in extremely low concentrations. Protactinium was first identified in 1913 by Kasimir Fajans and O.H. Gohring (as the isotope protactinium-234m), and protactinium-231 was identified in 1917. The name comes from the Greek work protos (meaning first) and the element actinium, because protactinium is the precursor of actinium. Of the 20 known isotopes of protactinium, only protactinium-231 has a half-life greater than one year and is a concern for Department of Energy (DOE) environmental management sites. The half-lives of all other protactinium isotopes Radioactive Properties of the Key Protactinium Isotope are less than a month. Protactinium-231 is a and Associated Radionuclides Natural decay product of Specific Radiation Energy (MeV) Abun- Decay uranium-235 and is Isotope Half-Life Activity dance Mode Alpha Beta Gamma present at sites that (Ci/g) (%) (α) (β) (γ) processed uranium α ores and associated Pa231 33,000 yr >99 .048 5.0 0.065 0.048 wastes.