(2007): 831 [PD Cantino , JA Doyle, and MJ D
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Approved Plant List 10/04/12
FLORIDA The best time to plant a tree is 20 years ago, the second best time to plant a tree is today. City of Sunrise Approved Plant List 10/04/12 Appendix A 10/4/12 APPROVED PLANT LIST FOR SINGLE FAMILY HOMES SG xx Slow Growing “xx” = minimum height in Small Mature tree height of less than 20 feet at time of planting feet OH Trees adjacent to overhead power lines Medium Mature tree height of between 21 – 40 feet U Trees within Utility Easements Large Mature tree height greater than 41 N Not acceptable for use as a replacement feet * Native Florida Species Varies Mature tree height depends on variety Mature size information based on Betrock’s Florida Landscape Plants Published 2001 GROUP “A” TREES Common Name Botanical Name Uses Mature Tree Size Avocado Persea Americana L Bahama Strongbark Bourreria orata * U, SG 6 S Bald Cypress Taxodium distichum * L Black Olive Shady Bucida buceras ‘Shady Lady’ L Lady Black Olive Bucida buceras L Brazil Beautyleaf Calophyllum brasiliense L Blolly Guapira discolor* M Bridalveil Tree Caesalpinia granadillo M Bulnesia Bulnesia arboria M Cinnecord Acacia choriophylla * U, SG 6 S Group ‘A’ Plant List for Single Family Homes Common Name Botanical Name Uses Mature Tree Size Citrus: Lemon, Citrus spp. OH S (except orange, Lime ect. Grapefruit) Citrus: Grapefruit Citrus paradisi M Trees Copperpod Peltophorum pterocarpum L Fiddlewood Citharexylum fruticosum * U, SG 8 S Floss Silk Tree Chorisia speciosa L Golden – Shower Cassia fistula L Green Buttonwood Conocarpus erectus * L Gumbo Limbo Bursera simaruba * L -

The Lower Devonian Water Canyon Formation of Northeastern Utah
Utah State University DigitalCommons@USU All Graduate Theses and Dissertations Graduate Studies 5-1963 The Lower Devonian Water Canyon formation of Northeastern Utah Michael E. Taylor Follow this and additional works at: https://digitalcommons.usu.edu/etd Part of the Geology Commons Recommended Citation Taylor, Michael E., "The Lower Devonian Water Canyon formation of Northeastern Utah" (1963). All Graduate Theses and Dissertations. 6626. https://digitalcommons.usu.edu/etd/6626 This Thesis is brought to you for free and open access by the Graduate Studies at DigitalCommons@USU. It has been accepted for inclusion in All Graduate Theses and Dissertations by an authorized administrator of DigitalCommons@USU. For more information, please contact [email protected]. THE LOWER DEVONIAN WATER CANYON FORMATION OF NORTHEASTERN UTAH by Michael E. Taylor A thesis submitted in partial fulfillment of the requirements for the degree of MASTER OF SCIENCE in Geology UTAH STATE UNIVERSITY Logan, Utah 1963 ACKNOWLEDGMENTS The writer greatly appreciates the advice and criticism offered by Dean J. Stewart Williams, Dr. Clyde T . Hardy , and Dr. Donald R. Olsen, of the Department of Geology of Utah State University. He is also grateful to his parents for their encouragement during the course of this investigation and for capable assistance with the preparation of the manuscript. Michael E. Taylor TABLE OF CONTENTS Page INTRODUCTION o 1 General statement 1 Geolog ic setting o 3 External stratigraphic relations 4 WATER CANYON FORMATION 8 General statement 8 Card Member 9 Grassy Flat Member 13 Areal distribution 22 Paleonto logy 27 Age 33 Correlat i on 35 Environment of deposition 37 Source of detr itus 40 SUMMARY , 42 LITE RATURE CITED 43 APPENDIX 46 LIST OF FIGURES Figure Page 1. -

Chemical Element Concentrations of Cycad Leaves: Do We Know Enough?
horticulturae Review Chemical Element Concentrations of Cycad Leaves: Do We Know Enough? Benjamin E. Deloso 1 , Murukesan V. Krishnapillai 2 , Ulysses F. Ferreras 3, Anders J. Lindström 4, Michael Calonje 5 and Thomas E. Marler 6,* 1 College of Natural and Applied Sciences, University of Guam, Mangilao, GU 96923, USA; [email protected] 2 Cooperative Research and Extension, Yap Campus, College of Micronesia-FSM, Colonia, Yap 96943, Micronesia; [email protected] 3 Philippine Native Plants Conservation Society Inc., Ninoy Aquino Parks and Wildlife Center, Quezon City 1101, Philippines; [email protected] 4 Plant Collections Department, Nong Nooch Tropical Botanical Garden, 34/1 Sukhumvit Highway, Najomtien, Sattahip, Chonburi 20250, Thailand; [email protected] 5 Montgomery Botanical Center, 11901 Old Cutler Road, Coral Gables, FL 33156, USA; [email protected] 6 Western Pacific Tropical Research Center, University of Guam, Mangilao, GU 96923, USA * Correspondence: [email protected] Received: 13 October 2020; Accepted: 16 November 2020; Published: 19 November 2020 Abstract: The literature containing which chemical elements are found in cycad leaves was reviewed to determine the range in values of concentrations reported for essential and beneficial elements. We found 46 of the 358 described cycad species had at least one element reported to date. The only genus that was missing from the data was Microcycas. Many of the species reports contained concentrations of one to several macronutrients and no other elements. The cycad leaves contained greater nitrogen and phosphorus concentrations than the reported means for plants throughout the world. Magnesium was identified as the macronutrient that has been least studied. -

Heterospory: the Most Iterative Key Innovation in the Evolutionary History of the Plant Kingdom
Biol. Rej\ (1994). 69, l>p. 345-417 345 Printeii in GrenI Britain HETEROSPORY: THE MOST ITERATIVE KEY INNOVATION IN THE EVOLUTIONARY HISTORY OF THE PLANT KINGDOM BY RICHARD M. BATEMAN' AND WILLIAM A. DiMlCHELE' ' Departments of Earth and Plant Sciences, Oxford University, Parks Road, Oxford OXi 3P/?, U.K. {Present addresses: Royal Botanic Garden Edinburiih, Inverleith Rojv, Edinburgh, EIIT, SLR ; Department of Geology, Royal Museum of Scotland, Chambers Street, Edinburgh EHi ijfF) '" Department of Paleohiology, National Museum of Natural History, Smithsonian Institution, Washington, DC^zo^bo, U.S.A. CONTENTS I. Introduction: the nature of hf^terospon' ......... 345 U. Generalized life history of a homosporous polysporangiophyle: the basis for evolutionary excursions into hetcrospory ............ 348 III, Detection of hcterospory in fossils. .......... 352 (1) The need to extrapolate from sporophyte to gametophyte ..... 352 (2) Spatial criteria and the physiological control of heterospory ..... 351; IV. Iterative evolution of heterospory ........... ^dj V. Inter-cladc comparison of levels of heterospory 374 (1) Zosterophyllopsida 374 (2) Lycopsida 374 (3) Sphenopsida . 377 (4) PtiTopsida 378 (5) f^rogymnospermopsida ............ 380 (6) Gymnospermopsida (including Angiospermales) . 384 (7) Summary: patterns of character acquisition ....... 386 VI. Physiological control of hetcrosporic phenomena ........ 390 VII. How the sporophyte progressively gained control over the gametophyte: a 'just-so' story 391 (1) Introduction: evolutionary antagonism between sporophyte and gametophyte 391 (2) Homosporous systems ............ 394 (3) Heterosporous systems ............ 39(1 (4) Total sporophytic control: seed habit 401 VIII. Summary .... ... 404 IX. .•Acknowledgements 407 X. References 407 I. I.NIRODUCTION: THE NATURE OF HETEROSPORY 'Heterospory' sensu lato has long been one of the most popular re\ie\v topics in organismal botany. -
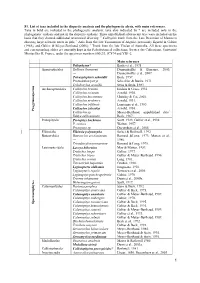
S1. List of Taxa Included in the Disparity Analysis and the Phylogenetic Alysis, with Main References
S1. List of taxa included in the disparity analysis and the phylogenetic alysis, with main references. Taxa in bold are included in the phylogenetic analysis; taxa also indicated by * are included only in the phylogenetic analysis and not in the disparity analysis. Three unpublished arborescent taxa were included on the basis that they showed additional anatomical diversity. 1 Callixylon trunk from the Late Devonian of Marrocco showing large sclerotic nests in pith; 2 Axis from the late Tournaisian of Algeria, previously figured in Galtier (1988), and Galtier & Meyer-Berthaud (2006); 3 Trunk from the late Viséan of Australia. All these specimens and corresponding slides are currently kept in the Paleobotanical collections, Service des Collections, Université Montpellier II, France, under the specimen numbers 600/2/3, JC874 and YB1-2. Main reference Psilophyton* Banks et al., 1975 Aneurophytales Rellimia thomsonii Dannenhoffer & Bonamo, 2003; --- Dannenhoffer et al., 2007. Tetraxylopteris schmidtii Beck, 1957. Proteokalon petryi Scheckler & Banks, 1971. Triloboxylon arnoldii Stein & Beck, 1983. s m Archaeopteridales Callixylon brownii Hoskin & Cross, 1951. r e Callixylon erianum Arnold, 1930. p s o Callixylon huronensis Chitaley & Cai, 2001. n Callixylon newberry Arnold, 1931. m y g Callixylon trifilievii Lemoigne et al., 1983. o r Callixylon zalesskyi Arnold, 1930. P Callixylon sp. Meyer-Berthaud, unpublished data1. Eddya sullivanensis Beck, 1967. Protopityales Protopitys buchiana Scott, 1923; Galtier et al., 1998. P. scotica Walton, 1957. Protopitys sp. Decombeix et al., 2005. Elkinsiales Elkinsia polymorpha Serbet & Rothwell, 1992. Buteoxylales Buteoxylon gordonianum Barnard &Long, 1973; Matten et al., --- 1980. Triradioxylon primaevum Barnard & Long, 1975. Lyginopteridales Laceya hibernica May & Matten, 1983. Tristichia longii Galtier, 1977. -

Structure, Development and Reproduction in Flowering Plants
Structure, Development and Reproduction in Flowering Plants Body Plan and Diversity in Form S.V.S Chauhan Professor Department of Botany B.R. Ambedkar University Khandari Campus Agra – 282002 [email protected] 1 Body Plan and Diversity in Form Every living organism has a fixed form and it is because of this reason that we are able to distinguish most of them just due to their external structure. Study of external morphology or external appearance of higher plants is necessary to describe the plants in an accurate fashion and to distinguish between almost similar looking plants. Therefore, the plants are identified by their morphological characters. Variation in plants is found not only in external forms but also in their anatomical characters which are represented by different types of tissue systems . Morphology along with anatomy constitute the base of studying pattern of life forms. Life Span of Plants On the basis of life span, plants are of three types: annuals, biennials and perennials. a) Annuals: These plants complete their life-cycle in a single growing season which varies from a few weeks to a few months. They pass the unfavourable period in the form of seeds. Examples are wheat, pea and sunflower, etc. b) Biennials: These plants complete their life-cycle in two growing seasons. In the first season; they grow only vegetatively and store food generally in the roots. In the second season, these plants grow at the expense of the stored food and form the flowering shoot bearing flowers, fruits and seeds. Then the plants die. radish, turnip, cabbage, etc. -

The Big Bloom—How Flowering Plants Changed the World
The Big Bloom—How Flowering Plants Changed the World Written by Michael Klesius Republished from the pages of National Geographic magazine -- July 2002 In the summer of 1973 sunflowers appeared in my father's vegetable garden. They seemed to sprout overnight in a few rows he had lent that year to new neighbors from California. Only six years old at the time, I was at first put off by these garish plants. Such strange and vibrant flowers seemed out of place among the respectable beans, peppers, spinach, and other vegetables we had always grown. Gradually, however, the brilliance of the sunflowers won me over. Their fiery halos relieved the green monotone that by late summer ruled the garden. I marveled at birds that clung upside down to the shaggy, gold disks, wings fluttering, looting the seeds. Sunflowers defined flowers for me that summer and changed my view of the world. Flowers have a way of doing that. They began changing the way the world looked almost as soon as they appeared on Earth about 130 million years ago, during the Cretaceous period. That's relatively recent in geologic time: If all Earth's history were compressed into an hour, flowering plants would exist for only the last 90 seconds. But once they took firm root about 100 million years ago, they swiftly diversified in an explosion of varieties that established most of the flowering plant families of the modern world. Today flowering plant species outnumber by twenty to one those of ferns and cone-bearing trees, or conifers, which had thrived for 200 million years before the first bloom appeared. -

Earliest Record of Megaphylls and Leafy Structures, and Their Initial Diversification
Review Geology August 2013 Vol.58 No.23: 27842793 doi: 10.1007/s11434-013-5799-x Earliest record of megaphylls and leafy structures, and their initial diversification HAO ShouGang* & XUE JinZhuang Key Laboratory of Orogenic Belts and Crustal Evolution, School of Earth and Space Sciences, Peking University, Beijing 100871, China Received January 14, 2013; accepted February 26, 2013; published online April 10, 2013 Evolutionary changes in the structure of leaves have had far-reaching effects on the anatomy and physiology of vascular plants, resulting in morphological diversity and species expansion. People have long been interested in the question of the nature of the morphology of early leaves and how they were attained. At least five lineages of euphyllophytes can be recognized among the Early Devonian fossil plants (Pragian age, ca. 410 Ma ago) of South China. Their different leaf precursors or “branch-leaf com- plexes” are believed to foreshadow true megaphylls with different venation patterns and configurations, indicating that multiple origins of megaphylls had occurred by the Early Devonian, much earlier than has previously been recognized. In addition to megaphylls in euphyllophytes, the laminate leaf-like appendages (sporophylls or bracts) occurred independently in several dis- tantly related Early Devonian plant lineages, probably as a response to ecological factors such as high atmospheric CO2 concen- trations. This is a typical example of convergent evolution in early plants. Early Devonian, euphyllophyte, megaphyll, leaf-like appendage, branch-leaf complex Citation: Hao S G, Xue J Z. Earliest record of megaphylls and leafy structures, and their initial diversification. Chin Sci Bull, 2013, 58: 27842793, doi: 10.1007/s11434- 013-5799-x The origin and evolution of leaves in vascular plants was phology and evolutionary diversification of early leaves of one of the most important evolutionary events affecting the basal euphyllophytes remain enigmatic. -

Ordovician Land Plants and Fungi from Douglas Dam, Tennessee
PROOF The Palaeobotanist 68(2019): 1–33 The Palaeobotanist 68(2019): xxx–xxx 0031–0174/2019 0031–0174/2019 Ordovician land plants and fungi from Douglas Dam, Tennessee GREGORY J. RETALLACK Department of Earth Sciences, University of Oregon, Eugene, OR 97403, USA. *Email: gregr@uoregon. edu (Received 09 September, 2019; revised version accepted 15 December, 2019) ABSTRACT The Palaeobotanist 68(1–2): Retallack GJ 2019. Ordovician land plants and fungi from Douglas Dam, Tennessee. The Palaeobotanist 68(1–2): xxx–xxx. 1–33. Ordovician land plants have long been suspected from indirect evidence of fossil spores, plant fragments, carbon isotopic studies, and paleosols, but now can be visualized from plant compressions in a Middle Ordovician (Darriwilian or 460 Ma) sinkhole at Douglas Dam, Tennessee, U. S. A. Five bryophyte clades and two fungal clades are represented: hornwort (Casterlorum crispum, new form genus and species), liverwort (Cestites mirabilis Caster & Brooks), balloonwort (Janegraya sibylla, new form genus and species), peat moss (Dollyphyton boucotii, new form genus and species), harsh moss (Edwardsiphyton ovatum, new form genus and species), endomycorrhiza (Palaeoglomus strotheri, new species) and lichen (Prototaxites honeggeri, new species). The Douglas Dam Lagerstätte is a benchmark assemblage of early plants and fungi on land. Ordovician plant diversity now supports the idea that life on land had increased terrestrial weathering to induce the Great Ordovician Biodiversification Event in the sea and latest Ordovician (Hirnantian) -

Cycas Revoluta1
Fact Sheet FPS-162 October, 1999 Cycas revoluta1 Edward F. Gilman2 Introduction The palm-like King Sago has a short, dark brown, unbranching trunk topped with graceful, arching, dark green, feathery leaves, two to three feet long (Fig. 1). King Sago is not a palm tree. It will eventually reach to about eight feet tall, but grows about one or two inches per year. The individual leaflets each end in a sharp tip. Although very slow-growing, King Sago is much prized for its tropical effect and easy care and makes an excellent specimen or container plant. It is easy to grow as a house plant. General Information Scientific name: Cycas revoluta Pronunciation: SYE-kus rev-voe-LOO-tuh Common name(s): King Sago Family: Cycadaceae Plant type: shrub USDA hardiness zones: 8B through 11 (Fig. 2) Planting month for zone 8: year round Planting month for zone 9: year round Figure 1. King Sago. Planting month for zone 10 and 11: year round Origin: not native to North America Uses: container or above-ground planter; border; mass Description planting; accent; suitable for growing indoors Height: 3 to 10 feet Availablity: somewhat available, may have to go out of the Spread: 4 to 8 feet region to find the plant Plant habit: palm Plant density: moderate Growth rate: slow Texture: fine 1.This document is Fact Sheet FPS-162, one of a series of the Environmental Horticulture Department, Florida Cooperative Extension Service, Institute of Food and Agricultural Sciences, University of Florida. Publication date: October 1999. Please visit the EDIS web site at http://edis.ifas.ufl.edu. -

Comparative Biology of Cycad Pollen, Seed and Tissue - a Plant Conservation Perspective
Bot. Rev. (2018) 84:295–314 https://doi.org/10.1007/s12229-018-9203-z Comparative Biology of Cycad Pollen, Seed and Tissue - A Plant Conservation Perspective J. Nadarajan1,2 & E. E. Benson 3 & P. Xaba 4 & K. Harding3 & A. Lindstrom5 & J. Donaldson4 & C. E. Seal1 & D. Kamoga6 & E. M. G. Agoo7 & N. Li 8 & E. King9 & H. W. Pritchard1,10 1 Royal Botanic Gardens, Kew, Wakehurst Place, Ardingly, West Sussex RH17 6TN, UK; e-mail: [email protected] 2 The New Zealand Institute for Plant & Food Research Ltd, Private Bag 11600, Palmerston North 4442, New Zealand; e-mail [email protected] 3 Damar Research Scientists, Damar, Cuparmuir, Fife KY15 5RJ, UK; e-mail: [email protected]; [email protected] 4 South African National Biodiversity Institute, Kirstenbosch National Botanical Garden, Cape Town, Republic of South Africa; e-mail: [email protected]; [email protected] 5 Nong Nooch Tropical Botanical Garden, Chonburi 20250, Thailand; e-mail: [email protected] 6 Joint Ethnobotanical Research Advocacy, P.O.Box 27901, Kampala, Uganda; e-mail: [email protected] 7 De La Salle University, Manila, Philippines; e-mail: [email protected] 8 Fairy Lake Botanic Garden, Shenzhen, Guangdong, People’s Republic of China; e-mail: [email protected] 9 UNEP-World Conservation Monitoring Centre, Cambridge, UK; e-mail: [email protected] 10 Author for Correspondence; e-mail: [email protected] Published online: 5 July 2018 # The Author(s) 2018 Abstract Cycads are the most endangered of plant groups based on IUCN Red List assessments; all are in Appendix I or II of CITES, about 40% are within biodiversity ‘hotspots,’ and the call for action to improve their protection is long- standing. -

Proliferated Megasporangiate Strobili of Zamia Furfuracea (Zamiaceae, Cycadales) and Its Possible Evolutionary Implications for the Origin of Cycad-Megasporophylls
Palaeodiversity 6: 135–147; Stuttgart, 30 December 2013. 135 Proliferated megasporangiate strobili of Zamia furfuracea (Zamiaceae, Cycadales) and its possible evolutionary implications for the origin of cycad-megasporophylls VEIT MARTIN DÖRKEN & BRIGITTE ROZYNEK Abstract At a 30-years-old individual of Zamia furfuracea (Zamiaceae, Cycadales) cultivated in the Botanic Garden Bo- chum (Germany), several proliferated megasporangiate strobili were found. The morphology of normal and prolif- erated strobili was compared. Within the proliferated strobili the sequence of megasporophylls, cataphylls, tropho- phyll-like leaves, followed again by a flush of cataphylls, was similar to those developed at the stems of extant Cycas species. However, all proliferated megasporangiate strobili were sterile. Within the proliferated strobili the pinnate trophophyll-like leaves that were replacing the terminal megasporophylls can be regarded as an atavism possibly reflecting the primitive character of megasporophylls in cycads. Thus, the results of the morphological examinations and also the comparison with fossil taxa may deliver new data supporting the idea that pinnate cycad-megasporo- phylls are a plesiomorphic feature within cycads. Keywords: Zamia, Cycadales, strobilus, megasporophyll, proliferation. 1. Introduction often wedge-shaped with a hexagonal outer face. In some species they have one or two distal spine-like appendages. Due to the morphology of megasporangiate strobili, me- Each megasporophyll bears only two ovules, which are gasporophylls and the attachment of ovules, the systemat- developed deeply within the strobilus. The micropyles are ics among extant cycads is still debated. Some authors sug- pointing towards the axis of the strobilus. gest a concept composing of three families: Cycadaceae, In contrast with the Zamiaceae, among the Cycadaceae Stangeriaceae, and Zamiaceae (e.g.