Signal Peptide Peptidase‐Like 2C Impairs Vesicular Transport And
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

A Novel 65-Bp Indel in the GOLGB1 Gene Is Associated with Chicken Growth and Carcass Traits
animals Article A Novel 65-bp Indel in the GOLGB1 Gene Is Associated with Chicken Growth and Carcass Traits Rong Fu 1,2, Tuanhui Ren 1,2, Wangyu Li 1,2, Jiaying Liang 1,2, Guodong Mo 1,2, Wen Luo 1,2, Danlin He 1,2, Shaodong Liang 1,2 and Xiquan Zhang 1,2,* 1 Department of Animal Genetics, Breeding and Reproduction, College of Animal Science, South China Agricultural University, Guangzhou 510642, China; [email protected] (R.F.); [email protected] (T.R.); [email protected] (W.L.); [email protected] (J.L.); [email protected] (G.M.); [email protected] (W.L.); [email protected] (D.H.); [email protected] (S.L.) 2 Guangdong Provincial Key Lab of Agro-Animal Genomics and Molecular Breeding, and Key Laboratory of Chicken Genetics, Breeding and Reproduction, Ministry of Agriculture, Guangzhou 510642, China * Correspondence: [email protected] Received: 20 February 2020; Accepted: 4 March 2020; Published: 12 March 2020 Simple Summary: Many Chinese-local chickens show slow-growing and low-producing performance, which is not conductive to the development of the poultry industry. The identification of thousands of indels in the last twenty years has helped us to make progress in animal genetics and breeding. Golgin subfamily B member 1 (GOLGB1) is located on chromosome 1 in chickens. Previous study showed that a large number of QTLs on the chicken chromosome 1 were related to the important economic traits. However, the biological function of GOLGB1 gene in chickens is still unclear. In this study, we detected a novel 65-bp indel in the fifth intron of the chicken GOLGB1 gene. -
![Viewed Previously [4]](https://docslib.b-cdn.net/cover/6213/viewed-previously-4-126213.webp)
Viewed Previously [4]
Barlow et al. BMC Biology (2018) 16:27 https://doi.org/10.1186/s12915-018-0492-9 RESEARCHARTICLE Open Access A sophisticated, differentiated Golgi in the ancestor of eukaryotes Lael D. Barlow1, Eva Nývltová2,3, Maria Aguilar1, Jan Tachezy2 and Joel B. Dacks1,4* Abstract Background: The Golgi apparatus is a central meeting point for the endocytic and exocytic systems in eukaryotic cells, and the organelle’s dysfunction results in human disease. Its characteristic morphology of multiple differentiated compartments organized into stacked flattened cisternae is one of the most recognizable features of modern eukaryotic cells, and yet how this is maintained is not well understood. The Golgi is also an ancient aspect of eukaryotes, but the extent and nature of its complexity in the ancestor of eukaryotes is unclear. Various proteins have roles in organizing the Golgi, chief among them being the golgins. Results: We address Golgi evolution by analyzing genome sequences from organisms which have lost stacked cisternae as a feature of their Golgi and those that have not. Using genomics and immunomicroscopy, we first identify Golgi in the anaerobic amoeba Mastigamoeba balamuthi. We then searched 87 genomes spanning eukaryotic diversity for presence of the most prominent proteins implicated in Golgi structure, focusing on golgins. We show some candidates as animal specific and others as ancestral to eukaryotes. Conclusions: None of the proteins examined show a phyletic distribution that correlates with the morphology of stacked cisternae, suggesting the possibility of stacking as an emergent property. Strikingly, however, the combination of golgins conserved among diverse eukaryotes allows for the most detailed reconstruction of the organelle to date, showing a sophisticated Golgi with differentiated compartments and trafficking pathways in the common eukaryotic ancestor. -

Knock-Out of ACBD3 Leads to Dispersed Golgi Structure, but Unaffected Mitochondrial Functions in HEK293 and Hela Cells
International Journal of Molecular Sciences Article Knock-Out of ACBD3 Leads to Dispersed Golgi Structure, but Unaffected Mitochondrial Functions in HEK293 and HeLa Cells Tereza Da ˇnhelovská 1 , Lucie Zdražilová 1 , Hana Štufková 1, Marie Vanišová 1, Nikol Volfová 1, Jana Kˇrížová 1 , OndˇrejKuda 2 , Jana Sládková 1 and Markéta Tesaˇrová 1,* 1 Department of Paediatrics and Inherited Metabolic Disorders, Charles University, First Faculty of Medicine and General University Hospital in Prague, 128 01 Prague, Czech Republic; [email protected] (T.D.); [email protected] (L.Z.); [email protected] (H.Š.); [email protected] (M.V.); [email protected] (N.V.); [email protected] (J.K.); [email protected] (J.S.) 2 Institute of Physiology, Academy of Sciences of the Czech Republic, 142 00 Prague, Czech Republic; [email protected] * Correspondence: [email protected] Abstract: The Acyl-CoA-binding domain-containing protein (ACBD3) plays multiple roles across the cell. Although generally associated with the Golgi apparatus, it operates also in mitochondria. In steroidogenic cells, ACBD3 is an important part of a multiprotein complex transporting cholesterol into mitochondria. Balance in mitochondrial cholesterol is essential for proper mitochondrial protein biosynthesis, among others. We generated ACBD3 knock-out (ACBD3-KO) HEK293 and HeLa cells and characterized the impact of protein absence on mitochondria, Golgi, and lipid profile. In ACBD3- Citation: Daˇnhelovská,T.; KO cells, cholesterol level and mitochondrial structure and functions are not altered, demonstrating Zdražilová, L.; Štufková, H.; that an alternative pathway of cholesterol transport into mitochondria exists. However, ACBD3- Vanišová, M.; Volfová, N.; Kˇrížová,J.; Kuda, O.; Sládková, J.; Tesaˇrová,M. -

Defining the Kv2.1–Syntaxin Molecular Interaction Identifies a First-In-Class Small Molecule Neuroprotectant
Defining the Kv2.1–syntaxin molecular interaction identifies a first-in-class small molecule neuroprotectant Chung-Yang Yeha,b,1, Zhaofeng Yec,d,1, Aubin Moutale, Shivani Gaura,b, Amanda M. Hentonf,g, Stylianos Kouvarosf,g, Jami L. Salomana, Karen A. Hartnett-Scotta,b, Thanos Tzounopoulosa,f,g, Rajesh Khannae, Elias Aizenmana,b,g,2, and Carlos J. Camachoc,2 aDepartment of Neurobiology, University of Pittsburgh School of Medicine, Pittsburgh, PA 15261; bPittsburgh Institute for Neurodegenerative Diseases, University of Pittsburgh School of Medicine, Pittsburgh, PA 15261; cDepartment of Computational and Systems Biology, University of Pittsburgh School of Medicine, Pittsburgh, PA 15261; dSchool of Medicine, Tsinghua University, Beijing 100871, China; eDepartment of Pharmacology, College of Medicine, University of Arizona, Tucson, AZ 85724; fDepartment of Otolaryngology, University of Pittsburgh School of Medicine, Pittsburgh, PA 15261; and gPittsburgh Hearing Research Center, University of Pittsburgh School of Medicine, Pittsburgh, PA 15261 Edited by Lily Yeh Jan, University of California, San Francisco, CA, and approved June 19, 2019 (received for review February 27, 2019) + The neuronal cell death-promoting loss of cytoplasmic K follow- (13). The Kv2.1-dependent cell death pathway is normally initiated ing injury is mediated by an increase in Kv2.1 potassium channels in by the oxidative liberation of zinc from intracellular metal-binding the plasma membrane. This phenomenon relies on Kv2.1 binding to proteins (14), leading to the sequential phosphorylation of syntaxin 1A via 9 amino acids within the channel intrinsically disor- Kv2.1 residues Y124 and S800 by Src and p38 kinases, respectively dered C terminus. Preventing this interaction with a cell and blood- (15–17). -

The Murine Orthologue of the Golgi-Localized TPTE Protein Provides Clues to the Evolutionary History of the Human TPTE Gene Family
View metadata, citation and similar papers at core.ac.uk brought to you by CORE provided by RERO DOC Digital Library Hum Genet (2001) 109:569–575 DOI 10.1007/s004390100607 ORIGINAL INVESTIGATION Michel Guipponi · Caroline Tapparel · Olivier Jousson · Nathalie Scamuffa · Christophe Mas · Colette Rossier · Pierre Hutter · Paolo Meda · Robert Lyle · Alexandre Reymond · Stylianos E. Antonarakis The murine orthologue of the Golgi-localized TPTE protein provides clues to the evolutionary history of the human TPTE gene family Received: 5 June 2001 / Accepted: 17 August 2001 / Published online: 27 October 2001 © Springer-Verlag 2001 Abstract The human TPTE gene encodes a testis-spe- events. The Y chromosome copy of TPTE is a pseudo- cific protein that contains four potential transmembrane gene and is not therefore involved in the testis expression domains and a protein tyrosine phosphatase motif, and of this gene family. shows homology to the tumor suppressor PTEN/MMAC1. Chromosomal mapping revealed multiple copies of the TPTE gene present on the acrocentric chromosomes 13, Introduction 15, 21 and 22, and the Y chromosome. Zooblot analysis suggests that mice may possess only one copy of TPTE. We have recently identified a testis-specific cDNA, TPTE In the present study, we report the isolation and initial (Transmembrane Phosphatase with TEnsin homology), characterization of the full-length cDNA of the mouse ho- that encodes a predicted protein of 551 amino acids con- mologue Tpte. At least three different mRNA transcripts taining four potential transmembrane domains and a tyro- (Tpte.a, b, c) are produced via alternative splicing, encod- sine phosphatase motif (Chen H et al. -

Incredibly Close—A Newly Identified Peroxisome–ER Contact Site in Humans
JCB: Spotlight Incredibly close—A newly identified peroxisome–ER contact site in humans Maya Schuldiner and Einat Zalckvar Department of Molecular Genetics, Weizmann Institute of Science, Rehovot 7610001, Israel Peroxisomes are tiny organelles that control important residents of contact sites and to mediate membrane association and diverse metabolic processes via their interplay with through their major sperm protein (MSP) domain (Wyles and Ridgway, 2004), they were exciting candidates for tethers. The other organelles, including the endoplasmic reticulum MSP domain of VAPs interacts with proteins that contain two (ER). In this issue, Costello et al. (2017. J. Cell Biol. phenylalanines (FF) in an acidic tract (FFAT) motif. When the https://doi.org/10.1083/jcb.201607055) and Hua et MSP and FFAT motifs are located in proteins that are anchored al. (2017. J. Cell Biol. https://doi.org/10.1083/jcb to opposing organelle membranes, the MSP–FFAT interac- tion zippers up the contact. Hua et al. (2017) looked among .201608128) identify a peroxisome–ER contact site in their PEX16-binding candidates for proteins with a FFAT do- human cells held together by a tethering complex of main and identified ACBD5. The observation that peroxisomal VAPA/B (vesicle-associated membrane protein–associated ACBD5 contained a FFAT domain and could be found in the same protein complex as the ER tethering proteins VAPA and proteins A/B) and ACBD5 (acyl Co-A binding protein 5). VAPB led both groups to investigate whether or not these pro- teins play a role in peroxisome–ER tethering. Peroxisomes are organelles enclosed by a single membrane We have recently put in place a suggestion to term a pro- that exist in most eukaryotic organisms and cells and are in- tein a tether only if it abides by three criteria (Eisenberg-Bord volved in various metabolic, as well as nonmetabolic, cellular et al., 2016). -

Behavioral and Other Phenotypes in a Cytoplasmic Dynein Light Intermediate Chain 1 Mutant Mouse
The Journal of Neuroscience, April 6, 2011 • 31(14):5483–5494 • 5483 Cellular/Molecular Behavioral and Other Phenotypes in a Cytoplasmic Dynein Light Intermediate Chain 1 Mutant Mouse Gareth T. Banks,1* Matilda A. Haas,5* Samantha Line,6 Hazel L. Shepherd,6 Mona AlQatari,7 Sammy Stewart,7 Ida Rishal,8 Amelia Philpott,9 Bernadett Kalmar,2 Anna Kuta,1 Michael Groves,3 Nicholas Parkinson,1 Abraham Acevedo-Arozena,10 Sebastian Brandner,3,4 David Bannerman,6 Linda Greensmith,2,4 Majid Hafezparast,9 Martin Koltzenburg,2,4,7 Robert Deacon,6 Mike Fainzilber,8 and Elizabeth M. C. Fisher1,4 1Department of Neurodegenerative Disease, 2Sobell Department of Motor Science and Movement Disorders, 3Division of Neuropathology, and 4Medical Research Council (MRC) Centre for Neuromuscular Diseases, University College London (UCL) Institute of Neurology, London WC1N 3BG, United Kingdom, 5MRC National Institute for Medical Research, London NW7 1AA, United Kingdom, 6Department of Experimental Psychology, University of Oxford, Oxford OX1 3UD, United Kingdom, 7UCL Institute of Child Health, London WC1N 1EH, United Kingdom, 8Department of Biological Chemistry, Weizmann Institute of Science, 76100 Rehovot, Israel, 9School of Life Sciences, University of Sussex, Brighton BN1 9QG, United Kingdom, and 10MRC Mammalian Genetics Unit, Harwell OX11 ORD, United Kingdom The cytoplasmic dynein complex is fundamentally important to all eukaryotic cells for transporting a variety of essential cargoes along microtubules within the cell. This complex also plays more specialized roles in neurons. The complex consists of 11 types of protein that interact with each other and with external adaptors, regulators and cargoes. Despite the importance of the cytoplasmic dynein complex, weknowcomparativelylittleoftherolesofeachcomponentprotein,andinmammalsfewmutantsexistthatallowustoexploretheeffects of defects in dynein-controlled processes in the context of the whole organism. -

Multifaceted Polo-Like Kinases: Drug Targets and Antitargets for Cancer Therapy
REVIEWS Multifaceted polo-like kinases: drug targets and antitargets for cancer therapy Klaus Strebhardt Abstract | The polo-like kinase 1 (PLK1) acts in concert with cyclin-dependent kinase 1–cyclin B1 and Aurora kinases to orchestrate a wide range of critical cell cycle events. Because PLK1 has been preclinically validated as a cancer target, small-molecule inhibitors of PLK1 have become attractive candidates for anticancer drug development. Although the roles of the closely related PLK2, PLK3 and PLK4 in cancer are less well understood, there is evidence showing that PLK2 and PLK3 act as tumour suppressors through their functions in the p53 signalling network, which guards the cell against various stress signals. In this article, recent insights into the biology of PLKs will be reviewed, with an emphasis on their role in malignant transformation, and progress in the development of small-molecule PLK1 inhibitors will be examined. More than two decades ago polo, the founding member Early observations on the overexpression of PLK1 of the family of polo-like kinases (PLKs), was identified in human tumours12 and on the inhibition of cellu- as having an essential role in the ordered execution of lar proliferation by microinjecting antibodies against mitotic events in Drosophila melanogaster 1,2. Since the PLK1 into HeLa cells13 initiated a series of follow-on discovery of polo, a wealth of functional information on studies using a broad spectrum of inhibitors (for exam- PLKs has been collected in a wide phylogenetic space. ple, dominant-negative forms of PLK1, antisense oli- Five mammalian PLK family members have been iden- gonucleotides and small interfering RNAs) aiming to tified so far, PLK1 (also known as STPK13), PLK2 (also evaluate PLK1 as a potential target for the treatment known as SNK), PLK3 (also known as CNK, FNK and of cancer14–17. -

Datasheet: MCA3923Z Product Details
Datasheet: MCA3923Z Description: MOUSE ANTI HUMAN ACBD3:Preservative Free Specificity: ACBD3 Format: Preservative Free Product Type: Monoclonal Antibody Clone: 2G2 Isotype: IgG1 Quantity: 0.1 mg Product Details Applications This product has been reported to work in the following applications. This information is derived from testing within our laboratories, peer-reviewed publications or personal communications from the originators. Please refer to references indicated for further information. For general protocol recommendations, please visit www.bio-rad-antibodies.com/protocols. Yes No Not Determined Suggested Dilution Immunohistology - Paraffin (1) 0.1 - 10 ug/ml Western Blotting Immunofluorescence 0.1 - 10 ug/ml Where this product has not been tested for use in a particular technique this does not necessarily exclude its use in such procedures. Suggested working dilutions are given as a guide only. It is recommended that the user titrates the product for use in their own system using appropriate negative/positive controls. (1)This product requires antigen retrieval using heat treatment prior to staining of paraffin sections.Sodium citrate buffer pH 6.0 is recommended for this purpose. Target Species Human Species Cross Reacts with: Rat, Mouse Reactivity N.B. Antibody reactivity and working conditions may vary between species. Product Form Purified IgG - liquid Preparation Purified IgG prepared by affinity chromatography on Protein A Buffer Solution Phosphate buffered saline Preservative None present Stabilisers Approx. Protein Ig concentration 0.5 mg/ml Concentrations Immunogen Recombinant protein corresponding to aa 73-172 of human ACBD3 Page 1 of 3 External Database Links UniProt: Q9H3P7 Related reagents Entrez Gene: 64746 ACBD3 Related reagents Synonyms GCP60, GOCAP1, GOLPH1 Fusion Partners Spleen cells from immunised Balb/c mice were fused with cells from the Sp2/0 myeloma cell line. -
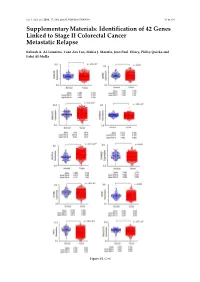
Identification of 42 Genes Linked to Stage II Colorectal Cancer Metastatic Relapse
Int. J. Mol. Sci. 2016, 17, 598; doi:10.3390/ijms17040598 S1 of S16 Supplementary Materials: Identification of 42 Genes Linked to Stage II Colorectal Cancer Metastatic Relapse Rabeah A. Al-Temaimi, Tuan Zea Tan, Makia J. Marafie, Jean Paul Thiery, Philip Quirke and Fahd Al-Mulla Figure S1. Cont. Int. J. Mol. Sci. 2016, 17, 598; doi:10.3390/ijms17040598 S2 of S16 Figure S1. Mean expression levels of fourteen genes of significant association with CRC DFS and OS that are differentially expressed in normal colon compared to CRC tissues. Each dot represents a sample. Table S1. Copy number aberrations associated with poor disease-free survival and metastasis in early stage II CRC as predicted by STAC and SPPS combined methodologies with resident gene symbols. CN stands for copy number, whereas CNV is copy number variation. Region Cytoband % of CNV Count of Region Event Gene Symbols Length Location Overlap Genes chr1:113,025,076–113,199,133 174,057 p13.2 CN Loss 0.0 2 AKR7A2P1, SLC16A1 chr1:141,465,960–141,822,265 356,305 q12–q21.1 CN Gain 95.9 1 SRGAP2B MIR5087, LOC10013000 0, FLJ39739, LOC10028679 3, PPIAL4G, PPIAL4A, NBPF14, chr1:144,911,564–146,242,907 1,331,343 q21.1 CN Gain 99.6 16 NBPF15, NBPF16, PPIAL4E, NBPF16, PPIAL4D, PPIAL4F, LOC645166, LOC388692, FCGR1C chr1:177,209,428–177,226,812 17,384 q25.3 CN Gain 0.0 0 chr1:197,652,888–197,676,831 23,943 q32.1 CN Gain 0.0 1 KIF21B chr1:201,015,278–201,033,308 18,030 q32.1 CN Gain 0.0 1 PLEKHA6 chr1:201,289,154–201,298,247 9093 q32.1 CN Gain 0.0 0 chr1:216,820,186–217,043,421 223,235 q41 CN -

Human Induced Pluripotent Stem Cell–Derived Podocytes Mature Into Vascularized Glomeruli Upon Experimental Transplantation
BASIC RESEARCH www.jasn.org Human Induced Pluripotent Stem Cell–Derived Podocytes Mature into Vascularized Glomeruli upon Experimental Transplantation † Sazia Sharmin,* Atsuhiro Taguchi,* Yusuke Kaku,* Yasuhiro Yoshimura,* Tomoko Ohmori,* ‡ † ‡ Tetsushi Sakuma, Masashi Mukoyama, Takashi Yamamoto, Hidetake Kurihara,§ and | Ryuichi Nishinakamura* *Department of Kidney Development, Institute of Molecular Embryology and Genetics, and †Department of Nephrology, Faculty of Life Sciences, Kumamoto University, Kumamoto, Japan; ‡Department of Mathematical and Life Sciences, Graduate School of Science, Hiroshima University, Hiroshima, Japan; §Division of Anatomy, Juntendo University School of Medicine, Tokyo, Japan; and |Japan Science and Technology Agency, CREST, Kumamoto, Japan ABSTRACT Glomerular podocytes express proteins, such as nephrin, that constitute the slit diaphragm, thereby contributing to the filtration process in the kidney. Glomerular development has been analyzed mainly in mice, whereas analysis of human kidney development has been minimal because of limited access to embryonic kidneys. We previously reported the induction of three-dimensional primordial glomeruli from human induced pluripotent stem (iPS) cells. Here, using transcription activator–like effector nuclease-mediated homologous recombination, we generated human iPS cell lines that express green fluorescent protein (GFP) in the NPHS1 locus, which encodes nephrin, and we show that GFP expression facilitated accurate visualization of nephrin-positive podocyte formation in -

The Rhodococcus Equi Virulence Protein Vapa Disrupts Endolysosome Function and Stimulates Lysosome Biogenesis
This is a repository copy of The Rhodococcus equi virulence protein VapA disrupts endolysosome function and stimulates lysosome biogenesis. White Rose Research Online URL for this paper: https://eprints.whiterose.ac.uk/104956/ Version: Accepted Version Article: Rofe, Adam P, Davis, Luther J, Whittingham, Jean L et al. (3 more authors) (2016) The Rhodococcus equi virulence protein VapA disrupts endolysosome function and stimulates lysosome biogenesis. Microbiology Open. e00416. ISSN 2045-8827 https://doi.org/10.1002/mbo3.416 Reuse This article is distributed under the terms of the Creative Commons Attribution (CC BY) licence. This licence allows you to distribute, remix, tweak, and build upon the work, even commercially, as long as you credit the authors for the original work. More information and the full terms of the licence here: https://creativecommons.org/licenses/ Takedown If you consider content in White Rose Research Online to be in breach of UK law, please notify us by emailing [email protected] including the URL of the record and the reason for the withdrawal request. [email protected] https://eprints.whiterose.ac.uk/ The Rhodococcus equi virulence protein VapA disrupts endolysosome function and stimulates lysosome biogenesis. Adam P. Rofe1, Luther J. Davis2, Jean L. Whittingham3, Elizabeth C. Latimer- Bowman2, Anthony J. Wilkinson3 and Paul R. Pryor1,4* 1 Department of Biology, Wentworth Way, University of York, York. YO10 5DD. United Kingdom; 2 Cambridge Institute for Medical Research and Department of Clinical Biochemistry, University of Cambridge, Addenbrooke’s Hospital, Hills Rd, Cambridge, CB2 0XY; 3 Structural Biology Laboratory, Department of Chemistry, University of York York.