VIII. Peptide Hormones and Behavior: Cholecystokinin and Prolactin
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Therapeutic Approaches to Preserve Islet Mass in Type 2 Diabetes
24 Dec 2005 16:24 AR ANRV262-ME57-17.tex XMLPublishSM(2004/02/24) P1: OJO 10.1146/annurev.med.57.110104.115624 Annu. Rev. Med. 2006. 57:265–81 doi: 10.1146/annurev.med.57.110104.115624 Copyright c 2006 by Annual Reviews. All rights reserved THERAPEUTIC APPROACHES TO PRESERVE ISLET MASS IN TYPE 2DIABETES Laurie L. Baggio and Daniel J. Drucker Department of Medicine, Toronto General Hospital, Banting and Best Diabetes Center, University of Toronto, Toronto, Ontario, Canada M5S 2S2; email: [email protected] KeyWords β-cell mass, apoptosis, neogenesis, proliferation ■ Abstract Type 2 diabetes is characterized by hyperglycemia resulting from in- sulin resistance in the setting of inadequate β-cell compensation. Currently available therapeutic agents lower blood glucose through multiple mechanisms but do not directly reverse the decline in β-cell mass. Glucagon-like peptide-1 (GLP-1) receptor agonists, exemplified by Exenatide (exendin-4), not only acutely lower blood glucose but also engage signaling pathways in the islet β-cell that lead to stimulation of β-cell repli- cation and inhibition of β-cell apoptosis. Similarly, glucose-dependent insulinotropic polypeptide (GIP) receptor activation stimulates insulin secretion, enhances β-cell proliferation, and reduces apoptosis. Moreover, potentiation of the endogenous post- prandial levels of GLP-1 and GIP via inhibition of dipeptidyl peptidase-IV (DPP-IV) also expands β-cell mass via related mechanisms. The thiazolidinediones (TZDs) en- hance insulin sensitivity, reduce blood glucose levels, and also preserve β-cell mass, although it remains unclear whether TZDs affect β-cell mass via direct mechanisms. Complementary approaches to regeneration of β-cell mass involve combinations of factors, exemplified by epidermal growth factor and gastrin, which promote islet neo- genesis and ameliorate diabetes in rodent studies. -

Corticotropin-Releasing Hormone Physiology
European Journal of Endocrinology (2006) 155 S71–S76 ISSN 0804-4643 Corticotropin-releasing hormone physiology Joseph A Majzoub Division of Endocrinology, Children’s Hospital Boston, Thomas Morgan Rotch Professor of Pediatrics, Harvard Medical School, 300 Longwood Avenue, Boston, Massachusetts 02115, USA (Correspondence should be addressed to J A Majzoub; Email: [email protected]) Abstract Corticotropin-releasing hormone (CRH), also known as corticotropin-releasing factor, is a highly conserved peptide hormone comprising 41 amino acid residues. Its name derives from its role in the anterior pituitary, where it mediates the release of corticotropin (ACTH) leading to the release of adrenocortical steroids. CRH is the major hypothalamic activator of the hypothalamic–pituitary– adrenal (HPA)axis. Major functions of the HPAinclude: (i) influencing fetal development of major organ systems including lung, liver, and gut, (ii) metabolic functions, including the maintenance of normal blood glucose levels during the fasting state via glycogenolysis and gluconeogenesis, (iii) modulation of immune function, and (iv) maintenance of cardiovascular tone. In addition, CRH, acting both directly and via the HPA, has a role in regulating several neuroendocrine functions including behavior, food intake, reproduction, growth, immune function, and autonomic function. CRH has been localized to the paraventricular nucleus (PVN) of the hypothalamus, which projects to the median eminence and other hypothalamic and midbrain targets. The CRH gene is composed of two exons. The CRH promoter contains a cAMP-response element, and the intron contains a restrictive element-1/neuron restrictive silencing element (RE-1/NRSE) sequence. Recently, a family of CRH-related peptides, termed the urocortins, has been identified. -

Leptin Stimulates Pituitary Prolactin Release Through an Extracellular Signal-Regulated Kinase-Dependent Pathway
275 Leptin stimulates pituitary prolactin release through an extracellular signal-regulated kinase-dependent pathway Christian K Tipsmark, Christina N Strom, Sean T Bailey and Russell J Borski Department of Zoology, North Carolina State University, Raleigh, North Carolina 27695-7617, USA (Correspondence should be addressed to C K Tipsmark who is now at Institute of Biology, University of Southern Denmark, Campusvej 55, DK-5230 Odense M, Denmark; Email: [email protected]) Abstract Leptin was initially identified as a regulator of appetite and leptin are mediated by the activation of extracellular signal- weight control centers in the hypothalamus, but appears to be regulated kinase (ERK1/2) but nothing is known about the involved in a number of physiological processes. This study cellular mechanisms by which leptin might regulate PRL was carried out to examine the possible role of leptin in secretion in vertebrates. We therefore tested whether regulating prolactin (PRL) release using the teleost pituitary ERK1/2 might be involved in the leptin PRL response and model system. This advantageous system allows isolation of a found that the ERK inhibitor, PD98059, hindered leptin- nearly pure population of lactotropes in their natural, in situ induced PRL release. We further analyzed leptin response by aggregated state. The rostral pars distalis were dissected from quantifying tyrosine and threonine phosphorylation of tilapia pituitaries and exposed to varying concentrations of ERK1/2 using western blots. One hour incubation with leptin (0, 1, 10, 100 nM) for 1 h. Release of PRL was leptin induced a concentration-dependent increase in stimulated by leptin in a potent and concentration-dependent phosphorylated, and thus active, ERK1/2. -

Interleukin 6 Inhibits Mouse Placental Lactogen II but Not Mouse Placental Lactogen I Secretion in Vitro (Trophoblast/Pregnancy/Cytokine) M
Proc. Natl. Acad. Sci. USA Vol. 90, pp. 11905-11909, December 1993 Physiology Interleukin 6 inhibits mouse placental lactogen II but not mouse placental lactogen I secretion in vitro (trophoblast/pregnancy/cytokine) M. YAMAGUCHI*t, L. OGREN*, J. N. SOUTHARD*, H. KURACHI*, A. MIYAKEt, AND F. TALAMANTES*§ *Department of Biology, University of California, Santa Cruz, CA 95064; and tDepartment of Obstetrics and Gynecology, Osaka University Medical School, Osaka, Japan 565 Communicated by George E. Seidel, Jr., September 7, 1993 (receivedfor review June 9, 1993) ABSTRACT The mouse placenta produces several poly- members ofthe PRL-GH gene family. We have used primary peptides belonging to the prolactin-growth hormone gene fam- cultures of placental cells from several days of pregnancy to ily, including mouse placental lactogen (mPL) I and mPL-II. demonstrate that IL-6 regulates the secretion of mPL-II, but The present study was undertaken to determine whether the not mPL-I, and that the sensitivity of mPL-II secretion to secretion of mPL-I and mPL-H is regulated by interleukin 6 IL-6 varies during gestation. (IL-6), which is present in the placenta and has previously been reported to stimulate the secretion ofpituitary members of this gene family. Effects of human and mouse IL-6 on mPL-I and MATERIALS AND METHODS mPL-II secretion were examined in primary cultures of pla- Hormones, Cytokines, and Antisera. mPL-II and recombi- cental cells from days 7, 9, and 12 of pregnancy. IL-6 caused nant mPL-I were purified as described (10, 16). Rabbit a dose-dependent reduction in the mPL-HI concentration in the anti-mPL-I and rabbit anti-mPL-II antisera have been de- medium of cells from days 9 and 12 of pregnancy but did not scribed (11, 16). -

Expression of Urocortin and Corticotropin-Releasing Hormone Receptors in the Horse Thyroid Gland
Cell Tissue Res DOI 10.1007/s00441-012-1450-4 REGULAR ARTICLE Expression of urocortin and corticotropin-releasing hormone receptors in the horse thyroid gland Caterina Squillacioti & Adriana De Luca & Sabrina Alì & Salvatore Paino & Giovanna Liguori & Nicola Mirabella Received: 11 January 2012 /Accepted: 3 May 2012 # Springer-Verlag 2012 Abstract Urocortin (UCN) is a 40-amino-acid peptide and UCN, CRHR1 and CRHR2 and that UCN plays a role in a member of the corticotropin-releasing hormone (CRH) the regulation of thyroid physiological functions through a family, which includes CRH, urotensin I, sauvagine, paracrine mechanism. UCN2 and UCN3. The biological actions of CRH family peptides are mediated via two types of G-protein-coupled Keywords Follicular cells . C-cells . RT-PCR . receptors, namely CRH type 1 receptor (CRHR1) and CRH Immunohistochemistry . Horse type 2 receptor (CRHR2). The biological effects of these peptides are mediated and modulated not only by CRH receptors but also via a highly conserved CRH-binding Introduction protein (CRHBP). Our aim was to investigate the expression of UCN, CRHR1, CRHR2 and CRHBP by immunohisto- Urocortin (UCN) is a peptide of 40 amino acids and is a chemistry, Western blot and reverse transcription with the member of the corticotropin-releasing hormone (CRH) fam- polymerase chain reaction (RT-PCR) in the horse thyroid ily, which includes CRH, urotensin I, sauvagine, UCN2 and gland. The results showed that UCN, CRHR1 and CRHR2 UCN3. Vaughan et al. (1995) were the first to identify UCN, were expressed in the thyroid gland, whereas CRHBP was which exhibits 45% homology to CRH (Latchman 2002; not expressed. -

Searching for Novel Peptide Hormones in the Human Genome Olivier Mirabeau
Searching for novel peptide hormones in the human genome Olivier Mirabeau To cite this version: Olivier Mirabeau. Searching for novel peptide hormones in the human genome. Life Sciences [q-bio]. Université Montpellier II - Sciences et Techniques du Languedoc, 2008. English. tel-00340710 HAL Id: tel-00340710 https://tel.archives-ouvertes.fr/tel-00340710 Submitted on 21 Nov 2008 HAL is a multi-disciplinary open access L’archive ouverte pluridisciplinaire HAL, est archive for the deposit and dissemination of sci- destinée au dépôt et à la diffusion de documents entific research documents, whether they are pub- scientifiques de niveau recherche, publiés ou non, lished or not. The documents may come from émanant des établissements d’enseignement et de teaching and research institutions in France or recherche français ou étrangers, des laboratoires abroad, or from public or private research centers. publics ou privés. UNIVERSITE MONTPELLIER II SCIENCES ET TECHNIQUES DU LANGUEDOC THESE pour obtenir le grade de DOCTEUR DE L'UNIVERSITE MONTPELLIER II Discipline : Biologie Informatique Ecole Doctorale : Sciences chimiques et biologiques pour la santé Formation doctorale : Biologie-Santé Recherche de nouvelles hormones peptidiques codées par le génome humain par Olivier Mirabeau présentée et soutenue publiquement le 30 janvier 2008 JURY M. Hubert Vaudry Rapporteur M. Jean-Philippe Vert Rapporteur Mme Nadia Rosenthal Examinatrice M. Jean Martinez Président M. Olivier Gascuel Directeur M. Cornelius Gross Examinateur Résumé Résumé Cette thèse porte sur la découverte de gènes humains non caractérisés codant pour des précurseurs à hormones peptidiques. Les hormones peptidiques (PH) ont un rôle important dans la plupart des processus physiologiques du corps humain. -

Placental Growth Hormone-Related Proteins and Prolactin-Related Proteins
Placental Growth Hormone-Related Proteins and Prolactin-Related Proteins The Harvard community has made this article openly available. Please share how this access benefits you. Your story matters Citation Haig, D. 2008. Placental growth hormone-related proteins and prolactin-related proteins. Placenta 29: 36-41. Published Version doi:10.1016/j.placenta.2007.09.010 Citable link http://nrs.harvard.edu/urn-3:HUL.InstRepos:11148777 Terms of Use This article was downloaded from Harvard University’s DASH repository, and is made available under the terms and conditions applicable to Other Posted Material, as set forth at http:// nrs.harvard.edu/urn-3:HUL.InstRepos:dash.current.terms-of- use#LAA Placental growth hormone-related proteins and prolactin-related proteins. David Haig Department of Organismic and Evolutionary Biology, Harvard University, 26 Oxford Street, Cambridge MA 02138. e-mail: [email protected] phone: 617-496-5125 fax: 617-495-5667 Keywords: GH, PRL, placenta, endometrial glands, placental lactogen The placentas of ruminants and muroid rodents express prolactin (PRL)-related genes whereas the placentas of anthropoid primates express growth hormone (GH)-related genes. The evolution of placental expression is associated with acclerated evolution of the corresponding pituitary hormone and destabilization of conserved endocrine systems. In particular, placental hormones often evolve novel interactions with new receptors. The adaptive functions of some placental hormones may be revealed only under conditions of physiological stress. Introduction Placental hormones are produced by offspring, but act on receptors of mothers. As such, placental hormones and maternal receptors are prime candidates for the expression of parent-offspring conflict [1,2]. -
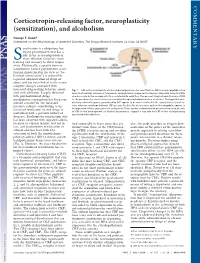
Corticotropin-Releasing Factor, Neuroplasticity (Sensitization), and Alcoholism
COMMENTARY Corticotropin-releasing factor, neuroplasticity (sensitization), and alcoholism George F. Koob* Committee on the Neurobiology of Addictive Disorders, The Scripps Research Institute, La Jolla, CA 92037 ensitization is a ubiquitous bio- logical phenomenon that has a role in the neuroadaptation of many different functions, from Slearning and memory to stress respon- sivity. Historically, a specific form of sensitization termed psychomotor sensi- tization (mistermed in my view as ‘‘be- havioral sensitization’’) is induced by repeated administration of drugs of abuse and has been linked to the neuro- adaptive changes associated with increased drug-seeking behavior associ- Fig. 1. CNS actions relevant to alcohol-induced psychomotor sensitization. CRF is a neuropeptide in the ated with addiction. Largely observed brain that controls autonomic, hormonal, and behavioral responses to stressors. New data show that CRF with psychostimulant drugs, also has a role in the neuroplasticity associated with addiction. The present study extends the role of CRF psychomotor sensitization has been con- to the psychomotor sensitization associated with repeated administration of alcohol. The hypothalamic- sidered a model for the increased pituitary-adrenal responses produced by CRF appear to be more involved in the acquisition of sensitiza- incentive salience contributing to the tion, whereas extrahypothalamic CRF systems, likely to be in structures such as the amygdala, appear to increased motivation to seek drugs in be important for the expression of sensitization. These results, combined with previous studies on the role of CRF in the development of alcohol dependence, suggest a key role for CRF in the neuroplasticity individuals with a previous history of associated with addiction. -

Urocortin 2 Modulates Glucose Utilization and Insulin Sensitivity in Skeletal Muscle
Urocortin 2 modulates glucose utilization and insulin sensitivity in skeletal muscle Alon Chen*†, Bhawanjit Brar*, Cheol Soo Choi‡, David Rousso*, Joan Vaughan*, Yael Kuperman†, Shee Ne Kim‡, Cindy Donaldson*, Sean M. Smith*, Pauline Jamieson*, Chien Li*, Tim R. Nagy§, Gerald I. Shulman‡, Kuo-Fen Lee*, and Wylie Vale*¶ *Clayton Foundation Laboratories for Peptide Biology, The Salk Institute for Biological Studies, La Jolla, CA 92037; †Department of Neurobiology, The Weizmann Institute of Science, Rehovot 76100, Israel; ‡Department of Internal Medicine and Cellular and Molecular Physiology and Howard Hughes Medical Institute, Yale University School of Medicine, New Haven, CT 06536; and §Department of Nutrition Sciences, University of Alabama at Birmingham, Birmingham, AL 35294 Contributed by Wylie Vale, August 28, 2006 Skeletal muscle is the principal tissue responsible for insulin- Results and Discussion stimulated glucose disposal and is a major site of peripheral insulin Generation of Ucn 2-Deficient Mice. To explore the physiological resistance. Urocortin 2 (Ucn 2), a member of the corticotropin- role of Ucn 2, we generated mice that are deficient in this releasing factor (CRF) family, and its cognate type 2 CRF receptor peptide. A genomic DNA clone containing Ucn 2 was isolated, (CRFR2) are highly expressed in skeletal muscle. To determine the and a targeting construct in which the full Ucn 2 coding sequence physiological role of Ucn 2, we generated mice that are deficient in was replaced with a neomycin-resistant gene cassette was gen- this peptide. Using glucose-tolerance tests (GTTs), insulin-tolerance erated (Fig. 1a). J1 ES cells were electroporated with the tests (ITTs), and hyperinsulinemic euglycemic glucose clamp stud- targeting construct and were selected as described in ref. -

Multi-Functionality of Proteins Involved in GPCR and G Protein Signaling: Making Sense of Structure–Function Continuum with In
Cellular and Molecular Life Sciences (2019) 76:4461–4492 https://doi.org/10.1007/s00018-019-03276-1 Cellular andMolecular Life Sciences REVIEW Multi‑functionality of proteins involved in GPCR and G protein signaling: making sense of structure–function continuum with intrinsic disorder‑based proteoforms Alexander V. Fonin1 · April L. Darling2 · Irina M. Kuznetsova1 · Konstantin K. Turoverov1,3 · Vladimir N. Uversky2,4 Received: 5 August 2019 / Revised: 5 August 2019 / Accepted: 12 August 2019 / Published online: 19 August 2019 © Springer Nature Switzerland AG 2019 Abstract GPCR–G protein signaling system recognizes a multitude of extracellular ligands and triggers a variety of intracellular signal- ing cascades in response. In humans, this system includes more than 800 various GPCRs and a large set of heterotrimeric G proteins. Complexity of this system goes far beyond a multitude of pair-wise ligand–GPCR and GPCR–G protein interactions. In fact, one GPCR can recognize more than one extracellular signal and interact with more than one G protein. Furthermore, one ligand can activate more than one GPCR, and multiple GPCRs can couple to the same G protein. This defnes an intricate multifunctionality of this important signaling system. Here, we show that the multifunctionality of GPCR–G protein system represents an illustrative example of the protein structure–function continuum, where structures of the involved proteins represent a complex mosaic of diferently folded regions (foldons, non-foldons, unfoldons, semi-foldons, and inducible foldons). The functionality of resulting highly dynamic conformational ensembles is fne-tuned by various post-translational modifcations and alternative splicing, and such ensembles can undergo dramatic changes at interaction with their specifc partners. -

The Importance of Determining Human
JMB 2009; 28 (2) DOI: 10.2478/v10011-009-0003-1 UDK 577.1 : 61 ISSN 1452-8258 JMB 28: 97–100, 2009 Original paper Originalni nau~ni rad THE IMPORTANCE OF DETERMINING HUMAN PLACENTAL LACTOGEN IN THE THIRD TRIMESTER OF PREGNANCY ZNA^AJ ODRE\IVANJA HUMANOG PLACENTALNOG LAKTOGENA U TRE]EM TRIMESTRU TRUDNO]E Jasmina Durkovi}1, Bojana Mandi}2 1Department of Genetics , Town Hospital, Subotica, Serbia 2Mega Lab, Biochemical Laboratory, Subotica, Serbia Summary: Human placental lactogen (HPL) is a hormone Kratak sadr`aj: Humani placentalni laktogen (HPL) jeste produced by the placenta with a role in the re gu lation of hormon koji izlu~uje placenta i regulator je fetoplacen- fetoplacental growth. In this paper, the results of HPL de- talnog rasta. U radu su prikazani rezultati odre|ivanja HPL- termination in the third trimester of pregnancy are pre- a u tre}em trimestru trudno}e, sa ciljem da se ispita senzi - sented with the aim of testing the sensitivity of this bio - tivnost tog biohemijskog markera za otkrivanje poreme}aja chemical marker for detecting placental dysfunction, fetal funkcije placente, vitaliteta fetusa i rizika za lo{ ishod. vitality and risk of bad outcome. The tests were performed Ispitivanje je obavljeno na uzorku od 370 rizi~nih trudno}a on 370 women with high-risk pregnancy, between the 20th izme|u 20 i 36 nedelje trudno}e. HPL je odre|en ELISA and 36th week of pregnancy. HPL was determined by an metodom, testovima »Bioserv Diagnostics«, a rezultati su ELISA method using Bioserv Diagnostics tests and the o~itani na rideru »STAT–FAX 303+«. -

The Endocrine Function of the Placenta: Interactions Between Trophic Peptides and Hormones
Placenlal Function and Fetal Nutrition, edited by Frederick C. Battaglia, Nestle Nutrition Workshop Series. Vol. 39. Nestec Ltd.. Vevey/Lippincott-Raven Publishers. Philadelphia. © 1997. The Endocrine Function of the Placenta: Interactions Between Trophic Peptides and Hormones Lise Cedard U. 361 INSERM, Maternite Baudelocque, 121 Boulevard de Port-Royal Paris, France The placenta is classically considered to be a source of hormones that play an important role in the establishment and maintenance of pregnancy. Because of its hemochorial structure, the human placenta produces hormones and easily secretes them into the maternal circulation. They include steroid hormones, such as estrogens and progesterone, and protein hormones, the most important of which are the chori- onic gonadotrophins (hCG), discovered in 1927. During the period from 1960 to 1980, the concept of a fetoplacental unit (1) with a complementary role for the fetus in steroid production offered a new approach to steroid metabolism, and since then new proteins and neuropeptides have been discovered (2,3). However, the lack of innervation of placental tissue highlights the importance of understanding neuroen- docrine regulation of placental physiology. Sophisticated culture methods enabling the study of biologic phenomena occurring during transformation of cytotrophoblast cells into a syncytiotrophoblast (4) have been combined with electron microscopy and histochemistry. Possibilities offered by molecular biology, including in situ hybridization and molecular cloning studies,