Apple Regulated Substances Specification 069-0135-L
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Chemical Characterization and Thermal Stressing Studies of Perfluorohexane Fluids for Space-Based Applications
Chemical Characterization and Thermal Stressing Studies of Perfluorohexane Fluids for Space-Based Applications William A. Arnold, Ph.D.1 ZIN Technologies, Inc., Brook Park, Ohio, 44142, USA Thomas G. Hartman, Ph.D.2 CAFT, Cook College, Rutgers the Sate University of New Jersey, New Brunswick, NJ, 08901 USA John McQuillen,3 NASA Glenn Research Center, Cleveland, Ohio, 44135, USA Perfluorohexane (PFH), C6F14, is a perfluorocarbon fluid. Several PFH fluids with different isomer concentrations were evaluated for use in an upcoming NASA space experiment. Samples tested included two commercially obtained high-purity n-perfluorohexane (n-PFH) fluids and a technical grade mixture of C6F14 branched and linear isomers (FC-72). These fluids were evaluated for exact chemical composition, impurity purity and high temperature degradation behavior (pyrolysis). Our investigation involved simulated thermal stressing studies of PFH fluids under conditions likely to occur in the event of an atmospheric breach within the International Space Station (ISS) and subsequent exposure of the vapors to the high temperature and catalyst present in its Trace Contaminant Control Subsystem (TCCS). Exposure to temperatures in the temperature range of 200-450°C in an inert or oxidizing atmosphere, with and without the presence of catalyst was investigated. The most aggressive conditions studied were exposure of PFH vapors to 450°C in air and in the presence of TCCS (palladium) catalyst. Gas chromatography-mass spectrometry (GC-MS) and gas chromatography (GC) analyses were conducted on the perfluorohexane samples before and after pyrolysis. The FC-72 and n-PFH samples showed no significant degradation following pyrolysis even under the most aggressive study conditions. -

Transport of Dangerous Goods
ST/SG/AC.10/1/Rev.16 (Vol.I) Recommendations on the TRANSPORT OF DANGEROUS GOODS Model Regulations Volume I Sixteenth revised edition UNITED NATIONS New York and Geneva, 2009 NOTE The designations employed and the presentation of the material in this publication do not imply the expression of any opinion whatsoever on the part of the Secretariat of the United Nations concerning the legal status of any country, territory, city or area, or of its authorities, or concerning the delimitation of its frontiers or boundaries. ST/SG/AC.10/1/Rev.16 (Vol.I) Copyright © United Nations, 2009 All rights reserved. No part of this publication may, for sales purposes, be reproduced, stored in a retrieval system or transmitted in any form or by any means, electronic, electrostatic, magnetic tape, mechanical, photocopying or otherwise, without prior permission in writing from the United Nations. UNITED NATIONS Sales No. E.09.VIII.2 ISBN 978-92-1-139136-7 (complete set of two volumes) ISSN 1014-5753 Volumes I and II not to be sold separately FOREWORD The Recommendations on the Transport of Dangerous Goods are addressed to governments and to the international organizations concerned with safety in the transport of dangerous goods. The first version, prepared by the United Nations Economic and Social Council's Committee of Experts on the Transport of Dangerous Goods, was published in 1956 (ST/ECA/43-E/CN.2/170). In response to developments in technology and the changing needs of users, they have been regularly amended and updated at succeeding sessions of the Committee of Experts pursuant to Resolution 645 G (XXIII) of 26 April 1957 of the Economic and Social Council and subsequent resolutions. -

Disinfection Byproducts in Chlorinated Drinking Water
International Journal of Water and Wastewater Treatment SciO p Forschene n HUB for Sc i e n t i f i c R e s e a r c h ISSN 2381-5299 | Open Access RESEARCH ARTICLE Volume 4 - Issue 2 | DOI: http://dx.doi.org/10.16966/2381-5299.156 Disinfection Byproducts in Chlorinated Drinking Water Malia Vester1, Hany F Sobhi2 and Mintesinot Jiru1* 1Department of Natural Sciences, Coppin State University, Maryland, USA 2Center for Organic Synthesis, Department of Natural Sciences, Coppin State University, Maryland, USA *Corresponding author: Mintesinot Jiru, Department of Natural Sciences, Coppin State University, Maryland, USA, Tel: 410-951-4139; E-mail: [email protected] Received: 07 Jul, 2018 | Accepted: 14 Nov, 2018 | Published: 20 Nov, 2018 Citation: Vester M, Sobhi HF, Jiru M (2018) Disinfection Byproducts in Chlorinated Drinking Water. Int J Water Wastewater Treat 4(2): dx.doi. org/10.16966/2381-5299.156 Copyright: © Vester M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Abstract Trihalomethanes (THMs) are disinfection byproducts formed through the reaction of chlorine and organic matter. There are four forms of Trihalomethanes: chloroform, bromoform, dibromochloromethane and bromodichloromethane. Studies have shown that chloroform, a principal disinfection byproduct, is carcinogenic in rodents. Although the presence of these hazardous materials are constantly being monitored by Water Treatment Plant and the Environmental Protection Agency (EPA), they continue to be present at an unpredictable levels and the full effects of these byproducts are not fully understood. -

Cylinder Valve Selection Quick Reference for Valve Abbreviations
SHERWOOD VALVE COMPRESSED GAS PRODUCTS Appendix Cylinder Valve Selection Quick Reference for Valve Abbreviations Use the Sherwood Cylinder Valve Series Abbreviation Chart on this page with the Sherwood Cylinder Valve Selection Charts found on pages 73–80. The Sherwood Cylinder Valve Selection Chart are for reference only and list: • The most commonly used gases • The Compressed Gas Association primary outlet to be used with each gas • The Sherwood valves designated for use with this gas • The Pressure Relief Device styles that are authorized by the DOT for use with these gases PLEASE NOTE: The Sherwood Cylinder Valve Selection Charts are partial lists extracted from the CGA V-1 and S-1.1 pamphlets. They can change without notice as the CGA V-1 and S-1.1 pamphlets are amended. Sherwood will issue periodic changes to the catalog. If there is any discrepancy or question between these lists and the CGA V-1 and S-1.1 pamphlets, the CGA V-1 and S-1.1 pamphlets take precedence. Sherwood Cylinder Valve Series Abbreviation Chart Abbreviation Sherwood Valve Series AVB Small Cylinder Acetylene Wrench-Operated Valves AVBHW Small Cylinder Acetylene Handwheel-Operated Valves AVMC Small Cylinder Acetylene Wrench-Operated Valves AVMCHW Small Cylinder Acetylene Handwheel-Operated Valves AVWB Small Cylinder Acetylene Wrench-Operated Valves — WB Style BV Hi/Lo Valves with Built-in Regulator DF* Alternative Energy Valves GRPV Residual Pressure Valves GV Large Cylinder Acetylene Valves GVT** Vertical Outlet Acetylene Valves KVAB Post Medical Valves KVMB Post Medical Valves NGV Industrial and Chrome-Plated Valves YVB† Vertical Outlet Oxygen Valves 1 * DF Valves can be used with all gases; however, the outlet will always be ⁄4"–18 NPT female. -

Estimation of Enthalpy of Bio-Oil Vapor and Heat Required For
Edinburgh Research Explorer Estimation of Enthalpy of Bio-Oil Vapor and Heat Required for Pyrolysis of Biomass Citation for published version: Yang, H, Kudo, S, Kuo, H-P, Norinaga, K, Mori, A, Masek, O & Hayashi, J 2013, 'Estimation of Enthalpy of Bio-Oil Vapor and Heat Required for Pyrolysis of Biomass', Energy & Fuels, vol. 27, no. 5, pp. 2675-2686. https://doi.org/10.1021/ef400199z Digital Object Identifier (DOI): 10.1021/ef400199z Link: Link to publication record in Edinburgh Research Explorer Document Version: Early version, also known as pre-print Published In: Energy & Fuels General rights Copyright for the publications made accessible via the Edinburgh Research Explorer is retained by the author(s) and / or other copyright owners and it is a condition of accessing these publications that users recognise and abide by the legal requirements associated with these rights. Take down policy The University of Edinburgh has made every reasonable effort to ensure that Edinburgh Research Explorer content complies with UK legislation. If you believe that the public display of this file breaches copyright please contact [email protected] providing details, and we will remove access to the work immediately and investigate your claim. Download date: 04. Oct. 2021 Estimation of Enthalpy of Bio-oil Vapor and Heat Required for Pyrolysis of Biomass Hua Yang,† Shinji Kudo,§ Hsiu-Po Kuo,‡ Koyo Norinaga, ξ Aska Mori, ξ Ondřej Mašek,|| and Jun-ichiro Hayashi †, ξ, §, * †Interdisciplinary Graduate School of Engineering Sciences, Kyushu University, -

Toxicological Profile for Bromodichloromethane
BROMODICHLOROMETHANE 89 CHAPTER 5. POTENTIAL FOR HUMAN EXPOSURE 5.1 OVERVIEW Bromodichloromethane has been identified in at least 238 of the 1,854 hazardous waste sites that have been proposed for inclusion on the EPA National Priorities List (NPL) (ATSDR 2017). However, the number of sites in which bromodichloromethane has been evaluated is not known. The number of sites in each state is shown in Figure 5-1. Of these sites, 233 are located within the United States, 2 are located in the Virgin Islands, and 3 are located in Puerto Rico (not shown). Figure 5-1. Number of NPL Sites with Bromodichloromethane Contamination • The most likely route of exposure for the general public to bromodichloromethane is through ingestion, inhalation, and dermal contact of chlorinated drinking water. • A median bromodichloromethane intake of 2.8–4.2 µg/day from drinking water has been estimated; inhalation and dermal exposure would add to this daily intake. BROMODICHLOROMETHANE 90 5. POTENTIAL FOR HUMAN EXPOSURE • Bromodichloromethane is formed as a byproduct of water disinfection methods using chlorination. This is the primary source of bromodichloromethane in the environment. • Its principal use is as a chemical intermediate for organic synthesis and as a chemical reagent. • Volatilization is an important fate process. Bromodichloromethane evaporates from sources and enters the environment as a gas, which is slowly broken down in air. Residual bromodichloromethane may be broken down slowly by bacteria. • In the atmosphere, bromodichloromethane is thought to undergo slow degradation through oxidative pathways, with a half-life of about 2–3 months. 5.2 PRODUCTION, IMPORT/EXPORT, USE, AND DISPOSAL 5.2.1 Production The principal anthropogenic source of bromodichloromethane is its unintentional formation as a byproduct during the chlorination of water containing organic materials and bromide. -

Human Health Toxicity Values for Perfluorobutane Sulfonic Acid (CASRN 375-73-5) and Related Compound Potassium Perfluorobutane Sulfonate (CASRN 29420 49 3)
EPA-823-R-18-307 Public Comment Draft Human Health Toxicity Values for Perfluorobutane Sulfonic Acid (CASRN 375-73-5) and Related Compound Potassium Perfluorobutane Sulfonate (CASRN 29420-49-3) This document is a Public Comment draft. It has not been formally released by the U.S. Environmental Protection Agency and should not at this stage be construed to represent Agency policy. This information is distributed solely for the purpose of public review. This document is a draft for review purposes only and does not constitute Agency policy. DRAFT FOR PUBLIC COMMENT – DO NOT CITE OR QUOTE NOVEMBER 2018 Human Health Toxicity Values for Perfluorobutane Sulfonic Acid (CASRN 375-73-5) and Related Compound Potassium Perfluorobutane Sulfonate (CASRN 29420 49 3) Prepared by: U.S. Environmental Protection Agency Office of Research and Development (8101R) National Center for Environmental Assessment Washington, DC 20460 EPA Document Number: 823-R-18-307 NOVEMBER 2018 This document is a draft for review purposes only and does not constitute Agency policy. DRAFT FOR PUBLIC COMMENT – DO NOT CITE OR QUOTE NOVEMBER 2018 Disclaimer This document is a public comment draft for review purposes only. This information is distributed solely for the purpose of public comment. It has not been formally disseminated by EPA. It does not represent and should not be construed to represent any Agency determination or policy. Mention of trade names or commercial products does not constitute endorsement or recommendation for use. i This document is a draft for review purposes only and does not constitute Agency policy. DRAFT FOR PUBLIC COMMENT – DO NOT CITE OR QUOTE NOVEMBER 2018 Authors, Contributors, and Reviewers CHEMICAL MANAGERS Jason C. -

Evaluating Analytical Methods for Detecting Unknown Chemicals in Recycled Water
PROJECT NO. 4992 Evaluating Analytical Methods for Detecting Unknown Chemicals in Recycled Water Evaluating Analytical Methods for Detecting Unknown Chemicals in Recycled Water Prepared by: Keith A. Maruya Charles S. Wong Southern California Coastal Water Research Project Authority 2020 The Water Research Foundation (WRF) is a nonprofit (501c3) organization which provides a unified source for One Water research and a strong presence in relationships with partner organizations, government and regulatory agencies, and Congress. The foundation conducts research in all areas of drinking water, wastewater, stormwater, and water reuse. The Water Research Foundation’s research portfolio is valued at over $700 million. The Foundation plays an important role in the translation and dissemination of applied research, technology demonstration, and education, through creation of research‐based educational tools and technology exchange opportunities. WRF serves as a leader and model for collaboration across the water industry and its materials are used to inform policymakers and the public on the science, economic value, and environmental benefits of using and recovering resources found in water, as well as the feasibility of implementing new technologies. For more information, contact: The Water Research Foundation Alexandria, VA Office Denver, CO Office 1199 North Fairfax Street, Suite 900 6666 West Quincy Avenue Alexandria, VA 22314‐1445 Denver, Colorado 80235‐3098 Tel: 571.384.2100 Tel: 303.347.6100 www.waterrf.org [email protected] ©Copyright 2020 by The Water Research Foundation. All rights reserved. Permission to copy must be obtained from The Water Research Foundation. WRF ISBN: 978‐1‐60573‐503‐0 WRF Project Number: 4992 This report was prepared by the organization(s) named below as an account of work sponsored by The Water Research Foundation. -
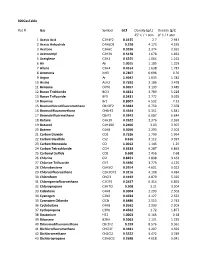
Gas Conversion Factor for 300 Series
300GasTable Rec # Gas Symbol GCF Density (g/L) Density (g/L) 25° C / 1 atm 0° C / 1 atm 1 Acetic Acid C2H4F2 0.4155 2.7 2.947 2 Acetic Anhydride C4H6O3 0.258 4.173 4.555 3 Acetone C3H6O 0.3556 2.374 2.591 4 Acetonitryl C2H3N 0.5178 1.678 1.832 5 Acetylene C2H2 0.6255 1.064 1.162 6 Air Air 1.0015 1.185 1.293 7 Allene C3H4 0.4514 1.638 1.787 8 Ammonia NH3 0.7807 0.696 0.76 9 Argon Ar 1.4047 1.633 1.782 10 Arsine AsH3 0.7592 3.186 3.478 11 Benzene C6H6 0.3057 3.193 3.485 12 Boron Trichloride BCl3 0.4421 4.789 5.228 13 Boron Triflouride BF3 0.5431 2.772 3.025 14 Bromine Br2 0.8007 6.532 7.13 15 Bromochlorodifluoromethane CBrClF2 0.3684 6.759 7.378 16 Bromodifluoromethane CHBrF2 0.4644 5.351 5.841 17 Bromotrifluormethane CBrF3 0.3943 6.087 6.644 18 Butane C4H10 0.2622 2.376 2.593 19 Butanol C4H10O 0.2406 3.03 3.307 20 Butene C4H8 0.3056 2.293 2.503 21 Carbon Dioxide CO2 0.7526 1.799 1.964 22 Carbon Disulfide CS2 0.616 3.112 3.397 23 Carbon Monoxide CO 1.0012 1.145 1.25 24 Carbon Tetrachloride CCl4 0.3333 6.287 6.863 25 Carbonyl Sulfide COS 0.668 2.456 2.68 26 Chlorine Cl2 0.8451 2.898 3.163 27 Chlorine Trifluoride ClF3 0.4496 3.779 4.125 28 Chlorobenzene C6H5Cl 0.2614 4.601 5.022 29 Chlorodifluoroethane C2H3ClF2 0.3216 4.108 4.484 30 Chloroform CHCl3 0.4192 4.879 5.326 31 Chloropentafluoroethane C2ClF5 0.2437 6.314 6.892 32 Chloropropane C3H7Cl 0.308 3.21 3.504 33 Cisbutene C4H8 0.3004 2.293 2.503 34 Cyanogen C2N2 0.4924 2.127 2.322 35 Cyanogen Chloride ClCN 0.6486 2.513 2.743 36 Cyclobutane C4H8 0.3562 2.293 2.503 37 Cyclopropane C3H6 0.4562 -

Prepared by 3M
DRAFT ECOTOXICOLOGY AND ENVIRONMENTAL FATE TESTING OF SHORT CHAIN PERFLUOROALKYL COMPOUNDS RELATED TO 3M CHEMISTRIES XXX XX, 2008 Prepared by 3M Page 1 CONFIDENTIAL - SUBJECT TO A PROTECTIVE ORDER ENTERED IN 3M MN01537089 HENNEPIN COUNTY DISTRICT COURT, NO. 27-CV-10-28862 2231.0001 DRAFT TABLE OF CONTENTS Page Introduction (describes propose, goals, document organization .... ) 3 1) Degradation modes 4-27 Basic degradation pathways, Conversion routes from polymer to monomer & degradants or residual intermediates to degradants) 2) Degradants & Intermediates - Phys. Chem properties and test data by section & chapter i) Fluorinated Sulfonic acids and derivatives a) PFBS (PBSK) 28-42 b) PFBSI 43-44 ii) Fluorinated Carboxylates a) TFA 46-55 b) PFPA 56-59 c) PFBA (APFB) 60-65 d) MeFBSAA (MeFBSE Acid, M370) 66-68 iii) Fluorinated Non-ionics a) MeFBSE 70-74 b) MeFBSA 75-80 c) FBSE 81-84 d) FBSA 85-88 e) HxFBSA 89-91 iv) Fluorinated Inerts & volatiles a) PBSF 94-97 b) NFB, C4 hydride, CF3CF2CF2CF2-H 98-99 3) Assessments X Glossary 100-102 References/Endnotes 105- Page 2 CONFIDENTIAL - SUBJECT TO A PROTECTIVE ORDER ENTERED IN 3M MN01537090 HENNEPIN COUNTY DISTRICT COURT, NO. 27-CV-10-28862 2231.0002 DRAFT Introduction to Environmental White Papers on Perfluoroalkyl acids related to 3M chemistries Perfluorochemicals have been commonplace in chemical industry over 50 years but until recently there has been little information on environmental fate and effects avialble in open literature. The following chapters summarize the findings of"list specific C4 intermediates PFBS, PFBSI, PFBA, PFPA, MeFBSAA, TFA, MeFBSE, FBSA, HxFBSA, FBSE, PBSF, NFB " As background, 3M announced on May 16, 2000 the voluntary manufacturing phase out of perfluorooctanyl chemicals which included perfluorooctanoic acid (PFOA), perfluorooctane sulfonate (PFOS), and PFOS-related chemistries. -

Summary of Information for ABC for Polycyclic Aromatic Hydrocarbons
Air Toxics Science Advisory Committee Summary of Information for ABC for Polycyclic Aromatic Hydrocarbons September 16, 2015 ATSAC Meeting #10 Presenter: Sue MacMillan, DEQ ATSAC lead Sue MacMillan | Oregon Department of Environmental Quality 26 Individual PAHs to Serve as Basis of ABC for Total PAHs Acenaphthene Cyclopenta(c,d)pyrene Acenaphthylene Dibenzo(a,h)anthracene Anthracene Dibenzo(a,e)pyrene Anthanthrene Dibenzo(a,h)pyrene Benzo(a)pyrene Dibenzo(a,i)pyrene Naphthalene Benz(a)anthracene Dibenzo(a,l)pyrene has separate Benzo(b)fluoranthene Fluoranthene ABC. Benzo(k)fluoranthene Fluorene Benzo( c)pyrene Indeno(1,2,3-c,d)pyrene Benzo(e)pyrene Phenanthrene Benzo(g,h,i)perylene Pyrene Benzo(j)fluoranthene 5-Methylchrysene Chrysene 6-Nitrochrysene Use of Toxic Equivalency Factors for PAHs • Benzo(a)pyrene serves as the index PAH, and has a documented toxicity value to which other PAHs are adjusted • Other PAHs adjusted using Toxic Equivalency Factors (TEFs), aka Potency Equivalency Factors (PEFs). These values are multipliers and are PAH-specific. • Once all PAH concentrations are adjusted to account for their relative toxicity as compared to BaP, the concentrations are summed • This summed concentration is then compared to the toxicity value for BaP, which is used as the ABC for total PAHs. Source of PEFs for PAHs • EPA provides a range of values of PEFs for each PAH • Original proposal suggested using upper-bound value of each PEF range as the PEF to use for adjustment of our PAHs • Average PEF value for each PAH is a better approximation of central tendency, and is consistent with the use of PEFs by other agencies • Result of using average, rather than upper-bound PEFs: slightly lower summed concentrations for adjusted PAHs, thus less apt to exceed ABC for total PAHs Documents can be provided upon request in an alternate format for individuals with disabilities or in a language other than English for people with limited English skills. -
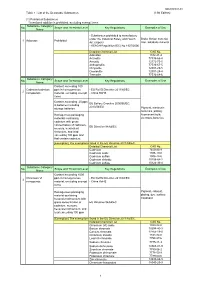
Table 1 : List of the Declarable Substances (1) Prohibited
DQ2000001-03 Table 1 : List of the Declarable Substances (13th Edition) (1) Prohibited Substances *Intentional addition is prohibited, excluding exempt items Substance Category/ No. Scope and Threshold Level Key Regulations Examples of Use Name - Substances prohibited to manufacture under the Industrial Safety and Health Brake friction material, 1 Asbestos Prohibited Act (Japan) filler, adiabatic material - REACH Regulation (EC) No 1907/2006 Detailed Chemical List CAS No. Asbestos 1332-21-4 Actinolite 77536-66-4 Amosite 12172-73-5 Anthophyllite 77536-67-5 Chrysotile 12001-29-5 Crocidolite 12001-28-4 Tremolite 77536-68-6 Substance Category/ No. Scope and Threshold Level Key Regulations Examples of Use Name Content exceeding 100 Cadmium/cadmium ppm in homogeneous - EU RoHS Directive 2011/65/EC 2 compounds material, excluding exempt - China RoHS items Content exceeding 20 ppm EU Battery Directive 2006/66/EC, in batteries including 2013/56/EU storage batteries Pigment, electronic materials, plating, Homogenous packaging fluorescent bulb, materials containing electrode,batteries cadmium with gross concentration of cadmium, EU Directive 94/62/EC mercury, hexavalent chromium, and lead exceeding 100 ppm and that contain cadmium [Exemption] The exemptions listed in the EU Directive 2011/65/EC. Detailed Chemical List CAS No. Cadmium 7440-43-9 Cadmium oxide 1306-19-0 Cadmium sulfide 1306-23-6 Cadmium chloride 10108-64-2 Cadmium sulfate 10124-36-4 Substance Category/ No. Scope and Threshold Level Key Regulations Examples of Use Name Content exceeding 1000 Chromium VI ppm in homogeneous - EU RoHS Directive 2011/65/EC 3 compounds material, excluding exempt - China RoHS items Homogenous packaging Pigment, catalyst, material containing plating, dye, surface hexavalent chromium with treatment gross concentration of EU Directive 94/62/EC cadmium, mercury, hexavalent chromium, and lead exceeding 100 ppm [Exemption] The exemptions listed in the EU Directive 2011/65/EC.