Third Amended Complaint1 Against Defendants Purdue Pharma
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Pharmaceutical and Medical Device Tort Practice Trusted Advisors to the Health Sciences Industry
Pharmaceutical and Medical Device Tort Practice Trusted Advisors to the Health Sciences Industry Today’s product liability cases reach far beyond one courtroom, requiring more than Product Liability a case-by-case approach. Because we take a strategic approach to litigation, King & “Law Firm of the Spalding’s product liability practice is recognized as one of the best in the nation. Year” in 2016 Recognizing that our clients need to protect their businesses, including the continued viability of their products in the marketplace, our lawyers work together with both Law360 our FDA & Life Sciences practice group and our Government Investigations team to provide a 360° defense. As one client noted, “Cases can spill over into other things _________________ like regulatory issues … King & Spalding can bring it all.” And we do. • The largest health law practice in the nation, according to the American Health Mass Tort / Class Lawyers Association, providing additional depth of experience on healthcare Action “Law Firm regulatory and litigation issues. of the Year” in 2015 • A stable of specialists in the relevant sciences, as well as former FDA and DEA regulatory officials who provide highly sophisticated support to our litigation defense. U.S. News & World • IP attorneys who successfully represent biotechnology and pharmaceutical Report clients in prosecuting, obtaining and defending patents. _________________ Trial Ready When Needed We add to our strategic approach a range of trial experience that is increasingly Life Sciences “Law unusual in firms of our size. We try cases every year throughout the nation. With four Firm of the Year” in Fellows in the American College of Trial Lawyers, an organization whose members 2017 are elected based on actual courtroom experience, we serve notice to opposing counsel that they should not count their cases as settled. -

Purdue Pharma to Pay $270 Million to Settle Historic Oklahoma Opioid Lawsuit
Purdue Pharma to pay $270 million to settle historic Oklahoma opioid lawsuit (CNN) — Purdue Pharma has agreed to pay $270 million to settle a historic lawsuit brought by the Oklahoma attorney general, who accused the OxyContin maker of aggressively marketing the opioid painkiller and fueling a drug epidemic that left thousands dead in the state. The settlement comes after Purdue fought the attorney general in court, seeking to delay the start of the trial, which is scheduled for May 28. "It is a new day in Oklahoma, and for the nation, in our battle against addiction and the opioid epidemic," Attorney General Mike Hunter said Tuesday in Tulsa. Hunter said that $102.5 million of the settlement would be used to help establish a national addiction treatment and research center at Oklahoma State University, with additional payments of $15 million each year for the next five years beginning in 2020. The company will also provide $20 million of addiction treatment and opioid rescue medications to the center over the same five-year time frame. A remaining $12.5 million from the settlement will be used directly to help cities and counties with the opioid crisis. The Sackler family, who founded and own Purdue Pharma, will also contribute $75 million over the next five years to the treatment and research center. "Purdue is very pleased to have reached an agreement with Oklahoma that will help those who are battling addiction now and in the future," Dr. Craig Landau, president and CEO of Purdue Pharma, said in a statement. The lawsuit was brought by Hunter against some of the nation's leading makers of opioid pain medications, alleging that deceptive marketing over the past decade fueled the epidemic in the state. -

Product Hopping 2.0: Getting the FDA to Yank Your Original License Beats Stacking Patents Lars Noah
Marquette Intellectual Property Law Review Volume 19 | Issue 2 Article 2 Product Hopping 2.0: Getting the FDA To Yank Your Original License Beats Stacking Patents Lars Noah Follow this and additional works at: http://scholarship.law.marquette.edu/iplr Part of the Intellectual Property Commons Repository Citation Lars Noah, Product Hopping 2.0: Getting the FDA To Yank Your Original License Beats Stacking Patents, 19 Marq. Intellectual Property L. Rev. 161 (2015). Available at: http://scholarship.law.marquette.edu/iplr/vol19/iss2/2 This Article is brought to you for free and open access by the Journals at Marquette Law Scholarly Commons. It has been accepted for inclusion in Marquette Intellectual Property Law Review by an authorized administrator of Marquette Law Scholarly Commons. For more information, please contact [email protected]. NOAH FINAL (DO NOT DELETE) 5/9/2015 4:17 PM ARTICLES PRODUCT HOPPING 2.0: GETTING THE FDA TO YANK YOUR ORIGINAL LICENSE BEATS STACKING PATENTS LARS NOAH* INTRODUCTION .............................................................................................. 165 I.A PRIMER ON PRODUCT HOPPING IN THE PHARMACEUTICAL INDUSTRY ... 165 II. NEW & IMPROVED PRODUCT HOPPING: THE OXYCONTIN MANEUVER ... 172 CONCLUSION .................................................................................................. 179 * Professor of Law, University of Florida. NOAH FINAL (DO NOT DELETE) 5/9/2015 4:17 PM 162 MARQ. INTELL. PROP. L. REV. [Vol. 19:2 NOAH FINAL (DO NOT DELETE) 5/9/2015 4:17 PM 2015] PRODUCT HOPPING 2.0 163 Lars Noah is a Professor of Law at the Univeersity of Florida, where he has taught courses in Administrative Law, Biommedical Innoovation, Medical Technology, Public Health Law, and Torts, among other subjects. -

PURDUE PHARMA, L.P., a Delaware Limited Partnership, PURDUE PHARMA INC., a New York Corporation, and RICHARD SACKLER, M.D
IN THE CIRCUIT COURT OF BOONE COUNTY, WEST VIRGINIA STATE OF WEST VIRGINIA ex rel. PATRICK MORRISEY, Attorney General, Plaintiff, v. CIVIL ACTION NO. JUDGE PURDUE PHARMA, L.P., a Delaware limited partnership, PURDUE PHARMA INC., a New York corporation, and RICHARD SACKLER, M.D. Defendants. COMPLAINT This action is brought in the name of the State of West Virginia in its sovereign capacity by Patrick Morrisey, Attorney General, pursuant to W. Va. Code § 46A-7-108 of the West Virginia Consumer Credit and Protection Act, W. Va. Code § 46A- 1-101 et seq. ("WVCCPA"), against Purdue Pharma L.P., Purdue Pharma Inc., and Richard Sackler, M.D., to protect consumers and the integrity of the commercial marketplace in West Virginia. This action is brought against Richard Sackler individually and as a director on the Board of Purdue Pharma, Inc. The State also brings suit pursuant to the Attorney General's common law police power to abate and remedy the statewide public nuisance created by the Defendants' interference with the commercial marketplace and endangerment of the public health. BACKGROUND 1. In June 2001, the State of West Virginia, the Bureau of Employment Programs, West Virginia Department of Health and Human Services and the West Virginia Public Employees Insurance Agency sued Purdue Pharma L.P., Purdue Pharma Inc., and Purdue Frederick Company in the Circuit Court of McDowell County, West Virginia, Civil Action No. 01-C-137-S, for violations of the WVCCPA, in connection with the sale and marketing of OxyContin®. The parties entered into a settlement agreement that was approved by Final Order dated December 22, 2004 ("2004 Settlement"). -

Case 1:17-Cv-00450-UNA Document 1 Filed 04/20/17 Page 1 of 18 Pageid #: 1
Case 1:17-cv-00450-UNA Document 1 Filed 04/20/17 Page 1 of 18 PageID #: 1 IN THE UNITED STATES DISTRICT COURT FOR THE DISTRICT OF DELAWARE PURDUE PHARMA L.P., PURDUE ) PHARMACEUTICALS L.P., THE P.F. ) LABORATORIES, INC., and RHODES ) TECHNOLOGIES, ) ) Plaintiffs, ) C.A. No. __________________ v. ) ) ABHAI, LLC and KVK-TECH, INC., ) ) Defendants. ) COMPLAINT Plaintiffs Purdue Pharma L.P., Purdue Pharmaceuticals L.P., The P.F. Laboratories, Inc. (collectively, “Purdue”), and Rhodes Technologies (“Rhodes”) (collectively, “Plaintiffs”), for their Complaint against Defendants Abhai, LLC (“Abhai”) and KVK-TECH, Inc. (“KVK”) (collectively, “Defendants”), aver as follows: NATURE OF THE ACTION 1. This is an action for patent infringement arising under the patent laws of the United States, Title 35, United States Code, for infringement of United States Patent Nos. 9,492,389 (“the ’389 patent”); 9,492,391 (“the ’391 patent”); 9,492,392 (“the ’392 patent”); 9,492,393 (“the ’393 patent”) and 9,522,919 (“the ’919 patent”) (collectively, “the patents-in- suit”). This action relates to Abbreviated New Drug Application (“ANDA”) No. 207493 (“Defendants’ ANDA”) submitted upon information and belief in the name of Defendants to the United States Food and Drug Administration (“FDA”). 2. Plaintiffs seek judgment that Defendants have infringed the ’389, ’391, ’392, ’393, and ’919 patents, which are listed in the FDA Approved Drug Products With Case 1:17-cv-00450-UNA Document 1 Filed 04/20/17 Page 2 of 18 PageID #: 2 Therapeutic Equivalence Evaluations (“Orange Book”) as covering Purdue’s OxyContin® (oxycodone hydrochloride) (“OxyContin®”), an extended-release pain medication. -

Signatory Companies
PhRMA Direct to Consumer Advertising Principles Signatory Companies In October 2018, the PhRMA BoArd of Directors Adopted meAsures to enhAnce the PhRMA Guiding Principles on Direct to Consumer Advertisements About Prescription Medicines. The revised, voluntAry Principles become effective on April 15, 2019. The revised, voluntAry Principles include A new guiding principle stAting thAt “[A]ll DTC television Advertising thAt identifies A prescription medicine by nAme should include direction as to where patients can find information about the cost of the medicine, such as a company- developed website, including the list price And Average, estimAted or typicAl pAtient out-of- pockets costs, or other context About the potentiAl cost of the medicine.” The Principles Also stAte thAt PhRMA will identify on its website All compAnies thAt voluntArily And independently commit to Abide by the Principles And will identify compAnies thAt complete, At the AppropriAte time, AnnuAl certificAtions thAt they hAve policies And procedures in plAce to foster compliAnce with the Principles. The following is A list of All signAtory compAnies who hAve Announced thAt they intend to Abide by the Principles: AbbVie GlAxoSmithKline Alexion Pharmaceuticals, Inc. Incyte CorporAtion Alkermes plc. Ipsen BiophArmAceuticAls, Inc. Allergan plc Johnson & Johnson Amgen Inc. Lundbeck Inc. Astellas Americas Merck & Co., Inc. AstrAZenecA PhArmAceuticAls LP NovArtis PhArmAceuticAls CorporAtion Bayer CorporAtion Novo Nordisk Inc. Biogen OtsukA AmericA PhArmAceuticAl, Inc. (OAPI) BioMArin PhArmAceuticAl Inc. Pfizer Inc Boehringer Ingelheim PharmAceuticAls, Inc. Purdue PhArmA L.P. Bristol-Myers Squibb CompAny Sanofi Celgene CorporAtion Sunovion Pharmaceuticals Inc. Daiichi SAnkyo, Inc. TakedA PhArmAceuticAls USA, Inc. EisAi Inc. TevA PhArmAceuticAls Eli Lilly And CompAny UCB EMD Serono . -

Bio/Pharmaceutical Outsourcing Report: November 2016
Volume 21, Number 11, November 2016 Bio/Pharmaceutical Outsourcing Report Part of PharmSource STRATEGIC ADVANTAGE Business Conditions 1 Business Conditions 1 PE-Owned Companies in Line to Change Hands PE-Owned Companies in Line to Change Hands 2 Commercial Dose Manufacturing and Mergers and acquisitions are a hot topic in the contract services industry these Packaging days, and there is a lot of curiosity about which companies are likely to change 2 New Manufacturing Deal, Temporary hands. Of course, any company is a target at any time, especially when the price is Closures and Recall for Patheon right, but insight to likely acquisition candidates can be gained by looking at the list 3 Commercial Dose Manufacturing and Packaging in Brief 3 Clinical Dose Manufacturing and leastof companies four years owned ago are by likelyprivate targets equity in (PE) the nextfirms. year On oraverage, two. PE firms tend to hold Packaging onto their investments for about five years, so companies that were acquired at 3 Marken to be Acquired by UPS The table on page 2 lists 23 CMC services companies that are PE-owned; the list 5 Clinical Dose Manufacturing and was derived from the PharmSource Strategic Advantage database “Search by Packaging in Brief Ownership” feature. It is sorted by the year each company was acquired, and 6 Side Effects: Impacts of Key Events on suggests that at least 13 CMOs and CDMOs are ripe for a transaction in the foresee- CMOs and CROs able future. 7 API — Large Molecule The sale of one company, Marken (Durham, N.C., USA), was announced just this 7 Samsung BioLogics Completes IPO month (see news item, Page 3); another, Capsugel (Morristown, N.J., USA), has been 8 API — Large Molecule in Brief mentioned in the business press as being prepared for an ownership change. -

Comparison of Oral Contraceptives and Non-Oral Alternatives
PL Detail-Document #290305 −This PL Detail-Document gives subscribers additional insight related to the Recommendations published in− PHARMACIST’S LETTER / PRESCRIBER’S LETTER March 2013 Comparison of Oral Contraceptives and Non-Oral Alternatives —More information about the use of contraceptives is available in our PL Detail-Document, Hormonal Contraception— Productsa Manufacturerb Estrogen Progestin LOW-DOSE MONOPHASIC PILLS Aviane-28 Teva EE 20 mcg Levonorgestrel 0.1 mg Falmina Novast Lessina Teva Lutera Actavis Orsythia Qualitest Sronyx Actavis Gildess Fe 1/20 Qualitest EE 20 mcg Norethindrone acetate Junel 1/20 Teva 1 mg Junel Fe 1/20 Teva Loestrin-21 1/20 Warner Chilcott/Teva Loestrin Fe 1/20 Warner Chilcott/Teva Microgestin 1/20 Actavis Microgestin Fe 1/20 Actavis Generess Fe chewable Actavis EE 25 mcg Norethindrone 0.8 mg Altavera Sandoz EE 30 mcg Levonorgestrel 0.15 mg Kurvelo Lupin Levora Actavis Marlissa Glenmark Nordette-28 Duramed/Teva Portia-28 Teva Cryselle-28 Teva EE 30 mcg Norgestrel 0.3 mg Elinest Novast Low-Ogestrel-21 Actavis Low-Ogestrel-28 Actavis Lo/Ovral-28 Wyeth Gildess Fe 1.5/30 Qualitest EE 30 mcg Norethindrone acetate Junel 1.5/30 Teva 1.5 mg Junel Fe 1.5/30 Teva Loestrin 1.5/30-21 Warner Chilcott/Teva Loestrin Fe 1.5/30 Warner Chilcott/Teva Microgestin 1.5/30 Actavis Microgestin Fe 1.5/30 Actavis More. Copyright © 2013 by Therapeutic Research Center 3120 W. March Lane, Stockton, CA 95219 ~ Phone: 209-472-2240 ~ Fax: 209-472-2249 www.PharmacistsLetter.com ~ www.PrescribersLetter.com ~ www.PharmacyTechniciansLetter.com -

How Purdue Opioid Win Could Bolster J&J in Okla. Trial
How Purdue Opioid Win Could Bolster J&J In Okla. Trial By Cameron Turner Purdue Pharma LP, the manufacturer of OxyContin, currently embroiled in opioid litigation around the country, scored a major victory recently, as District Judge James Hill in Burleigh County, North Dakota, dismissed that state’s action against the opioid defendant. North Dakota’s complaint against Purdue Pharma alleged consumer fraud and public nuisance. Purdue Pharma argued that North Dakota’s claims were preempted by the Federal Food, Drug and Cosmetic Act, and the approval process outlined by the act for Purdue Pharma’s packaging and labeling of its opioid products. Judge Hill agreed, concluding that “the marketing practices of Purdue that the State claims are improper … were consistent with the FDA-approved product labeling.”[1] Cameron Turner The court went on to find that there was “’clear evidence’ that the FDA would not have approved changes to Purdue’s labels to comport with the State’s claims.”[2] As to the consumer fraud claims, the court held that North Dakota was obligated to plead causation that the damages it sought based on the alleged consumer fraud were caused directly by Purdue Pharma’s advertising practices.[3] Instead, the court held that the state’s claims amounted to a “fraud-on-the market” theory that was attenuated at best, given the number of intervening causes and the availability of other lawful and unlawful opioids.[4] Finally, the court refused to extend North Dakota’s public nuisance statute to cases involving the sale of goods under the facts presented by the state.[5] Purdue Pharma’s motion to dismiss was treated as a motion for summary judgment because of the evidence submitted by the parties in their briefs.[6] The state has indicated it will appeal the court’s decision. -

United States House of Representatives House Committee on Oversight and Reform Representative Carolyn B. Maloney, Chair
United States House of Representatives House Committee on Oversight and Reform Representative Carolyn B. Maloney, chair 2157 Rayburn House Office Building Washington, D.C. 20515 December 8, 2020 Dear esteemed committee members, thank you for letting me submit a statement in regards to the criminal Sackler family. I lost my only son, Eddie, on President’s Day 2001 due to an OxyContin he took along with friends at a high school party. August 2001, I attended the very first Congressional hearing on OxyContin and I was shocked when it was revealed that Purdue Pharma had NOT shared the overprescribing data they had on the Pill Mill Doctor who was flooding my neighborhood with OXY’s for the previous two years. Had they of been an ETHICALLY run company and warned of this Pill Mill doctor who later received a 30- year sentence, I would not be living every parent’s worst nightmare and writing you now. I also attended many other hearings/meetings/employee whistle blower trials and gave testimony at the 2007 sentencing in Abingdon, VA. None of these previous hearings, not even the 2007- 600 million plus fine and probation, slowed down the Sackler’s bloodlust for profits. In 2007, a prosecution memo calling for jail time was buried by the brass at the DOJ and the judge never saw this game changing memo. Judge Jones was apologetic to the many families in attendance that he was following the submitted (false) recommendation of NO jail time. The Sackler’s succeeded in gaining immunity for any crimes before the 2007 sentencing but they also pledged in writing things would be different going forward. -
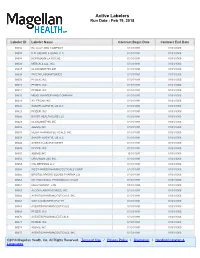
Active Labelers Run Date : Feb 19, 2018
Active Labelers Run Date : Feb 19, 2018 Labeler ID Labeler Name Contract Begin Date Contract End Date 00002 ELI LILLY AND COMPANY 01/01/1991 01/01/3000 00003 E.R. SQUIBB & SONS, LLC. 01/01/1991 01/01/3000 00004 HOFFMANN-LA ROCHE 01/01/1991 01/01/3000 00006 MERCK & CO., INC. 01/01/1991 01/01/3000 00007 GLAXOSMITHKLINE 01/01/1991 01/01/3000 00008 WYETH LABORATORIES 01/01/1991 01/01/3000 00009 PFIZER, INC 01/01/1991 01/01/3000 00013 PFIZER, INC. 01/01/1991 01/01/3000 00014 PFIZER, INC 01/01/1991 01/01/3000 00015 MEAD JOHNSON AND COMPANY 01/01/1991 01/01/3000 00023 ALLERGAN INC 01/01/1991 01/01/3000 00024 SANOFI-AVENTIS, US LLC 01/01/1991 01/01/3000 00025 PFIZER, INC. 01/01/1991 01/01/3000 00026 BAYER HEALTHCARE LLC 01/01/1991 01/01/3000 00029 GLAXOSMITHKLINE 01/01/1991 01/01/3000 00032 ABBVIE INC. 01/01/1991 01/01/3000 00037 MEDA PHARMACEUTICALS, INC. 01/01/1991 01/01/3000 00039 SANOFI-AVENTIS, US LLC 01/01/1991 01/01/3000 00046 AYERST LABORATORIES 01/01/1991 01/01/3000 00049 PFIZER, INC 01/01/1991 01/01/3000 00051 ABBVIE INC 10/01/1997 01/01/3000 00052 ORGANON USA INC. 01/01/1991 01/01/3000 00053 CSL BEHRING LLC 01/01/1991 01/01/3000 00054 WEST-WARD PHARMACEUTICALS CORP. 01/01/1991 01/01/3000 00056 BRISTOL-MYERS SQUIBB PHARMA CO. 01/01/1991 01/01/3000 00062 ORTHO MCNEIL PHARMACEUTICALS 01/01/1991 01/01/3000 00064 HEALTHPOINT, LTD. -

Weekly Capitol Hill Report October 23, 2020
Issues for the week ending October 23, 2020 In this Issue: Federal Issues Legislative Federal Issues Legislative President Signs National Suicide Hotline Bill • President Signs National Suicide Hotline Bill Into Law Into Law On October 17, President Trump signed into law the National Suicide Hotline Designation Act, Regulatory designating 988 as the nationwide phone number to • CMS to Delay Start Date for Radiation connect people experiencing mental health crises to Oncology Model the National Suicide Prevention Lifeline. • HHS Again Updates PRF Reporting Requirements-Restores Flexibility on “Lost The Lifeline is a network of 163 crisis centers that last Revenue” year answered more than 2.2 million calls and 100,000 • Expanded Eligibility Phase 3 General online chats. The Federal Communications Distribution – Applications Due November 6, Commission in July designated the number for this 2020 purpose, meaning telecommunications carriers and • Federal COVID-19 Policy Guidance and Other one-way Voice over Internet Protocol service providers Developments must direct 988 calls to 1-800-273-TALK (8255) by July 16, 2022. State Issues Under the bill, the Department of Health and Human Pennsylvania Services must also develop a strategy to provide Legislative access to competent, specialized services for high-risk • Senate and House Finalize Health Care Bills populations such as LGBTQ+ youth, minorities, and • Legislation Regulating PBM/Medicaid MCO individuals in rural communities. Arrangements Clears Senate • House Endorses CRNP Independent Practice Why this matters: This pandemic has highlighted the Pilot for Rural Areas need for modernized mental health and suicide West Virginia prevention resources, and this easy-to-remember Regulatory number will help connect callers in need of help with • COVID-19 Vaccine Plans Not on Agenda, experts who can offer resources and hope to make it West Virginia Health Officer Says through a mental health crisis.