Characterization of the Chromatin Accessibility in an Alzheimer's
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

CD29 Identifies IFN-Γ–Producing Human CD8+ T Cells With
+ CD29 identifies IFN-γ–producing human CD8 T cells with an increased cytotoxic potential Benoît P. Nicoleta,b, Aurélie Guislaina,b, Floris P. J. van Alphenc, Raquel Gomez-Eerlandd, Ton N. M. Schumacherd, Maartje van den Biggelaarc,e, and Monika C. Wolkersa,b,1 aDepartment of Hematopoiesis, Sanquin Research, 1066 CX Amsterdam, The Netherlands; bLandsteiner Laboratory, Oncode Institute, Amsterdam University Medical Center, University of Amsterdam, 1105 AZ Amsterdam, The Netherlands; cDepartment of Research Facilities, Sanquin Research, 1066 CX Amsterdam, The Netherlands; dDivision of Molecular Oncology and Immunology, Oncode Institute, The Netherlands Cancer Institute, 1066 CX Amsterdam, The Netherlands; and eDepartment of Molecular and Cellular Haemostasis, Sanquin Research, 1066 CX Amsterdam, The Netherlands Edited by Anjana Rao, La Jolla Institute for Allergy and Immunology, La Jolla, CA, and approved February 12, 2020 (received for review August 12, 2019) Cytotoxic CD8+ T cells can effectively kill target cells by producing therefore developed a protocol that allowed for efficient iso- cytokines, chemokines, and granzymes. Expression of these effector lation of RNA and protein from fluorescence-activated cell molecules is however highly divergent, and tools that identify and sorting (FACS)-sorted fixed T cells after intracellular cytokine + preselect CD8 T cells with a cytotoxic expression profile are lacking. staining. With this top-down approach, we performed an un- + Human CD8 T cells can be divided into IFN-γ– and IL-2–producing biased RNA-sequencing (RNA-seq) and mass spectrometry cells. Unbiased transcriptomics and proteomics analysis on cytokine- γ– – + + (MS) analyses on IFN- and IL-2 producing primary human producing fixed CD8 T cells revealed that IL-2 cells produce helper + + + CD8 Tcells. -

Role of RUNX1 in Aberrant Retinal Angiogenesis Jonathan D
Page 1 of 25 Diabetes Identification of RUNX1 as a mediator of aberrant retinal angiogenesis Short Title: Role of RUNX1 in aberrant retinal angiogenesis Jonathan D. Lam,†1 Daniel J. Oh,†1 Lindsay L. Wong,1 Dhanesh Amarnani,1 Cindy Park- Windhol,1 Angie V. Sanchez,1 Jonathan Cardona-Velez,1,2 Declan McGuone,3 Anat O. Stemmer- Rachamimov,3 Dean Eliott,4 Diane R. Bielenberg,5 Tave van Zyl,4 Lishuang Shen,1 Xiaowu Gai,6 Patricia A. D’Amore*,1,7 Leo A. Kim*,1,4 Joseph F. Arboleda-Velasquez*1 Author affiliations: 1Schepens Eye Research Institute/Massachusetts Eye and Ear, Department of Ophthalmology, Harvard Medical School, 20 Staniford St., Boston, MA 02114 2Universidad Pontificia Bolivariana, Medellin, Colombia, #68- a, Cq. 1 #68305, Medellín, Antioquia, Colombia 3C.S. Kubik Laboratory for Neuropathology, Massachusetts General Hospital, 55 Fruit St., Boston, MA 02114 4Retina Service, Massachusetts Eye and Ear Infirmary, Department of Ophthalmology, Harvard Medical School, 243 Charles St., Boston, MA 02114 5Vascular Biology Program, Boston Children’s Hospital, Department of Surgery, Harvard Medical School, 300 Longwood Ave., Boston, MA 02115 6Center for Personalized Medicine, Children’s Hospital Los Angeles, Los Angeles, 4650 Sunset Blvd, Los Angeles, CA 90027, USA 7Department of Pathology, Harvard Medical School, 25 Shattuck St., Boston, MA 02115 Corresponding authors: Joseph F. Arboleda-Velasquez: [email protected] Ph: (617) 912-2517 Leo Kim: [email protected] Ph: (617) 912-2562 Patricia D’Amore: [email protected] Ph: (617) 912-2559 Fax: (617) 912-0128 20 Staniford St. Boston MA, 02114 † These authors contributed equally to this manuscript Word Count: 1905 Tables and Figures: 4 Diabetes Publish Ahead of Print, published online April 11, 2017 Diabetes Page 2 of 25 Abstract Proliferative diabetic retinopathy (PDR) is a common cause of blindness in the developed world’s working adult population, and affects those with type 1 and type 2 diabetes mellitus. -

1A Multiple Sclerosis Treatment
The Pharmacogenomics Journal (2012) 12, 134–146 & 2012 Macmillan Publishers Limited. All rights reserved 1470-269X/12 www.nature.com/tpj ORIGINAL ARTICLE Network analysis of transcriptional regulation in response to intramuscular interferon-b-1a multiple sclerosis treatment M Hecker1,2, RH Goertsches2,3, Interferon-b (IFN-b) is one of the major drugs for multiple sclerosis (MS) 3 2 treatment. The purpose of this study was to characterize the transcriptional C Fatum , D Koczan , effects induced by intramuscular IFN-b-1a therapy in patients with relapsing– 2 1 H-J Thiesen , R Guthke remitting form of MS. By using Affymetrix DNA microarrays, we obtained and UK Zettl3 genome-wide expression profiles of peripheral blood mononuclear cells of 24 MS patients within the first 4 weeks of IFN-b administration. We identified 1Leibniz Institute for Natural Product Research 121 genes that were significantly up- or downregulated compared with and Infection Biology—Hans-Knoell-Institute, baseline, with stronger changed expression at 1 week after start of therapy. Jena, Germany; 2University of Rostock, Institute of Immunology, Rostock, Germany and Eleven transcription factor-binding sites (TFBS) are overrepresented in the 3University of Rostock, Department of Neurology, regulatory regions of these genes, including those of IFN regulatory factors Rostock, Germany and NF-kB. We then applied TFBS-integrating least angle regression, a novel integrative algorithm for deriving gene regulatory networks from gene Correspondence: M Hecker, Leibniz Institute for Natural Product expression data and TFBS information, to reconstruct the underlying network Research and Infection Biology—Hans-Knoell- of molecular interactions. An NF-kB-centered sub-network of genes was Institute, Beutenbergstr. -

Functional Receptor Molecules Cd300lf and Cd300ld Within the CD300 Family Enable Murine Noroviruses to Infect Cells
Functional receptor molecules CD300lf and CD300ld within the CD300 family enable murine noroviruses to infect cells Kei Hagaa,1, Akira Fujimotoa,1, Reiko Takai-Todakaa, Motohiro Mikia,b, Yen Hai Doana, Kosuke Murakamia, Masaru Yokoyamac, Kazuyoshi Muratad, Akira Nakanishie,2, and Kazuhiko Katayamaa,2 aDepartment of Virology II, National Institute of Infectious Diseases, Tokyo 208-0011, Japan; bNew Business Planning Division, Innovative Diagnostics Research Center, Denka Innovation Center, Denka Company Limited, Tokyo 194-8560, Japan; cPathogen Genomics Center, National Institute of Infectious Diseases, Tokyo 208-0011, Japan; dNational Institute for Physiological Sciences, Aichi 444-8585, Japan; and eSection of Gene Therapy, Department of Aging Intervention, National Center for Geriatrics and Gerontology, Aichi 474-8511, Japan Edited by Mary K. Estes, Baylor College of Medicine, Houston, Texas, and approved August 11, 2016 (received for review April 9, 2016) Norovirus is the leading cause of acute gastroenteritis worldwide. mice that were deficient in STAT1 and RAG2 (11). MNV repli- Since the discovery of human norovirus (HuNoV), an efficient and cates within the murine macrophage cell line RAW264.7 cells. reproducible norovirus replication system has not been established in MNV strains MNV-1, WU11, and S99 bind macrophages ex vivo cultured cells. Although limited amounts of virus particles can be through a terminal sialic acid on the ganglioside GD1a, which produced when the HuNoV genome is directly transfected into cells, has been suggested as a binding receptor (12). Another MNV the HuNoV cycle of infection has not been successfully reproduced in strain, CR3, binds a different glycan, and the difference in glycan any currently available cell-culture system. -

Functional Receptor Molecules Cd300lf and Cd300ld Within the CD300 Family Enable Murine Noroviruses to Infect Cells
Functional receptor molecules CD300lf and CD300ld within the CD300 family enable murine noroviruses to infect cells Kei Hagaa,1, Akira Fujimotoa,1, Reiko Takai-Todakaa, Motohiro Mikia,b, Yen Hai Doana, Kosuke Murakamia, Masaru Yokoyamac, Kazuyoshi Muratad, Akira Nakanishie,2, and Kazuhiko Katayamaa,2 aDepartment of Virology II, National Institute of Infectious Diseases, Tokyo 208-0011, Japan; bNew Business Planning Division, Innovative Diagnostics Research Center, Denka Innovation Center, Denka Company Limited, Tokyo 194-8560, Japan; cPathogen Genomics Center, National Institute of Infectious Diseases, Tokyo 208-0011, Japan; dNational Institute for Physiological Sciences, Aichi 444-8585, Japan; and eSection of Gene Therapy, Department of Aging Intervention, National Center for Geriatrics and Gerontology, Aichi 474-8511, Japan Edited by Mary K. Estes, Baylor College of Medicine, Houston, Texas, and approved August 11, 2016 (received for review April 9, 2016) Norovirus is the leading cause of acute gastroenteritis worldwide. mice that were deficient in STAT1 and RAG2 (11). MNV repli- Since the discovery of human norovirus (HuNoV), an efficient and cates within the murine macrophage cell line RAW264.7 cells. reproducible norovirus replication system has not been established in MNV strains MNV-1, WU11, and S99 bind macrophages ex vivo cultured cells. Although limited amounts of virus particles can be through a terminal sialic acid on the ganglioside GD1a, which produced when the HuNoV genome is directly transfected into cells, has been suggested as a binding receptor (12). Another MNV the HuNoV cycle of infection has not been successfully reproduced in strain, CR3, binds a different glycan, and the difference in glycan any currently available cell-culture system. -
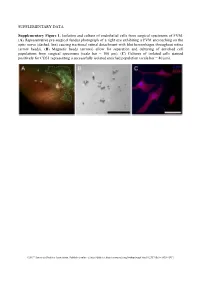
Supplementary Figures and Tables
SUPPLEMENTARY DATA Supplementary Figure 1. Isolation and culture of endothelial cells from surgical specimens of FVM. (A) Representative pre-surgical fundus photograph of a right eye exhibiting a FVM encroaching on the optic nerve (dashed line) causing tractional retinal detachment with blot hemorrhages throughout retina (arrow heads). (B) Magnetic beads (arrows) allow for separation and culturing of enriched cell populations from surgical specimens (scale bar = 100 μm). (C) Cultures of isolated cells stained positively for CD31 representing a successfully isolated enriched population (scale bar = 40 μm). ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Figure 2. Efficient siRNA knockdown of RUNX1 expression and function demonstrated by qRT-PCR, Western Blot, and scratch assay. (A) RUNX1 siRNA induced a 60% reduction of RUNX1 expression measured by qRT-PCR 48 hrs post-transfection whereas expression of RUNX2 and RUNX3, the two other mammalian RUNX orthologues, showed no significant changes, indicating specificity of our siRNA. Functional inhibition of Runx1 signaling was demonstrated by a 330% increase in insulin-like growth factor binding protein-3 (IGFBP3) RNA expression level, a known target of RUNX1 inhibition. Western blot demonstrated similar reduction in protein levels. (B) siRNA- 2’s effect on RUNX1 was validated by qRT-PCR and western blot, demonstrating a similar reduction in both RNA and protein. Scratch assay demonstrates functional inhibition of RUNX1 by siRNA-2. ns: not significant, * p < 0.05, *** p < 0.001 ©2017 American Diabetes Association. Published online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db16-1035/-/DC1 SUPPLEMENTARY DATA Supplementary Table 1. -

Brain Region-Dependent Gene Networks Associated with Selective Breeding for Increased Voluntary Wheel-Running Behavior
UC Riverside UC Riverside Previously Published Works Title Brain region-dependent gene networks associated with selective breeding for increased voluntary wheel-running behavior. Permalink https://escholarship.org/uc/item/8c49g8fd Journal PloS one, 13(8) ISSN 1932-6203 Authors Zhang, Pan Rhodes, Justin S Garland, Theodore et al. Publication Date 2018 DOI 10.1371/journal.pone.0201773 Peer reviewed eScholarship.org Powered by the California Digital Library University of California RESEARCH ARTICLE Brain region-dependent gene networks associated with selective breeding for increased voluntary wheel-running behavior Pan Zhang1,2, Justin S. Rhodes3,4, Theodore Garland, Jr.5, Sam D. Perez3, Bruce R. Southey2, Sandra L. Rodriguez-Zas2,6,7* 1 Illinois Informatics Institute, University of Illinois at Urbana-Champaign, Urbana, IL, United States of America, 2 Department of Animal Sciences, University of Illinois at Urbana-Champaign, Urbana, IL, United a1111111111 States of America, 3 Beckman Institute for Advanced Science and Technology, Urbana, IL, United States of a1111111111 America, 4 Center for Nutrition, Learning and Memory, University of Illinois at Urbana-Champaign, Urbana, a1111111111 IL, United States of America, 5 Department of Evolution, Ecology, and Organismal Biology, University of a1111111111 California, Riverside, CA, United States of America, 6 Department of Statistics, University of Illinois at Urbana-Champaign, Urbana, IL, United States of America, 7 Carle Woese Institute for Genomic Biology, a1111111111 University of Illinois at Urbana-Champaign, Urbana, IL, United States of America * [email protected] OPEN ACCESS Abstract Citation: Zhang P, Rhodes JS, Garland T, Jr., Perez SD, Southey BR, Rodriguez-Zas SL (2018) Brain Mouse lines selectively bred for high voluntary wheel-running behavior are helpful models region-dependent gene networks associated with for uncovering gene networks associated with increased motivation for physical activity and selective breeding for increased voluntary wheel- other reward-dependent behaviors. -

Identi Cation of Potential Biomarkers and Pathways
Identication of Potential Biomarkers and Pathways in Neonatal Hypoxic-Ischemic Brain Injury: Based on Bioinformatics Technology Shangbin Li The First Hospital of Hebei Medical University Shuangshuang Li Chengdu University of Traditional Chinese Medicine Qian Zhao The First Hospital of Hebei Medical University Jiayu Huang The First Hospital of Hebei Medical University Jinfeng Meng The First Hospital of Hebei Medical University Weichen Yan The First Hospital of Hebei Medical University Jie Wang The First Hospital of Hebei Medical University Changjun Ren ( [email protected] ) The First Hospital of Hebei Medical University Ling Hao The First Hospital of Hebei Medical University Research Article Keywords: neonatal hypoxic-ischemic brain injury, inammatory response, immune response, differentially expressed genes, bioinformatics analysis Posted Date: August 5th, 2021 DOI: https://doi.org/10.21203/rs.3.rs-777916/v1 License: This work is licensed under a Creative Commons Attribution 4.0 International License. Read Full License Page 1/22 Abstract Background Neonatal hypoxic-ischemic brain damage (HIBD) is one of the most common serious diseases in newborns, with a high mortality and disability rate. This study aims to use the bioinformatics analysis to identify potential hematologic/immune systems tissue-specic genes and related signaling pathways neonatal HIBD. Methods Microarray datasets in HIBD were downloaded from the Gene Expression Omnibus database, and DEGs were identied by R software.Enrichment analyses were performed and protein–protein interaction networks were constructed to understand the functions and enriched pathways of DEGs and to identify central genes and key modules. Results In the cerebral cortex tissue with HIBD, 2598 DEGs were identied, including 2362 up-regulated and 236 down-regulated DEGs. -

A Non-Synonymous SNP Within Membrane Metalloendopeptidase-Like 1 (MMEL1) Is Associated with Multiple Sclerosis
Genes and Immunity (2010) 11, 660–664 & 2010 Macmillan Publishers Limited All rights reserved 1466-4879/10 www.nature.com/gene ORIGINAL ARTICLE A non-synonymous SNP within membrane metalloendopeptidase-like 1 (MMEL1) is associated with multiple sclerosis M Ban1,15, JL McCauley2,15, R Zuvich3,15, A Baker1, L Bergamaschi4, M Cox5, A Kemppinen1, S D’Alfonso4, FR Guerini6, J Lechner-Scott5, F Dudbridge7, J Wason7, NP Robertson8, PL De Jager9,10, DA Hafler11, LF Barcellos12, AJ Ivinson13, D Sexton3, JR Oksenberg14, SL Hauser14, MA Pericak-Vance2, J Haines3, A Compston1 and S Sawcer1 1Department of Clinical Neuroscience, University of Cambridge, Addenbrooke’s Hospital, Cambridge, UK; 2Miami Institute for Human Genomics, University of Miami Miller School of Medicine, Miami, FL, USA; 3Center for Human Genetics Research, Vanderbilt University Medical Center, Nashville, TN, USA; 4Department of Medical Sciences and Interdisciplinary Research Center of Autoimmune Diseases, University of Eastern Piedmont, Novara, Italy; 5University of Newcastle, John Hunter Hospital, Newcastle, New South Wales, Australia; 6Laboratory of Molecular Medicine and Biotechnology, Don C Gnocchi Foundation IRCCS, S Maria Nascente, Milan, Italy; 7MRC Biostatistics Unit, Cambridge, UK; 8Department of Neurology, University Hospital of Wales, Heath Park, Cardiff, UK; 9Program in Translational NeuroPsychiatric Genomics, Center for Neurologic Diseases, Brigham and Women’s Hospital, Harvard Medical School, Boston, MA, USA; 10Broad Institute, Cambridge, MA, USA; 11Department of Neurology, Yale University School of Medicine, New Haven, CT, USA; 12Division of Epidemiology, School of Public Health, University of California at Berkeley, Berkeley, CA, USA; 13Harvard NeuroDiscovery Center, Harvard Medical School, Boston, MA, USA and 14Department of Neurology, University of California San Francisco, San Francisco, CA, USA Several single-nucleotide polymorphism (SNP) genome-wide association studies (GWASs) have been completed in multiple sclerosis (MS). -

Nº Ref Uniprot Proteína Péptidos Identificados Por MS/MS 1 P01024
Document downloaded from http://www.elsevier.es, day 26/09/2021. This copy is for personal use. Any transmission of this document by any media or format is strictly prohibited. Nº Ref Uniprot Proteína Péptidos identificados 1 P01024 CO3_HUMAN Complement C3 OS=Homo sapiens GN=C3 PE=1 SV=2 por 162MS/MS 2 P02751 FINC_HUMAN Fibronectin OS=Homo sapiens GN=FN1 PE=1 SV=4 131 3 P01023 A2MG_HUMAN Alpha-2-macroglobulin OS=Homo sapiens GN=A2M PE=1 SV=3 128 4 P0C0L4 CO4A_HUMAN Complement C4-A OS=Homo sapiens GN=C4A PE=1 SV=1 95 5 P04275 VWF_HUMAN von Willebrand factor OS=Homo sapiens GN=VWF PE=1 SV=4 81 6 P02675 FIBB_HUMAN Fibrinogen beta chain OS=Homo sapiens GN=FGB PE=1 SV=2 78 7 P01031 CO5_HUMAN Complement C5 OS=Homo sapiens GN=C5 PE=1 SV=4 66 8 P02768 ALBU_HUMAN Serum albumin OS=Homo sapiens GN=ALB PE=1 SV=2 66 9 P00450 CERU_HUMAN Ceruloplasmin OS=Homo sapiens GN=CP PE=1 SV=1 64 10 P02671 FIBA_HUMAN Fibrinogen alpha chain OS=Homo sapiens GN=FGA PE=1 SV=2 58 11 P08603 CFAH_HUMAN Complement factor H OS=Homo sapiens GN=CFH PE=1 SV=4 56 12 P02787 TRFE_HUMAN Serotransferrin OS=Homo sapiens GN=TF PE=1 SV=3 54 13 P00747 PLMN_HUMAN Plasminogen OS=Homo sapiens GN=PLG PE=1 SV=2 48 14 P02679 FIBG_HUMAN Fibrinogen gamma chain OS=Homo sapiens GN=FGG PE=1 SV=3 47 15 P01871 IGHM_HUMAN Ig mu chain C region OS=Homo sapiens GN=IGHM PE=1 SV=3 41 16 P04003 C4BPA_HUMAN C4b-binding protein alpha chain OS=Homo sapiens GN=C4BPA PE=1 SV=2 37 17 Q9Y6R7 FCGBP_HUMAN IgGFc-binding protein OS=Homo sapiens GN=FCGBP PE=1 SV=3 30 18 O43866 CD5L_HUMAN CD5 antigen-like OS=Homo -

The DNA Sequence and Comparative Analysis of Human Chromosome 20
articles The DNA sequence and comparative analysis of human chromosome 20 P. Deloukas, L. H. Matthews, J. Ashurst, J. Burton, J. G. R. Gilbert, M. Jones, G. Stavrides, J. P. Almeida, A. K. Babbage, C. L. Bagguley, J. Bailey, K. F. Barlow, K. N. Bates, L. M. Beard, D. M. Beare, O. P. Beasley, C. P. Bird, S. E. Blakey, A. M. Bridgeman, A. J. Brown, D. Buck, W. Burrill, A. P. Butler, C. Carder, N. P. Carter, J. C. Chapman, M. Clamp, G. Clark, L. N. Clark, S. Y. Clark, C. M. Clee, S. Clegg, V. E. Cobley, R. E. Collier, R. Connor, N. R. Corby, A. Coulson, G. J. Coville, R. Deadman, P. Dhami, M. Dunn, A. G. Ellington, J. A. Frankland, A. Fraser, L. French, P. Garner, D. V. Grafham, C. Grif®ths, M. N. D. Grif®ths, R. Gwilliam, R. E. Hall, S. Hammond, J. L. Harley, P. D. Heath, S. Ho, J. L. Holden, P. J. Howden, E. Huckle, A. R. Hunt, S. E. Hunt, K. Jekosch, C. M. Johnson, D. Johnson, M. P. Kay, A. M. Kimberley, A. King, A. Knights, G. K. Laird, S. Lawlor, M. H. Lehvaslaiho, M. Leversha, C. Lloyd, D. M. Lloyd, J. D. Lovell, V. L. Marsh, S. L. Martin, L. J. McConnachie, K. McLay, A. A. McMurray, S. Milne, D. Mistry, M. J. F. Moore, J. C. Mullikin, T. Nickerson, K. Oliver, A. Parker, R. Patel, T. A. V. Pearce, A. I. Peck, B. J. C. T. Phillimore, S. R. Prathalingam, R. W. Plumb, H. Ramsay, C. M. -

Transcriptome Analysis on Monocytes from Patients With
www.nature.com/scientificreports OPEN Transcriptome Analysis on Monocytes from Patients with Neovascular Age-Related Macular Received: 26 February 2016 Accepted: 10 June 2016 Degeneration Published: 04 July 2016 Michelle Grunin1, Shira-Hagbi-Levi1, Batya Rinsky1, Yoav Smith2 & Itay Chowers1 Mononuclear phagocytes (MPs), including monocytes/macrophages, play complex roles in age- related macular degeneration (AMD) pathogenesis. We reported altered gene-expression signature in peripheral blood mononuclear cells from AMD patients, and a chemokine receptor signature on AMD monocytes. To obtain comprehensive understanding of MP involvement, particularly in peripheral circulation in AMD, we performed global gene expression analysis in monocytes. We separated monocytes from treatment-naïve neovascular AMD (nvAMD) patients (n = 14) and age-matched controls (n = 15), and performed microarray and bioinformatics analysis. Quantitative real-time PCR was performed on other sets of nvAMD (n = 25), atrophic AMD (n = 21), and controls (n = 28) for validation. This validated microarray genes (like TMEM176A/B and FOSB) tested, including differences between nvAMD and atrophic AMD. We identified 2,165 differentially-expressed genes (P < 0.05), including 79 genes with log2 fold change ≥1.5 between nvAMD and controls. Functional annotation using DAVID and TANGO demonstrated immune response alterations in AMD monocytes (FDR-P <0.05), validated by randomized data comparison (P < 0.0001). GSEA, ISMARA, and MEME analysis found immune enrichment and specific involved microRNAs. Enrichment of differentially-expressed genes in monocytes was found in retina via SAGE data-mining. These genes were enriched in non-classical vs. classical monocyte subsets (P < 0.05). Therefore, global gene expression analysis in AMD monocytes reveals an altered immune-related signature, further implicating systemic MP activation in AMD.