Hydroboration-Oxidation of Styrene, 2,3 Dihydrofuran and Quadricyclene Dimethylester Promoted by Wilkinson's Catalyst Pingyun Feng
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Organoboranes in Organic Syntheses Including Suzuki Coupling Reaction
HETEROCYCLES, Vol. 80, No. 1, 2010 15 HETEROCYCLES, Vol. 80, No. 1, 2010, pp. 15 - 43. © The Japan Institute of Heterocyclic Chemistry DOI: 10.3987/COM-09-S(S)Summary ORGANOBORANES IN ORGANIC SYNTHESES INCLUDING SUZUKI COUPLING REACTION Akira Suzuki In 1962 I had a lively interest in Wacker reaction [the oxidation of ethylene to acetaldehyde in the presence of palladium chloride and cupric chloride (Angew. Chem. 1959, 71, 176)] and began a literature survey. One Saturday afternoon during that time, I went a bookstore in Sapporo to look at new chemistry books and found a red and black two-tone colored book on the shelf that did not look like a chemistry book. The book was "Hydroboration" written by Professor Herbert C. Brown of Purdue University. It seemed to be an interesting book, so, I bought it. This book changed the course of my career, and my fascination with the chemistry of hydroboration reaction and organoboron compounds thus prepared by hydroboration began after reading the book. I immediately wrote to Professor Brown requesting to work as a postdoctoral research fellow. At that time Professor Brown was at Heidelberg in Germany as a visiting professor. He kindly wrote me a letter of acceptance, and I began a study of the stereochemistry of hydroboration reaction at Purdue (1963-65). Through this work I came to understand hydroboration and the interesting characteristics of organoboranes. My family (wife and two small girls) and I had a very good time there and made good friends. Of course I enjoyed chemistry. After a stay of about two years at Purdue, I returned to Japan with my family at the end of March 1965. -

Syntheses and Eliminations of Cyclopentyl Derivatives David John Rausch Iowa State University
Iowa State University Capstones, Theses and Retrospective Theses and Dissertations Dissertations 1966 Syntheses and eliminations of cyclopentyl derivatives David John Rausch Iowa State University Follow this and additional works at: https://lib.dr.iastate.edu/rtd Part of the Organic Chemistry Commons Recommended Citation Rausch, David John, "Syntheses and eliminations of cyclopentyl derivatives " (1966). Retrospective Theses and Dissertations. 2875. https://lib.dr.iastate.edu/rtd/2875 This Dissertation is brought to you for free and open access by the Iowa State University Capstones, Theses and Dissertations at Iowa State University Digital Repository. It has been accepted for inclusion in Retrospective Theses and Dissertations by an authorized administrator of Iowa State University Digital Repository. For more information, please contact [email protected]. This dissertation has been microfilmed exactly as received 66—6996 RAUSCH, David John, 1940- SYNTHESES AND ELIMINATIONS OF CYCLOPENTYL DERIVATIVES. Iowa State University of Science and Technology Ph.D., 1966 Chemistry, organic University Microfilms, Inc., Ann Arbor, Michigan SYNTHESES AND ELIMINATIONS OF CYCLOPENTYL DERIVATIVES by David John Rausch A Dissertation Submitted to the Graduate Faculty in Partial Fulfillment of The Requirements for the Degree of DOCTOR OF PHILOSOPHY Major Subject: Organic Chemistry Approved : Signature was redacted for privacy. Signature was redacted for privacy. Head of Major Department Signature was redacted for privacy. Iowa State University Of Science and Technology Ames, Iowa 1966 ii TABLE OF CONTENTS VITA INTRODUCTION HISTORICAL Conformation of Cyclopentanes Elimination Reactions RESULTS AND DISCUSSION Synthetic Elimination Reactions EXPERIMENTAL Preparation and Purification of Materials Procedures and Data for Beta Elimination Reactions SUMMARY LITERATURE CITED ACKNOWLEDGEMENTS iii VITA The author was born in Aurora, Illinois, on October 24, 1940, to Mr. -

Chemistry 301-301A - Hour Examination #3, December 11, 2003
Chemistry 301-301A - Hour Examination #3, December 11, 2003 “.....as we know, there are known unknowns; there are things we know we know. We also know there are known unknowns; that is to say we know there are some things we do not know. But there are also unknown unknowns - the ones we don't know we don't know.” Donald Rumsfeld (winner of a British award given to the worst mangler of the English language in 2003) “I know, a proof is a proof. What kind of a proof is a proof? A proof is a proof and when you have a good proof it's because it's proven." Jean Chrétien (hon. mention for the same award) 1[18 points] (a) Acid-catalyzed addition of water to 3-methyl-1-butene (1) results in formation of large amounts of a rearranged alcohol (2), in addition to the expected alcohol (3). Explain, with excellent arrow formalisms. H2O + H O+ 3 OH OH 1 3 2 (b) On the other hand hydroboration of 1, followed by oxidation, does not lead to any rearranged product. Only alcohol 4 is formed. Explain. Detailed mechanisms are not needed here, but a drawing of the transition state for the hydroboration step is. 1. BH3 OH 2. HOOH/HO – 1 4 (c) But there are some strange things that happen in hydroboration. For example when 2-methyl-2-butene (5) is hydroborated at high temperature, then treated with HOOH/HO–, alcohol 4 is still one of the products. Explain mechanistcally. Hint: at high temperature hydroboration is reversible. -

Reactions of Alkenes and Alkynes
05 Reactions of Alkenes and Alkynes Polyethylene is the most widely used plastic, making up items such as packing foam, plastic bottles, and plastic utensils (top: © Jon Larson/iStockphoto; middle: GNL Media/Digital Vision/Getty Images, Inc.; bottom: © Lakhesis/iStockphoto). Inset: A model of ethylene. KEY QUESTIONS 5.1 What Are the Characteristic Reactions of Alkenes? 5.8 How Can Alkynes Be Reduced to Alkenes and 5.2 What Is a Reaction Mechanism? Alkanes? 5.3 What Are the Mechanisms of Electrophilic Additions HOW TO to Alkenes? 5.1 How to Draw Mechanisms 5.4 What Are Carbocation Rearrangements? 5.5 What Is Hydroboration–Oxidation of an Alkene? CHEMICAL CONNECTIONS 5.6 How Can an Alkene Be Reduced to an Alkane? 5A Catalytic Cracking and the Importance of Alkenes 5.7 How Can an Acetylide Anion Be Used to Create a New Carbon–Carbon Bond? IN THIS CHAPTER, we begin our systematic study of organic reactions and their mecha- nisms. Reaction mechanisms are step-by-step descriptions of how reactions proceed and are one of the most important unifying concepts in organic chemistry. We use the reactions of alkenes as the vehicle to introduce this concept. 129 130 CHAPTER 5 Reactions of Alkenes and Alkynes 5.1 What Are the Characteristic Reactions of Alkenes? The most characteristic reaction of alkenes is addition to the carbon–carbon double bond in such a way that the pi bond is broken and, in its place, sigma bonds are formed to two new atoms or groups of atoms. Several examples of reactions at the carbon–carbon double bond are shown in Table 5.1, along with the descriptive name(s) associated with each. -

Stapled Peptides—A Useful Improvement for Peptide-Based Drugs
molecules Review Stapled Peptides—A Useful Improvement for Peptide-Based Drugs Mattia Moiola, Misal G. Memeo and Paolo Quadrelli * Department of Chemistry, University of Pavia, Viale Taramelli 12, 27100 Pavia, Italy; [email protected] (M.M.); [email protected] (M.G.M.) * Correspondence: [email protected]; Tel.: +39-0382-987315 Received: 30 July 2019; Accepted: 1 October 2019; Published: 10 October 2019 Abstract: Peptide-based drugs, despite being relegated as niche pharmaceuticals for years, are now capturing more and more attention from the scientific community. The main problem for these kinds of pharmacological compounds was the low degree of cellular uptake, which relegates the application of peptide-drugs to extracellular targets. In recent years, many new techniques have been developed in order to bypass the intrinsic problem of this kind of pharmaceuticals. One of these features is the use of stapled peptides. Stapled peptides consist of peptide chains that bring an external brace that force the peptide structure into an a-helical one. The cross-link is obtained by the linkage of the side chains of opportune-modified amino acids posed at the right distance inside the peptide chain. In this account, we report the main stapling methodologies currently employed or under development and the synthetic pathways involved in the amino acid modifications. Moreover, we report the results of two comparative studies upon different kinds of stapled-peptides, evaluating the properties given from each typology of staple to the target peptide and discussing the best choices for the use of this feature in peptide-drug synthesis. Keywords: stapled peptide; structurally constrained peptide; cellular uptake; helicity; peptide drugs 1. -

Hydroboration-Oxidation Of
molecules Article Hydroboration-Oxidation of (±)-(1α,3α,3aβ,6aβ) -1,2,3,3a,4,6a-Hexahydro-1,3-pentalenedimethanol and Its O-Protected Derivatives: Synthesis of New Compounds Useful for Obtaining (iso)Carbacyclin Analogues and X-ray Analysis of the Products Constantin I. Tănase 1,* ID , Florea G. Cocu 1, Miron Teodor Căproiu 2, Constantin Drăghici 2 and Sergiu Shova 3 1 National Institute for Chemical-Pharmaceutical Research and Development, Department of bioactive substances and pharmaceutical technologies, 112 Vitan Av., 031299, Bucharest-3, Romania; [email protected] 2 Organic Chemistry Center “C.D.Nenitescu”, Spectroscopy Laboratory, 202 B Splaiul Independentei, 060023 Bucharest, Romania; [email protected] (M.T.C.); [email protected] (C.D.) 3 Institute of Macromolecular Chemistry “Petru Poni”, Crystallography Department, 700478 Iasi, Romania; [email protected] * Correspondence: [email protected]; Tel.: +40-21-321-2117; Fax: +40-21-322-2917 Received: 22 October 2017; Accepted: 16 November 2017; Published: 24 November 2017 Abstract: Hydroboration-oxidation of 2α,4α-dimethanol-1β,5β-bicyclo[3.3.0]oct-6-en dibenzoate (1) gave alcohols 2 (symmetric) and 3 (unsymmetric) in ~60% yield, together with the monobenzoate diol 4a (37%), resulting from the reduction of the closer benzoate by the intermediate alkylborane. The corresponding alkene and dialdehyde gave only the triols 8 and 9 in ~1:1 ratio. By increasing the reaction time and the temperature, the isomerization of alkylboranes favours the un-symmetrical triol 9. The PDC oxidation of the alcohols gave cleanly the corresponding ketones 5 and 6 and the deprotection of the benzoate groups gave the symmetrical ketone 14, and the cyclic hemiketal 15, all in high yields. -

Supporting Information for “Conjunctive Functionalization of Vinyl Boronate
Electronic Supplementary Material (ESI) for ChemComm. This journal is © The Royal Society of Chemistry 2017 Supporting Information for “Conjunctive Functionalization of Vinyl Boronate Complexes with Electrophiles: A Diastereoselective Three-Component Coupling” Roly J. Armstrong, Christopher Sandford, Cristina García-Ruiz and Varinder K. Aggarwal Contents 1. General Information S2 2. Optimization S4 2.1. Solvent Screen for Synthesis of 25 S4 2.2. Solvent Screen for Synthesis of 29 S5 3. General Procedure for Conjunctive Three Component Coupling S6 4. Experimental Procedures S7 4.1. Starting Material Synthesis S7 4.2. Conjunctive Three-Component Coupling S13 5. Conjunctive Coupling With an Aryldiazonium Salt S43 6. -Elimination Studies S44 7. X-ray crystallographic analysis of 2 S46 8. References S48 9. NMR spectra S49 S1 1. General Information Reactions were carried out in flame-dried glassware under an atmosphere of nitrogen unless stated otherwise. Room temperature refers to 20-25 °C. Temperatures of 0 °C were obtained using an ice/water bath. Temperatures of –78 °C were obtained using a dry ice/acetone bath. Temperatures of –45 °C were obtained using a dry ice/acetonitrile bath. Reflux conditions were obtained using an oil bath equipped with a contact thermometer. Dichloromethane, diethyl ether and tetrahydrofuran were purified by filtration through activated alumina columns employing the method of Grubbs et al.1 All other solvents were used as supplied without prior purification. n-Butyllithium was purchased from Acros Organics as a -

(12) United States Patent (10) Patent No.: US 6,204,405 B1 Brown (45) Date of Patent: Mar
USOO6204405B1 (12) United States Patent (10) Patent No.: US 6,204,405 B1 Brown (45) Date of Patent: Mar. 20, 2001 (54) ECONOMICAL AND CONVENIENT (56) References Cited PROCEDURES FOR THE SYNTHESIS OF CATECHOLBORANE U.S. PATENT DOCUMENTS 4,739,096 4/1988 Noth et al. ........................... 558/288 (75) Inventor: Fisher C. Brown, West Lafayette, IN 5,973,185 10/1999 Ma et al. ............................. 558/288 (73) Assignee: Sigma-Aldrich Co. sk cited- by examiner (*) Notice: Subject to any disclaimer, the term of this Primary Examiner Deborah C. Lambkin patent is extended or adjusted under 35 U.S.C. 154(b) by 0 days. (57) ABSTRACT New, economical and convenient procedures for the prepa (21) Appl. No.: 09/469,274 ration of catecholborane in high chemically pure form using 1-1. tri-O-phenylene bis borate readily prepared from the reac (22) Filed: Dec. 22, 1999 tion of catechol with boric acid, and diborane or borane (51) Int. Cl." .................................................... C07F 5/04 Lewis base complexes. (52) U.S. Cl. .............................................................. 558/288 (58) Field of Search ............................................... 558/288 8 Claims, No Drawings US 6,204,405 B1 1 2 ECONOMICAL AND CONVENIENT rants more economic and convenient options for the Syn PROCEDURES FOR THE SYNTHESIS OF thesis of this important borane reagent, with increased CATECHOLBORANE chemical purity. The present invention provides new, eco nomical and convenient procedures for the preparation of This invention relates to new, economical and convenient catecholborane. procedures for the preparation of catecholborane. These SUMMARY OF THE DISCLOSURE processes provide catecholborane in high yields and chemi New, economical and convenient procedures for the cally pure form. -
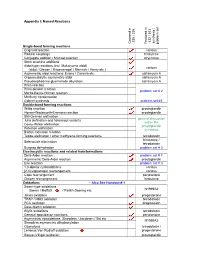
Appendix I: Named Reactions Single-Bond Forming Reactions Co
Appendix I: Named Reactions 235 / 335 432 / 533 synthesis / / synthesis Covered in Covered Featured in problem set problem Single-bond forming reactions Grignard reaction various Radical couplings hirstutene Conjugate addition / Michael reaction strychnine Stork enamine additions Aldol-type reactions (incl. Mukaiyama aldol) various (aldol / Claisen / Knoevenagel / Mannich / Henry etc.) Asymmetric aldol reactions: Evans / Carreira etc. saframycin A Organocatalytic asymmetric aldol saframycin A Pseudoephedrine glycinamide alkylation saframycin A Prins reaction Prins-pinacol reaction problem set # 2 Morita-Baylis-Hillman reaction McMurry condensation Gabriel synthesis problem set #3 Double-bond forming reactions Wittig reaction prostaglandin Horner-Wadsworth-Emmons reaction prostaglandin Still-Gennari olefination general discussion Julia olefination and heteroaryl variants within the Corey-Winter olefination prostaglandin Peterson olefination synthesis Barton extrusion reaction Tebbe olefination / other methylene-forming reactions tetrodotoxin hirstutene / Selenoxide elimination tetrodotoxin Burgess dehydration problem set # 3 Electrocyclic reactions and related transformations Diels-Alder reaction problem set # 1 Asymmetric Diels-Alder reaction prostaglandin Ene reaction problem set # 3 1,3-dipolar cycloadditions various [2,3] sigmatropic rearrangement various Cope rearrangement periplanone Claisen rearrangement hirstutene Oxidations – Also See Handout # 1 Swern-type oxidations (Swern / Moffatt / Parikh-Doering etc. N1999A2 Jones oxidation -

Manipulating Selectivity and Reactivity in Palladium-Catalyzed Oxidation Reactions
Manipulating Selectivity and Reactivity in Palladium-Catalyzed Oxidation Reactions Thesis by Zachary Kimble Wickens In Partial Fulfillment of the Requirements for the degree of Doctor of Philosophy CALIFORNIA INSTITUTE OF TECHNOLOGY Pasadena, California 2015 (Defended June 2nd, 2015) © 2015 Zachary Kimble Wickens All Rights Reserved 1 Dedicated to my parents for never pushing me to become a scientist 2 Acknowledgments The work I have the pleasure of sharing with you in this thesis would have been completely impossible without the help (both emotional and intellectual) of a tremendously large number of people. First, I want to thank my advisor, Bob Grubbs. I truly could not have asked for a more incredible thesis advisor. Since the beginning of my time at Caltech, you have subtly guided me towards the water but never forced me to drink. You have given me independence in choosing which projects to pursue and the directions these projects go, but always provided suggestions and hints along the way that have kept me on track. I know you will continue to provide great advice and strong support even long after I've left Caltech. Thanks for being my invisible safety net over the last five years. I would have never even considered pursuing a Ph.D in chemistry in the first place if it weren't for the fantastic chemistry faculty at Macalester College. Rebecca Hoye, Ronald Brisbois, Paul Fischer, Keith Kuwata, Thomas Varberg and Katherine Splan, you were all tremendously influential in my life. You taught me how to see the exciting puzzles that are scattered throughout the field of chemistry. -

Studies in Directed Catalytic Asymmetric Hydroboration of 1,2-Disubstituted Unsaturated Amide
University of Nebraska - Lincoln DigitalCommons@University of Nebraska - Lincoln Student Research Projects, Dissertations, and Chemistry, Department of Theses - Chemistry Department Summer 7-2017 STUDIES IN DIRECTED CATALYTIC ASYMMETRIC HYDROBORATION OF 1,2-DISUBSTITUTED UNSATURATED AMIDE Shuyang Zhang University of Nebraska - Lincoln, [email protected] Follow this and additional works at: http://digitalcommons.unl.edu/chemistrydiss Part of the Organic Chemistry Commons Zhang, Shuyang, "STUDIES IN DIRECTED CATALYTIC ASYMMETRIC HYDROBORATION OF 1,2-DISUBSTITUTED UNSATURATED AMIDE" (2017). Student Research Projects, Dissertations, and Theses - Chemistry Department. 85. http://digitalcommons.unl.edu/chemistrydiss/85 This Article is brought to you for free and open access by the Chemistry, Department of at DigitalCommons@University of Nebraska - Lincoln. It has been accepted for inclusion in Student Research Projects, Dissertations, and Theses - Chemistry Department by an authorized administrator of DigitalCommons@University of Nebraska - Lincoln. STUDIES IN DIRECTED CATALYTIC ASYMMETRIC HYDROBORATION OF 1,2- DISUBSTITUTED UNSATURATED AMIDE by Shuyang Zhang A THESIS Presented to the Faculty of The Graduate College at the University of Nebraska In Partial Fulfillment of Requirements For the Degree of Master of Science Major: Chemistry Under the Supervision of Professor James M. Takacs Lincoln, Nebraska July 2017 DIRECTED CATALYTIC ASYMMETRIC HYDROBORATION OF 1,2- DISUBSTITUTED ALKENES Shuyang Zhang, M.S. University of Nebraska 2017 Advisor: Professor James M. Takacs The Rh-catalyzed, substrate directed catalytic asymmetric hydroboration of γ,δ-unsaturated amides provides a direct route to enantioenriched acyclic secondary γ- borylated carbonyl derivatives with high regio- and enantioselectivity. The catalytic condition optimization and substrate scope study is discussed, including the effects in catalytical asymmetric hydroboration on pre-installed chiral γ,δ-unsaturated amides. -

Synthetic Applications of Organoboranes
ú 7lt) f SYNTHETIC APPLICATIONS OF ORGANOBORANES A Thesis Presented for the Degree of Doctor of Philosophy in The Uníversity of Adelaide by Roger Murphy, B.Sc. (Hons.) Department of Organíc Chemistry L976. CONTENTS Page SUMMARY (f) STATEMENT (fií) PUBL ICATIONS (iv) ACKNOI^JLEDGEMENTS (v) CHAPTER 1. Synthesis of Products Isolated from the Hydroboration-Cyanidation of Unsaturated Terpenes. / 1.1 Introduction. 1 L.2 Synthesis of Products Derived from L2 Geraniol. 1.3 Synthesis of Products Derived from L7 LÍnalyl AceËate. L.4 SynthesÍs of Products Derived from 22 Myrcene. CHAPTER 2. Cyanidation of Dialkyìboranes and Borinic Acids and Esters. 2.L Introduction. 43 2.2 Cyanidation of Dialkylboranes. 50 2.3 Cyanidation of BorÍnic Acíds and EsËers. 65 CHAPTER 3. Silver(I) 0xidation of Organoboranes. 3.1 Introduction. 70 3.2 Cycl-izatlon of Díenes vía Intramolecular 76 Alkyl Coupling. 3.3 AÈtenpted Reduction of Intemedl-ates 84 Obtaíned by Reaction of Organoboranes with Alkalíne SÍ1ver nltrate. CHAPTER 4. Asymmetric Induction by Hydroboration with Optical ly Active Dial llyl boranes. 4.L Introductlon. 88 4.2 AËterpted Resolution of (t)-414,6- 96 Trfnethylcaprolactam and (t)-4 16 16- Tr lmethylcapro lactam. 4.3 Attempted Resolutíon of (1)-31515- 100 Trinethylcyclohexanone . 4.4 Asynrmetric Hydroboratfon of 6r7-Dihydro- 110 416 r6-trTmethyl-5H-azepínone and 3r5,5- TrirneËhylcyclohex-2-enone (Isophorone) . CHAPTER 5. Experimental. 5.1 General. 118 5.2 I^Iork described in chapter 1. L2T 5.3 Ilork described in chapter 2. 163 5.4 Work descríbed ín chapËer 3. 178 5.5 lJork described Ín chapter 4.