The Origin of Neutron Radiation
Total Page:16
File Type:pdf, Size:1020Kb
Load more
Recommended publications
-

Neutron Interactions and Dosimetry Outline Introduction Tissue
Outline • Neutron dosimetry Neutron Interactions and – Thermal neutrons Dosimetry – Intermediate-energy neutrons – Fast neutrons Chapter 16 • Sources of neutrons • Mixed field dosimetry, paired dosimeters F.A. Attix, Introduction to Radiological • Rem meters Physics and Radiation Dosimetry Introduction Tissue composition • Consider neutron interactions with the majority tissue elements H, O, C, and N, and the resulting absorbed dose • Because of the short ranges of the secondary charged particles that are produced in such interactions, CPE is usually well approximated • Since no bremsstrahlung x-rays are generated, the • The ICRU composition for muscle has been assumed in absorbed dose can be assumed to be equal to the most cases for neutron-dose calculations, lumping the kerma at any point in neutron fields at least up to 1.1% of “other” minor elements together with oxygen to an energy E ~ 20 MeV make a simple four-element (H, O, C, N) composition Neutron kinetic energy Neutron kinetic energy • Neutron fields are divided into three • Thermal neutrons, by definition, have the most probable categories based on their kinetic energy: kinetic energy E=kT=0.025eV at T=20C – Thermal (E<0.5 eV) • Neutrons up to 0.5eV are considered “thermal” due to simplicity of experimental test after they emerge from – Intermediate-energy (0.5 eV<E<10 keV) moderator material – Fast (E>10 keV) • Cadmium ratio test: • Differ by their primary interactions in tissue – Gold foil can be activated through 197Au(n,)198Au interaction and resulting biological effects -

Neutron Stars
Chandra X-Ray Observatory X-Ray Astronomy Field Guide Neutron Stars Ordinary matter, or the stuff we and everything around us is made of, consists largely of empty space. Even a rock is mostly empty space. This is because matter is made of atoms. An atom is a cloud of electrons orbiting around a nucleus composed of protons and neutrons. The nucleus contains more than 99.9 percent of the mass of an atom, yet it has a diameter of only 1/100,000 that of the electron cloud. The electrons themselves take up little space, but the pattern of their orbit defines the size of the atom, which is therefore 99.9999999999999% Chandra Image of Vela Pulsar open space! (NASA/PSU/G.Pavlov et al. What we perceive as painfully solid when we bump against a rock is really a hurly-burly of electrons moving through empty space so fast that we can't see—or feel—the emptiness. What would matter look like if it weren't empty, if we could crush the electron cloud down to the size of the nucleus? Suppose we could generate a force strong enough to crush all the emptiness out of a rock roughly the size of a football stadium. The rock would be squeezed down to the size of a grain of sand and would still weigh 4 million tons! Such extreme forces occur in nature when the central part of a massive star collapses to form a neutron star. The atoms are crushed completely, and the electrons are jammed inside the protons to form a star composed almost entirely of neutrons. -
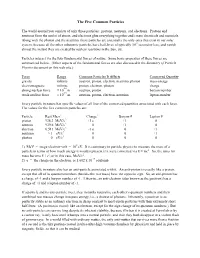
The Five Common Particles
The Five Common Particles The world around you consists of only three particles: protons, neutrons, and electrons. Protons and neutrons form the nuclei of atoms, and electrons glue everything together and create chemicals and materials. Along with the photon and the neutrino, these particles are essentially the only ones that exist in our solar system, because all the other subatomic particles have half-lives of typically 10-9 second or less, and vanish almost the instant they are created by nuclear reactions in the Sun, etc. Particles interact via the four fundamental forces of nature. Some basic properties of these forces are summarized below. (Other aspects of the fundamental forces are also discussed in the Summary of Particle Physics document on this web site.) Force Range Common Particles It Affects Conserved Quantity gravity infinite neutron, proton, electron, neutrino, photon mass-energy electromagnetic infinite proton, electron, photon charge -14 strong nuclear force ≈ 10 m neutron, proton baryon number -15 weak nuclear force ≈ 10 m neutron, proton, electron, neutrino lepton number Every particle in nature has specific values of all four of the conserved quantities associated with each force. The values for the five common particles are: Particle Rest Mass1 Charge2 Baryon # Lepton # proton 938.3 MeV/c2 +1 e +1 0 neutron 939.6 MeV/c2 0 +1 0 electron 0.511 MeV/c2 -1 e 0 +1 neutrino ≈ 1 eV/c2 0 0 +1 photon 0 eV/c2 0 0 0 1) MeV = mega-electron-volt = 106 eV. It is customary in particle physics to measure the mass of a particle in terms of how much energy it would represent if it were converted via E = mc2. -

Photons from 252Cf and 241Am-Be Neutron Sources H
Neutronenbestrahlungsraum Calibration Laboratory LB6411 Am-Be Quelle [email protected] Photons from 252Cf and 241Am-Be neutron sources H. Hoedlmoser, M. Boschung, K. Meier and S. Mayer Paul Scherrer Institut, CH-5232 Villigen PSI, Switzerland At the accredited PSI calibration laboratory neutron reference fields traceable to the standards of the Physikalisch-Technische Bundesanstalt (PTB) in Germany are available for the calibration of ambient and personal dose equivalent (rate) meters and passive dosimeters. The photon contribution to H*(10) in the neutron fields of the 252Cf and 241Am-Be sources was measured using various photon dose rate meters and active and passive dosimeters. Measuring photons from a neutron source involves considerable uncertainties due to the presence of neutrons, due to a non-zero neutron sensitivity of the photon detector and due to the energy response of the photon detectors. Therefore eight independent detectors and methods were used to obtain a reliable estimate for the photon contribution as an average of the individual methods. For the 241Am-Be source a photon contribution of approximately 4.9% was determined and for the 252Cf source a contribution of 3.6%. 1) Photon detectors 2) Photons from 252Cf and 241Am-Be neutron sources Figure 1 252Cf decays through -emission (~97%) and through spontaneous fission (~3%) with a half-life of 2.65 y. Both processes are accompanied by photon radiation. Furthermore the spectrum of fission products contains radioactive elements that produce additional gamma photons. In the 241Am-Be source 241Am decays through -emission and various low energy emissions: 241Am237Np + + with a half life of 432.6 y. -

MASTER 9700 South Cass Avenue Argonne, Illinois 60439 USA
ANL/KDH—SO DE84 001440 ANL/NDM-80 NEUTRON TOTAL CROSS SECTION MEASUREMENTS IN THE ENERGY REGION FROM 47 key to 20 MeV* by W. P. Poenitz and J. F. Whalon Applied Physics Division May, 1983 *This work supported by the U.S. Department of Energy Argonne National Laboratory MASTER 9700 South Cass Avenue Argonne, Illinois 60439 USA fflSTRtBtfUOU OF miS DOCUMENT IS UNLIMITED NUCLEAR DATA AMD MEASUREMENTS SERIES The Nuclear Data and Measurements Series presents results of studies in the field of microscopic nuclear data. The primary objective is the dissemination of information in the comprehensive form required for nuclear technology applications. This Series is devoted to: a) measured microscopic nuclear parameters, b) experimental techniques and facilities employed in measurements, c) the analysis, correlation and interpretation of nuclear data, and d) the evaluation of nuclear data. Contributions to this Series are reviewed to assure technical competence and, unless otherwise stated, the contents can be formally referenced. This Series does not supplant formal journal publication but it does provide the more extensive informa- tion required for technological applications (e.g., tabulated numerical data) in a timely manner. DISCLAIMER This report was prepared as an account of work sponsored by an agency of the United States Government. Neither the United States Government nor any agency thereof, nor any of their employees, makes any warranty, express or implied, or assumes any legal liability or retpoosi- bility for the accuracy, completeness, or usefulness of any information, apparatus, product, or process disclosed, or represents that its use would not infringe privately owned rights. Refer- ence herein to any specific commercial product, process, or service by trade name, trademark, manufacturer, or otherwise does not necessarily constitute or imply its endorsement, recom- mendation, or favoring by the United States Government or any agency thereof. -

Neutron Activation and Prompt Gamma Intensity in Ar/CO $ {2} $-Filled Neutron Detectors at the European Spallation Source
Neutron activation and prompt gamma intensity in Ar/CO2-filled neutron detectors at the European Spallation Source E. Diana,b,c,∗, K. Kanakib, R. J. Hall-Wiltonb,d, P. Zagyvaia,c, Sz. Czifrusc aHungarian Academy of Sciences, Centre for Energy Research, 1525 Budapest 114., P.O. Box 49., Hungary bEuropean Spallation Source ESS ERIC, P.O Box 176, SE-221 00 Lund, Sweden cBudapest University of Technology and Economics, Institute of Nuclear Techniques, 1111 Budapest, M}uegyetem rakpart 9. dMid-Sweden University, SE-851 70 Sundsvall, Sweden Abstract Monte Carlo simulations using MCNP6.1 were performed to study the effect of neutron activation in Ar/CO2 neutron detector counting gas. A general MCNP model was built and validated with simple analytical calculations. Simulations and calculations agree that only the 40Ar activation can have a considerable effect. It was shown that neither the prompt gamma intensity from the 40Ar neutron capture nor the produced 41Ar activity have an impact in terms of gamma dose rate around the detector and background level. Keywords: ESS, neutron detector, B4C, neutron activation, 41Ar, MCNP, Monte Carlo simulation 1. Introduction Ar/CO2 is a widely applied detector counting gas, with long history in ra- diation detection. Nowadays, the application of Ar/CO2-filled detectors is ex- tended in the field of neutron detection as well. However, the exposure of arXiv:1701.08117v2 [physics.ins-det] 16 Jun 2017 Ar/CO2 counting gas to neutron radiation carries the risk of neutron activa- tion. Therefore, detailed consideration of the effect and amount of neutron ∗Corresponding author Email address: [email protected] (E. -

Discovery of the Isotopes with Z<= 10
Discovery of the Isotopes with Z ≤ 10 M. Thoennessen∗ National Superconducting Cyclotron Laboratory and Department of Physics and Astronomy, Michigan State University, East Lansing, MI 48824, USA Abstract A total of 126 isotopes with Z ≤ 10 have been identified to date. The discovery of these isotopes which includes the observation of unbound nuclei, is discussed. For each isotope a brief summary of the first refereed publication, including the production and identification method, is presented. arXiv:1009.2737v1 [nucl-ex] 14 Sep 2010 ∗Corresponding author. Email address: [email protected] (M. Thoennessen) Preprint submitted to Atomic Data and Nuclear Data Tables May 29, 2018 Contents 1. Introduction . 2 2. Discovery of Isotopes with Z ≤ 10........................................................................ 2 2.1. Z=0 ........................................................................................... 3 2.2. Hydrogen . 5 2.3. Helium .......................................................................................... 7 2.4. Lithium ......................................................................................... 9 2.5. Beryllium . 11 2.6. Boron ........................................................................................... 13 2.7. Carbon.......................................................................................... 15 2.8. Nitrogen . 18 2.9. Oxygen.......................................................................................... 21 2.10. Fluorine . 24 2.11. Neon........................................................................................... -

TUTORIAL on NEUTRON PHYSICS in DOSIMETRY S. Pomp1
TUTORIAL ON NEUTRON PHYSICS IN DOSIMETRY S. Pomp1,* 1 Department of physics and astronomy, Uppsala University, Box 516, 751 20 Uppsala, Sweden. *Corresponding author. E‐mail address: [email protected] (S.Pomp) Abstract: Almost since the time of the discovery of the neutron more than 70 years ago, efforts have been made to understand the effects of neutron radiation on tissue and, eventually, to use neutrons for cancer treatment. In contrast to charged particle or photon radiations which directly lead to release of electrons, neutrons interact with the nucleus and induce emission of several different types of charged particles such as protons, alpha particles or heavier ions. Therefore, a fundamental understanding of the neutron‐nucleus interaction is necessary for dose calculations and treatment planning with the needed accuracy. We will discuss the concepts of dose and kerma, neutron‐nucleus interactions and have a brief look at nuclear data needs and experimental facilities and set‐ups where such data are measured. Keywords: Neutron physics; nuclear reactions; kerma coefficients; neutron beams; Introduction Dosimetry is concerned with the ability to determine the absorbed dose in matter and tissue resulting from exposure to directly and indirectly ionizing radiation. The absorbed dose is a measure of the energy deposited per unit mass in the medium by ionizing radiation and is measured in Gray, Gy, where 1 Gy = 1 J/kg. Radiobiology then uses information about dose to assess the risks and gains. A risk is increased probability to develop cancer due to exposure to a certain dose. A gain is exposure of a cancer tumour to a certain dose in order to cure it. -

Photofission Cross Sections of 238U and 235U from 5.0 Mev to 8.0 Mev Robert Andrew Anderl Iowa State University
Iowa State University Capstones, Theses and Retrospective Theses and Dissertations Dissertations 1972 Photofission cross sections of 238U and 235U from 5.0 MeV to 8.0 MeV Robert Andrew Anderl Iowa State University Follow this and additional works at: https://lib.dr.iastate.edu/rtd Part of the Nuclear Commons, and the Oil, Gas, and Energy Commons Recommended Citation Anderl, Robert Andrew, "Photofission cross sections of 238U and 235U from 5.0 MeV to 8.0 MeV " (1972). Retrospective Theses and Dissertations. 4715. https://lib.dr.iastate.edu/rtd/4715 This Dissertation is brought to you for free and open access by the Iowa State University Capstones, Theses and Dissertations at Iowa State University Digital Repository. It has been accepted for inclusion in Retrospective Theses and Dissertations by an authorized administrator of Iowa State University Digital Repository. For more information, please contact [email protected]. INFORMATION TO USERS This dissertation was produced from a microfilm copy of the original document. While the most advanced technological means to photograph and reproduce this document have been used, the quality is heavily dependent upon the quality of the original submitted. The following explanation of techniques is provided to help you understand markings or patterns which may appear on this reproduction, 1. The sign or "target" for pages apparently lacking from the document photographed is "Missing Page(s)". If it was possible to obtain the missing page(s) or section, they are spliced into the film along with adjacent pages. This may have necessitated cutting thru an image and duplicating adjacent pages to insure you complete continuity, 2. -

Opportunities for Neutrino Physics at the Spallation Neutron Source (SNS)
Opportunities for Neutrino Physics at the Spallation Neutron Source (SNS) Paper submitted for the 2008 Carolina International Symposium on Neutrino Physics Yu Efremenko1,2 and W R Hix2,1 1University of Tennessee, Knoxville TN 37919, USA 2Oak Ridge National Laboratory, Oak Ridge TN 37981, USA E-mail: [email protected] Abstract. In this paper we discuss opportunities for a neutrino program at the Spallation Neutrons Source (SNS) being commissioning at ORNL. Possible investigations can include study of neutrino-nuclear cross sections in the energy rage important for supernova dynamics and neutrino nucleosynthesis, search for neutrino-nucleus coherent scattering, and various tests of the standard model of electro-weak interactions. 1. Introduction It seems that only yesterday we gathered together here at Columbia for the first Carolina Neutrino Symposium on Neutrino Physics. To my great astonishment I realized it was already eight years ago. However by looking back we can see that enormous progress has been achieved in the field of neutrino science since that first meeting. Eight years ago we did not know which region of mixing parameters (SMA. LMA, LOW, Vac) [1] would explain the solar neutrino deficit. We did not know whether this deficit is due to neutrino oscillations or some other even more exotic phenomena, like neutrinos decay [2], or due to the some other effects [3]. Hints of neutrino oscillation of atmospheric neutrinos had not been confirmed in accelerator experiments. Double beta decay collaborations were just starting to think about experiments with sensitive masses of hundreds of kilograms. Eight years ago, very few considered that neutrinos can be used as a tool to study the Earth interior [4] or for non- proliferation [5]. -
R-Process: Observations, Theory, Experiment
r-process: observations, theory, experiment H. Schatz Michigan State University National Superconducting Cyclotron Laboratory Joint Institute for Nuclear Astrophysics 1. Observations: do we need s,r,p-process and LEPP? 2. r-process (and LEPP?) models 3. r-process experiments SNR 0103-72.6 Credit: NASA/CXC/PSU/S.Park et al. Origin of the heavy elements in the solar system s-process: secondary • nuclei can be studied Æ reliable calculations • site identified • understood? Not quite … r-process: primary • most nuclei out of reach • site unknown p-process: secondary (except for νp-process) Æ Look for metal poor`stars (Pagel, Fig 6.8) To learn about the r-process Heavy elements in Metal Poor Halo Stars CS22892-052 (Sneden et al. 2003, Cowan) 2 1 + solar r CS 22892-052 ) H / X CS22892-052 ( g o red (K) giant oldl stars - formed before e located in halo Galaxyc was mixed n distance: 4.7 kpc theya preserve local d mass ~0.8 M_sol n pollutionu from individual b [Fe/H]= −3.0 nucleosynthesisa events [Dy/Fe]= +1.7 recall: element number[X/Y]=log(X/Y)-log(X/Y)solar What does it mean: for heavy r-process? For light r-process? • stellar abundances show r-process • process is not universal • process is universal • or second process exists (not visible in this star) Conclusions depend on s-process Look at residuals: Star – solar r Solar – s-process – p-process s-processSimmerer from Simmerer (Cowan et etal.) al. /Lodders (Cowan et al.) s-processTravaglio/Lodders from Travaglio et al. -0.50 -0.50 -1.00 -1.00 -1.50 -1.50 log e log e -2.00 -2.00 -2.50 -2.50 30 40 50 60 70 80 90 30 40 50 60 70 80 90 Element number Element number ÆÆNeedNeed reliable reliable s-process s-process (models (models and and nu nuclearclear data, data, incl. -

Uses of Isotopic Neutron Sources in Elemental Analysis Applications
EG0600081 3rd Conference on Nuclear & Particle Physics (NUPPAC 01) 20 - 24 Oct., 2001 Cairo, Egypt USES OF ISOTOPIC NEUTRON SOURCES IN ELEMENTAL ANALYSIS APPLICATIONS A. M. Hassan Department of Reactor Physics Reactors Division, Nuclear Research Centre, Atomic Energy Authority. Cairo-Egypt. ABSTRACT The extensive development and applications on the uses of isotopic neutron in the field of elemental analysis of complex samples are largely occurred within the past 30 years. Such sources are used extensively to measure instantaneously, simultaneously and nondestruclively, the major, minor and trace elements in different materials. The low residual activity, bulk sample analysis and high accuracy for short lived elements are improved. Also, the portable isotopic neutron sources, offer a wide range of industrial and field applications. In this talk, a review on the theoretical basis and design considerations of different facilities using several isotopic neutron sources for elemental analysis of different materials is given. INTRODUCTION In principle there are two ways to use neutrons for elemental and isotopic abundance analysis in samples. One is the neutron activation analysis which we call it the "off-line" where the neutron - induced radioactivity is observed after the end of irradiation. The other one we call it the "on-line" where the capture gamma-rays is observed during the neutron bombardment. Actually, the sequence of events occurring during the most common type of nuclear reaction used in this analysis namely the neutron capture or (n, gamma) reaction, is well known for the people working in this field. The neutron interacts with the target nucleus via a non-elastic collision, a compound nucleus forms in an excited state.